Need Anxiety & Depression devices for your tests?
Introduction to the Striatum
The striatum is a brain region within the basal ganglia comprised of three major components: the caudate, the putamen and the ventral striatum. While the caudate and putamen are located dorsal of the internal capsule, the ventral striatum rests on the ventral side of the internal capsule and itself contains the nucleus accumbens as well as the olfactory tubercle.[1]
Comprised primarily of GABAergic (and therefore inhibitory) medium spiny neurons (MSNs), the striatum receives the majority of its input from the neocortex in addition to components of the limbic system including the amygdala, hippocampus, and thalamus. As such, synaptic activity in the striatum modulates these inputs before sending the resultant signals to other brain regions. Aside from MSNs, which comprise ~95% of the neurons found in the striatum, this structure also contains cholinergic, fast-spiking interneurons which may play a significant role in the learning behaviors governed by the balance of reward and punishment.[2]
Signals leaving the striatum are divided into two categories: direct pathway and indirect pathway efferents. The direct pathway efferents are carried by MSNs which express D1-type dopamine receptors and primarily target the substantia nigra pars reticulata. These neurons carry the signals which evoke voluntary movements based on motivational information received from afferent inputs. Conversely, the indirect pathway efferents are carried by MSNs which express D2-type dopamine receptors and project to the globus pallidus where they function to inhibit involuntary movements and thereby permit the prioritization of actions to carry out motivated behaviors.
Function of the Striatum
From a large-scale perspective, it can be said that the striatum functions as a relay-station for motivation. The majority of inputs arriving at the striatum originate in the limbic system structures which are known to mediate reward signals, while the outputs from the striatum primarily target structures of the motor assembly, thereby sending signals to carry out or inhibit actions based on the level of reward or alternatively, the absence of expected reward. Indeed, evidence suggests that the interneurons found in the striatum encode signals that determine action responses to either rewarding or negative-affect experiences. Thus, the behaviors associated with the striatum are those governing the balance of actions that guide our relationship with reward and punishment.
Furthermore, the striatum plays a critical role in behavioral development during adolescence in the so-called ‘‘dual systems’’ model. Risk-taking behavior during adolescent development necessarily results in both positive and negative emotional outcomes. These outcomes are then encoded in striatal circuitry, thereby developing an affective map for guiding future behavioral decisions based on previous experiences.
