You are what you eat, right? Why? Well, this is a complicated science! One big factor is the gut microbiome.
In this article, we will review:
- The structure of the human gut microbiome
- The relationship between the gut and behavior
- The history of microbiome studies
- What behavioral research has shown about the gut microbiome in rodents, and
- How the gut microbiome affects behavior in humans.
So, the first half of this article introduces you to the gut microbiome, including some key things you must know in order to understand its role in behavior.
The second half explores what research has shown about the relationship between behavior and the gut microbiome.
Part 1: The Human Gut Microbiome
What is the Microbiome?
Did you know that the human body has 10 times more microbial cells than it has human body cells? In fact, the human gut has 3.3 million unique microbial genes, amounting to 150 times more genes than that of the human genome. That means that most of the genetic material humans host is for microbial cells, not human cells.
The human gut hosts about 1000 bacterial species with the majority belong to the Bacteroides and Firmicutes families.[1]
Gut bacteria are crucial for regulating and maintaining optimal gut metabolism, but are also involved in the immune system and can affect behavior.
Gut Microbiome Bacteria
Commonly found bacteria in the human gut microbiome include:
- Prevotella family
- Ruminococcus family
- Bacteroides family
- Firmicutes family
Firmicutes and Bacteroides phyla account for at least ¾ of the human microbiome.[2]
Furthermore, in the colon, a low oxygen environment that is attractive to anaerobic bacteria, the following can be found:
- Peptostreptococcus
- Bifidobacterium
- Lactobacillus
- Clostridium[3]
These microbes are beneficial to the human organism because it is theorized that they inhibit potentially harmful bacteria from growing. This is accomplished by competing for the same attachment sites of the gut’s mucous membranes and nutrients.[4][5]
One of the challenges in studying the gut microbiome is the level of variability that exists between the distribution of bacteria from person to person, making it difficult to compare microbiome profiles across individuals.
In the next subsection, we cover some of the factors that contribute to the variability observed in the human microbiome.
Variation and Plasticity of Human Microbiome
While humans are about 99.9% similar in terms of their genome, they can be 80-90% different when it comes to their gut microbiome.[6][7] The following factors influence gut microbiome variation across individuals:
- Host immunity and environment. The human microbiome depends a lot on the host, its genotype, disease status, lifestyle such as dietary choices, the immune system, and the immediate environment.[8]
- Gut plasticity. The gut microbiome doesn’t only vary from person to person. It also varies on an individual level across time, a phenomenon known as gut plasticity. Gut plasticity is another challenging aspect of quantifying the bacteria in the human gut microbiome since it introduces a time component.[9]
When it comes to studies using rodents, some of these factors are largely controlled for since the rodents may be genetically engineered and/or kept under the same environmental conditions, thus limiting the number of variables available to influence the gut microbiome.
The Gut and Human Disease
The gut microbiome is an important factor to study and consider when investigating the pathology and potential treatment of human disease. Increasingly, preclinical studies are showing that changes in the bidirectional brain-gut-microbiome interactions are associated with many diseases, including: functional gastrointestinal disorders like irritable bowel syndrome (IBS)[10][11] and neurologic diseases like affective disorders, Parkinson’s disease,[12] multiple sclerosis,[13] chronic pain,[14] and autism spectrum disorders.[15]
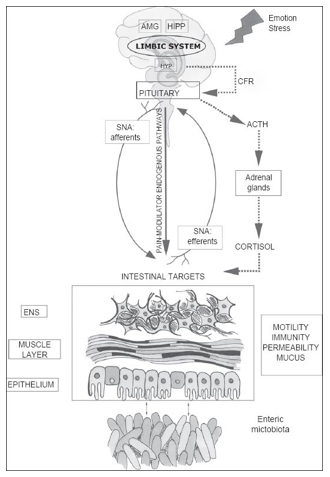
The Brain-Gut Axis: How the gut influences the brain
The brain-gut axis (also known as the ‘gut-brain axis’) refers to complex interrelationship and communication system between the brain and the gut. The brain-gut axis links the brain’s cognitive and emotional centers with intestinal mechanisms and functions such as: enteric reflex, intestinal permeability, immune activation, and entero-endocrine signaling.[16]
This physiological link is important for treating functional gastrointestinal disorders. However, the connection between the brain and the gut is especially important for treating neurologic and psychiatric diseases, especially since it may be somehow implicated in disease pathology.
Preclinical studies have found that the gut is associated with neurochemical alterations such as changes in the:
- Levels of brain-derived neurotrophic factor (BDNF) in the cortical and hippocampal brain regions[17][18]
- Expression of hippocampal serotonin (5-HT) 1A receptors[18]
- Turnover of striatal monoamine[19]
- Expression of synaptic plasticity genes[19]
Also, the gut has been linked to the following
- Stress responsiveness[20][21]
- Anxiety-like behaviors[18][19][22]
- Depression-like behaviors [23][24][25]
- Nociceptive response[14][16]
These findings demonstrate the diverse and powerful influence that the microbiome has over the central nervous system and behavior.
Pathways of Communication
In order to affect behavior, the gut microbiome must be able to communicate across different body systems. The following are characteristics of the gut microbiome’s pathways of communication:
- A bidirectional communication between the gut and brain exists and has been recognized for a long time.
- The brain and gut communicate through pathways within the following systems:
- The autonomic nervous system’s (ANS) sympathetic and parasympathetic nervous systems
- The enteric nervous system (ENS)
- The immune system[27]
These channels are interactive and work in parallel through various signaling mechanisms.[28]
Experiments on microbiota and behavior also quantify intestinal or gut permeability, the ability of the gut to be like a barrier. Gut permeability refers to how much control the gut has over materials passing through the gut wall into the rest of the body. Increased gut permeability means that more substances, such as toxins, microorganisms, or foreign antigens, can move through the intestine. This is a threat to the rest of the body since these substances may be potentially harmful.
The History of the Human Gut Microbiome Studies
Although the microbiome was always there, it was not until recently that it gained such a strong foothold in neuroscience and health research.
Studies focusing on human microbiome diversity started with Antonie
van Leeuwenhoek, who, as early as the 1680s, began establishing differences between the microbiomes of sick and diseased individuals.[9] Present-day research employs molecular techniques in order to learn why and how there are differences in the microbiome between the healthy and diseased.
Due to the potential of this area of research, the shift towards personalized medicine, and the advancement of technology, gut microbiome studies are quickly increasing in number.
Part 2: The Gut Microbiome and Behavior
Here comes the part where we focus intensely on what research has to say about the link between the gut microbiome and behavior.
In the sections below, we take a look at what (and how) rodent studies have taught us so far about the relationship between behavior and the gut microbiome.
Rodent Studies on the Gut Microbiome and Behavior
Diets and Memory in C57BL/6 mice
A study by Magnusson et al. showed that there is a relationship between gut microbiome diet-related changes and cognitive flexibility.[29] The experiment made use of male C57BL/6 mice which were 2-months old. The mice were assigned randomly to one of the following diet conditions:
- High-fat diet (42% fat and 43% carbohydrate)
- High-sucrose (12% fat and 70% carbohydrates mostly sucrose)
- Normal chow (13% kcal fat and 62% carbohydrates)
The mice were then subjected to fecal microbiome analysis and Morris Water Maze testing.
The researchers found that:
- The gut microbiome was significantly more altered in the high-sucrose diet than in the normal chow and high-fat diets.
- A high-sucrose diet was associated with impaired cognitive flexibility and poor spatial memory.
- In the Morris Water Maze, both high-fat and high-sucrose groups focused on the former platform position when it was time to perform the reversal probe trials.
- Decreased cognitive flexibility was associated with an increase in gut Clostridiales and a decrease in gut Bacteroidales.
- A higher percent of Lactobacillales was correlated with poorer performance in the Morris Water Maze’s first probe trial.
Voluntary Exercise Attenuates the Microbiome
Exercise can attenuate the gut microbiome. A study by Choi et al. demonstrated how changes in the gut microbiome structure and composition, created by exposure to polychlorinated biphenyls (PCBs), are further affected by exercise.
The researchers used male C57BL/6 mice for this experiment. The mice were divided into two conditions:
- Activity condition: individual housing with a Running Wheel.
- Sedentary condition: individual housing with a locked/immobile running wheel.
The mice were observed for 5 weeks, meaning that they had a 5-week timeframe for voluntarily exercising in the active condition, while the sedentary mice had no choice but to remain inactive.
Then, all mice were subjected to 2 days of PCBs (150μmol/kg). Oral intake of PCBs was significantly associated with an altered gut microbiome, substantially decreasing the gut’s levels of Proteobacteria.[30]
However, these PCB-induced changes were modulated by a mouse’s exercise levels. A mouse’s activity levels were significantly correlated with the gut microbiome’s shift in biodiversity, composition, and abundance of bacteria.
Mice that had access to the running wheel, and thus exercised more, had higher levels of Proteobacteria than the sedentary mice.
The effects of exercise on gut physiology and disease pathology have also been reported in other research, including colon cancer,[31] irritable bowel syndrome, and inflammatory bowel diseases.[32]
Such findings demonstrate how behavior, particularly voluntary exercise, can influence the state of the gut microbiome which in turn positively influences health by somehow lowering toxicity.
Involuntary Exercise Possibly Detrimental To Gut Health
However, in contrast to the previous section’s point, not all forms of exercise and activity were created equal when it comes to gut microbiota. When exercise is forced or involuntary, it is thought to be detrimental to gut health.[33]
In a study by Cook et al., the effect of exercise on a mouse model of colitis (inflammation of the colon) was explored. Male C57BL/6 mice were subjected to the following conditions:
- Forced treadmill running: 40 minutes of 8-12m/min, 6 weeks, 5x/week
- Voluntary wheel running: 30-day access to available wheels in the home cage
- Sedentary: no exercise
after being induced with colitis through dextran sodium sulfate exposure via drinking water over 5 days.
While the researchers expected both exercise conditions to have improved colitis symptoms as a result of activity, the forced treadmill running group ended up having more negative outcomes. The voluntary wheel running condition was associated with reduced inflammatory gene expression in these mice while the reverse was true for the forced treadmill running group condition
Such findings indicate that involuntary exercise does not help re-establish the gut microbiome balance while voluntary exercise can.[34]
Lactobacillus rhamnosus Reduces Anxiety- and Depression Related Behavior
In this next example, we will see how a specific lactic acid bacteria, can influence behavior and γ-aminobutyric acid (GABA) activity. GABA receptor expression is involved in psychological and physiological processes and has been implicated in depression and anxiety. Both depression and anxiety are comorbid with functional bowel disorders, suggesting a link between the gut, behavior, and GABA receptors.
An experiment by Bravo et al. explored the link between Lactobacillus rhamnosus and behavior by orally gavaging naturally-anxious BALB/c mice with the bacteria. The control group was BALB/c mice given broth without bacteria. The following behavioral characteristics were discovered in mice given Lactobacillus rhamnosus:[35]
- Reduced anxiety. The Elevated-Plus Maze was used to assess the behavioral consequences associated with changes in GABA neurotransmission. Mice that ingested Lactobacillus rhamnosus had a greater number of entries into the Elevated-Plus Maze’s open arms (an entry into the open arms is defined by having all four paws in the open area). This suggests that the bacteria studied may have a possible anxiolytic effect.
- Reduced depressive-like behaviors. Mice fed with the probiotic Lactobacillus rhamnosus fared better off in a Forced Swim Test, an indication of lower anxiety, than control-fed mice.
- Interaction between time and fear-conditioning. An interesting finding was uncovered during the Fear-Conditioning Task. In this task, mice exposed to aversive stimuli on Day 1 are trained to associate a stimulus (light/tone) and a foot shock. If the association has been learned, the mouse is expected to demonstrate freezing behavior when it encounters the stimulus again the next day. Experimental findings showed that Lactobacillus rhamnosus mice performed the same as control mice on Day 1 (the learning phase) indicating no differences in learning ability. On Day 2 (the memory testing phase) the Lactobacillus rhamnosus mice outperformed the controls and had a significantly higher percentage of freezing behavior exhibited, indicating they learned the association well. Finally, on Day 3 (the extinction phase), there were no differences between the two groups.[35]
This experiment also took a look at the physiological effects and differences of the gut-brain axis between the mice given Lactobacillus rhamnosus and control bacteria-free broth and showed that:
- Treatment was significantly associated with GABA mRNA expression. The mice fed with Lactobacillus rhamnosus showed higher expression of GABAB1b mRNA in the cortical regions and a reduction in the amygdala, hippocampus and locus coeruleus. Furthermore, these mice had reduced GABAAα2 mRNA reductions in the prefrontal cortex and amygdala but increased levels in the hippocampus.
- Corticosterone levels were different between the two conditions. The Lactobacillus rhamnosus-treated mice had reduced stress-induced corticosterone levels. Since corticosterone is the ‘stress hormone’ and ultimately affects behavior, such a finding demonstrates a potential pathway between the microbiome and behavior.
- Anxiety- and depression-reducing effects are reduced through partial vagotomy. The anxiety- and depression-reducing effects (both the behavioral and neurochemical) were obliterated by the partial removal of the vagus nerve (vagotomy), a central communication nerve between the gut and brain. This suggests that the vagus nerve transmits or modulates information from gut bacteria to the brain. Thus, the effects of Lactobacillus rhamnosus ingestion on behavior were made possible due to this physiological link.[35]
This experiment demonstrates how the relationship between a single bacteria strain, the brain, and behavior can be tested and quantified.
Social Behavior Attenuated by Gut Microbiome
The gut microbiome can also influence social behavior. In a study conducted by Arentsen et al., germ-free mice were compared with specific-pathogen free mice used in order to explore the link between social behavior and microbiota.[36] These mouse strains have the following differences in their microbiota:
- Germ-free mice: Completely lack any microbiota. Thus, their skin, intestines, and respiratory tract are all free from bacteria and microorganisms. These mice lack natural immunity from common infections since they have no prior exposure to them.
- Specific-pathogen free mice: These mice lack certain microbiota. They are bred in order to be totally free from specific pathogens. In order for a mouse to be a specific-pathogen mouse is must get certified by passing certain lab tests. Commonly excluded bacteria include Clostridium piliforme and Salmonella spp, for example. However, there are some lab-to-lab variabilities in their production.[37]
In order to assess social behavior, germ-free mice were tested using a Social Three-Chambered Apparatus. When placed in this apparatus, mice have a choice of whether they want to interact with a stimulus mouse or a novel object. Mice that interact with an object are classified as being less social than those that interact with the stimulus mouse.
In the experiment, the researchers found that the germ-free mice spent significantly more time with the stimulus mouse than the object, indicating higher levels of sociability when compared with specific-pathogen free mice.
Such findings suggest that gut microbiota are involved in social behavior.
Offspring Behavior Affected by Gut Microbiome
In addition to affecting social behavior, the gut microbiome can also affect an offspring’s behavior based on what the maternal microbiota profile is like. A mouse’s sex modulates this effect.
Maternal obesity predisposes the offspring to certain neurodevelopmental and metabolic abnormalities. Although the mechanisms behind this remain unknown, there is a link between high-fat diets and altered gut microbiota.
In order to determine the link between diet, gut microbiota, and maternal/offspring behavior, an experiment by Bruce-Keller et al. made use of transplanted control low diet- or high-fat diet-associated gut microbiota into female mice. The female mice then bred and their behavior was observed, as well as their offsprings’ behavior.
As a result of the microbiota transplants, only offspring behavior was affected. More behavioral effects were observed in male than female offspring from mothers with a high-fat diet. The researchers observed that male offspring from dams with microbiota had:[38]
- Higher anxiety levels. The experimental mice showed a decrease in the amount of time spent in the inner zone of the Open Field Test, as well as a lower frequency of inner zone entries.
- Poor learning abilities. The male offspring from high-fat fed dams also had a lower percentage of time spent exhibiting freezing behavior in response to a stimulus tone in the Fear-Conditioning Task, indicating an inability to learn to associate an aversive stimuli with a protective behavior.
- Stereotypical behaviors. Increased percentage of marbles buried in the Marble Burying Test.
Both female and male neonatal offspring from dams with high-fat diet microbiota vocalized less when they experienced maternal separation as compared to the controls.
These findings suggest that offspring from high-fat diet dams have their behavior and cognition affected as a result of the mother’s diet.[38]
Antibiotic-treated Mice Have Higher Anxiety-like Behaviors
The administration of antibiotics changes gut microbiota and influences behavior. A study by Desbonnet et al. showed that when adolescent NIH Swiss mice are chronically given a cocktail of antibiotics (ampicillin at 1 mg/ml, vancomycin at 5mg/ml, neomycin at 10 mg/ml, metronidazole at 10 mg/ml and supplemented with amphotericin-B at 0.1 mg/ml), significantly lower levels of anxiety-like behaviors are performed but with other changes in cognition accompanying the treatment.[39]
Changes in cognition were indicated by the following behavioral changes when compared with controls:
- Lower discrimination index. Antibiotic-treated mice have altered non-spatial cognition and exploratory behaviors when assessed using the Novel Object Recognition Test. In the Novel Object Recognition Test, mice naturally express the tendency to interact more with a novel object than a familiar object. However, antibiotic-treated mice do not display this natural tendency, indicating that they fail to discriminate between objects.
- More time spent in brightly lit spaces. Aversion of brightly lit spaces comes as an instinct in mice, an evolutionary mechanism serving species preservation. When given a choice, mice will choose a darkly lit area over a brightly lit area. Mice given antibiotics spent more time in brightly-lit areas of the Light/Dark Box than controls, indicating lower anxiety levels.
These behavioral changes were accompanied by significant differences at the phylum level with antibiotic treatment showing:
- A decreased relative abundance of Firmicutes and Bacteroidetes
- An increased relative abundance of Proteobacteria and Cyanobacteria
Schizophrenic Behaviors and the Gut Microbiome
The gut microbiome is not implicated only in intestinal disorders, but it may also be a key component of the pathophysiology behind neuropsychiatric diseases like schizophrenia.
A study by Zheng et al. transplanted microbiota from schizophrenic patients into recipient germ-free mice. Then, the germ-free mice with now schizophrenic microbiota were behaviorally tested using a variety of measures. These mice were compared with the control germ-free mice that received transplanted microbiota from healthy control humans.[40] The following behavioral findings were established:
- Hyperactivity and reduced anxiety in the Open Field Test. The mice with schizophrenic microbiota demonstrated hyperactivity by traveling a greater amount of total distance. Also, they showed reduced anxiety levels when compared to the control germ-free mice and spent more time in the maze’s center region, away from the walls.
- Reduced immobility in the Forced Swim Test. Another behavioral difference observed in mice with microbiota from schizophrenic patients was a significant reduction in the duration of immobility. While the control mice spent about 50% of the time being immobile, the schizophrenic mice were immobile for roughly 20% of the observed time. A reduction of immobility indicates that the mice with schizophrenic microbiota were more active and had less depressive-like behavior.
Together, these results indicate that mice hosting schizophrenic patients’ microbiota have increased locomotor activity, but decreased depressive- and anxiety-like behaviors.
Gut Bacteria Across BTBR Mice Modeling Autism
Altered gut microbiota are also apparent in the pathogenesis of autism spectrum disorders (ASD)[41] A study by Coretti et al. established this by comparing BTBR T+tf/J (BTBR) inbred mice with naturally social healthy C57BL/6J mice.
BTBR displayed a significantly altered behavioral profile, they:
- Spent significantly less time interacting with a mouse in the Three-Chamber Social Interaction Test and spend more time in the empty zone of the apparatus. By comparing, C57BL/6 mice spent more time interacting with the mouse and less time in the empty zone.
- Buried more marbles in the Marble Burying Task, indicating higher levels of repetitive behavior than C57BL/6 mice.
- Had higher levels of self-grooming when compared to C57BL/6 mice, but female BTBR showed even higher levels of self-grooming than male BTBR mice, indicating an effect of gender.
BTBR mice had increased gut permeability, meaning that it was easier for materials and microorganisms like bacteria to travel through the intestine into the rest of the body.
In addition to finding strain differences between the mice modeling ASD and the healthy C57BL/6J mice, the researchers noted an effect of gender:
- Altered behavior in female BTBR is associated with an increase of Sutterella and Parabacteroides and a decrease of Oscillospira and Dehalobacterium when compared to female C57BL/6 mice.
- Male BTBR mice were found to have lower levels of Desulfovibrio, Dehalobacterium, and Ruminococcus when compared to male C57BL/6 mice.
Altered microbiota can also be observed in other mouse models of ASD, including valproic acid exposure and maternal immune activation. Although the relationship between human disease and animal models needs deeper scrutiny, some findings already exist showing that several Proteobacteria can be found in children with ASD and gastrointestinal disturbances when compared to control non-ASD children with gastrointestinal disturbances.[42]
Conclusion
As showcased by these findings, the gut microbiome has a strong hold on behavior due to its intricate ties with the central nervous system.
The gut microbiome varies from person to person, but also within an individual over time. Disease status, environment, and lifestyle habits all influence the microbiome, making it challenging to study.
However, due to the advancement of techniques (such as transplanting microbiota from humans into rodents) and the use of rodents, the relationship between the gut microbiome and disease/health status and behavior can be better understood.
The gut microbiome is gaining increasing attention by researchers due to its relevance to personalized medicine.[9] Future studies will continue to explore the mechanisms between the gut microbiome, disease, and behavior.
References
- Zhu, Baoli, Xin Wang, and Lanjuan Li. “Human gut microbiome: the second genome of human body.” Protein & cell 1.8 (2010): 718-725.
- Eckburg, Paul B., et al. “Diversity of the human intestinal microbial flora.” science 308.5728 (2005): 1635-1638.
- Arumugam, Manimozhiyan, et al. “Enterotypes of the human gut microbiome.” nature 473.7346 (2011): 174.
- Canny, Geraldine O., and Beth A. McCormick. “Bacteria in the intestine, helpful residents or enemies from within?.” Infection and immunity 76.8 (2008): 3360-3373.
- Jandhyala, Sai Manasa, et al. “Role of the normal gut microbiota.” World journal of gastroenterology: WJG 21.29 (2015): 8787.
- Fierer, Noah, et al. “The influence of sex, handedness, and washing on the diversity of hand surface bacteria.” Proceedings of the National Academy of Sciences 105.46 (2008): 17994-17999.
- Turnbaugh, Peter J., et al. “A core gut microbiome in obese and lean twins.” nature 457.7228 (2009): 480.
- Turnbaugh, Peter J., et al. “The human microbiome project.” Nature 449.7164 (2007): 804.
- Ursell, Luke K., et al. “Defining the human microbiome.” Nutrition reviews 70.suppl_1 (2012): S38-S44.
- Rhee, Sang H., Charalabos Pothoulakis, and Emeran A. Mayer. “Principles and clinical implications of the brain–gut–enteric microbiota axis.” Nature reviews Gastroenterology & hepatology 6.5 (2009): 306.
- Mayer, Emeran A. “Gut feelings: the emerging biology of gut–brain communication.” Nature Reviews Neuroscience 12.8 (2011): 453.
- Sampson, Timothy R., et al. “Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease.” Cell 167.6 (2016): 1469-1480.
- Berer, Kerstin, et al. “Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination.” Nature 479.7374 (2011): 538.
- Amaral, F. A., et al. “Commensal microbiota is fundamental for the development of inflammatory pain.” Proceedings of the National Academy of Sciences 105.6 (2008): 2193-2197.
- Vuong, Helen E., and Elaine Y. Hsiao. “Emerging roles for the gut microbiome in autism spectrum disorder.” Biological psychiatry 81.5 (2017): 411-423.
- Carabotti, Marilia, et al. “The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems.” Annals of gastroenterology: quarterly publication of the Hellenic Society of Gastroenterology 28.2 (2015): 203.
- Sudo, Nobuyuki, et al. “Postnatal microbial colonization programs the hypothalamic–pituitary–adrenal system for stress response in mice.” The Journal of physiology 558.1 (2004): 263-275.
- Neufeld, K. M., et al. “Reduced anxiety‐like behavior and central neurochemical change in germ‐free mice.” Neurogastroenterology & Motility 23.3 (2011): 255-e119.
- Heijtz, Rochellys Diaz, et al. “Normal gut microbiota modulates brain development and behavior.” Proceedings of the National Academy of Sciences 108.7 (2011): 3047-3052.
- Desbonnet, Lieve, et al. “Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression.” Neuroscience 170.4 (2010): 1179-1188.
- Bravo, Javier A., et al. “Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve.” Proceedings of the National Academy of Sciences 108.38 (2011): 16050-16055.
- Clarke, Gerard, et al. “The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner.” Molecular psychiatry 18.6 (2013): 666.
- Savignac, H. M., et al. “B ifidobacteria exert strain‐specific effects on stress‐related behavior and physiology in BALB/c mice.” Neurogastroenterology & Motility 26.11 (2014): 1615-1627.
- De Palma, Giada, et al. “Microbiota and host determinants of behavioural phenotype in maternally separated mice.” Nature communications 6 (2015): 7735.
- Kelly, John R., et al. “Transferring the blues: depression-associated gut microbiota induces neurobehavioural changes in the rat.” Journal of psychiatric research 82 (2016): 109-118.
- Ait-Belgnaoui, Afifa, et al. “Lactobacillus farciminis treatment suppresses stress induced visceral hypersensitivity: a possible action through interaction with epithelial cell cytoskeleton contraction.” Gut 55.8 (2006): 1090-1094.
- Foster, Jane A., and Karen-Anne McVey Neufeld. “Gut–brain axis: how the microbiome influences anxiety and depression.” Trends in neurosciences 36.5 (2013): 305-312.
- Martin, Clair R., et al. “The brain-gut-microbiome axis.” Cellular and molecular gastroenterology and hepatology 6.2 (2018): 133-148.
- Magnusson, K. R., et al. “Relationships between diet-related changes in the gut microbiome and cognitive flexibility.” Neuroscience 300 (2015): 128-140.
- Choi, Jeong June, et al. “Exercise attenuates PCB-induced changes in the mouse gut microbiome.” Environmental health perspectives 121.6 (2013): 725-730.
- Friedenreich, Christine, et al. “Physical activity and risk of colon and rectal cancers: the European prospective investigation into cancer and nutrition.” Cancer Epidemiology and Prevention Biomarkers 15.12 (2006): 2398-2407.
- Lustyk, Kathleen MB, et al. “Does a physically active lifestyle improve symptoms in women with irritable bowel syndrome?.” Gastroenterology Nursing 24.3 (2001): 129-137.
- Allen, Jacob M., et al. “Voluntary and forced exercise differentially alters the gut microbiome in C57BL/6J mice.” Journal of applied physiology 118.8 (2015): 1059-1066.
- Cook, Marc D., et al. “Forced treadmill exercise training exacerbates inflammation and causes mortality while voluntary wheel training is protective in a mouse model of colitis.” Brain, behavior, and immunity 33 (2013): 46-56.
- Bravo, Javier A., et al. “Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve.” Proceedings of the National Academy of Sciences 108.38 (2011): 16050-16055.
- Arentsen, Tim, et al. “Host microbiota modulates development of social preference in mice.” Microbial ecology in health and disease 26.1 (2015): 29719.
- Dobson, Geoffrey P., et al. “Specific pathogen-free (SPF) animal status as a variable in biomedical research: Have we come full circle?.” EBioMedicine 41 (2019): 42.
- Bruce-Keller, Annadora J., et al. “Maternal obese-type gut microbiota differentially impact cognition, anxiety and compulsive behavior in male and female offspring in mice.” PLoS One 12.4 (2017): e0175577.
- Desbonnet, Lieve, et al. “Gut microbiota depletion from early adolescence in mice: implications for brain and behaviour.” Brain, behavior, and immunity 48 (2015): 165-173.
- Zheng, Peng, et al. “The gut microbiome from patients with schizophrenia modulates the glutamate-glutamine-GABA cycle and schizophrenia-relevant behaviors in mice.” Science advances 5.2 (2019): eaau8317.
- Coretti, Lorena, et al. “Sex-related alterations of gut microbiota composition in the BTBR mouse model of autism spectrum disorder.” Scientific reports 7 (2017): 45356.
- Williams, Brent L., et al. “Impaired carbohydrate digestion and transport and mucosal dysbiosis in the intestines of children with autism and gastrointestinal disturbances.” PloS one 6.9 (2011): e24585.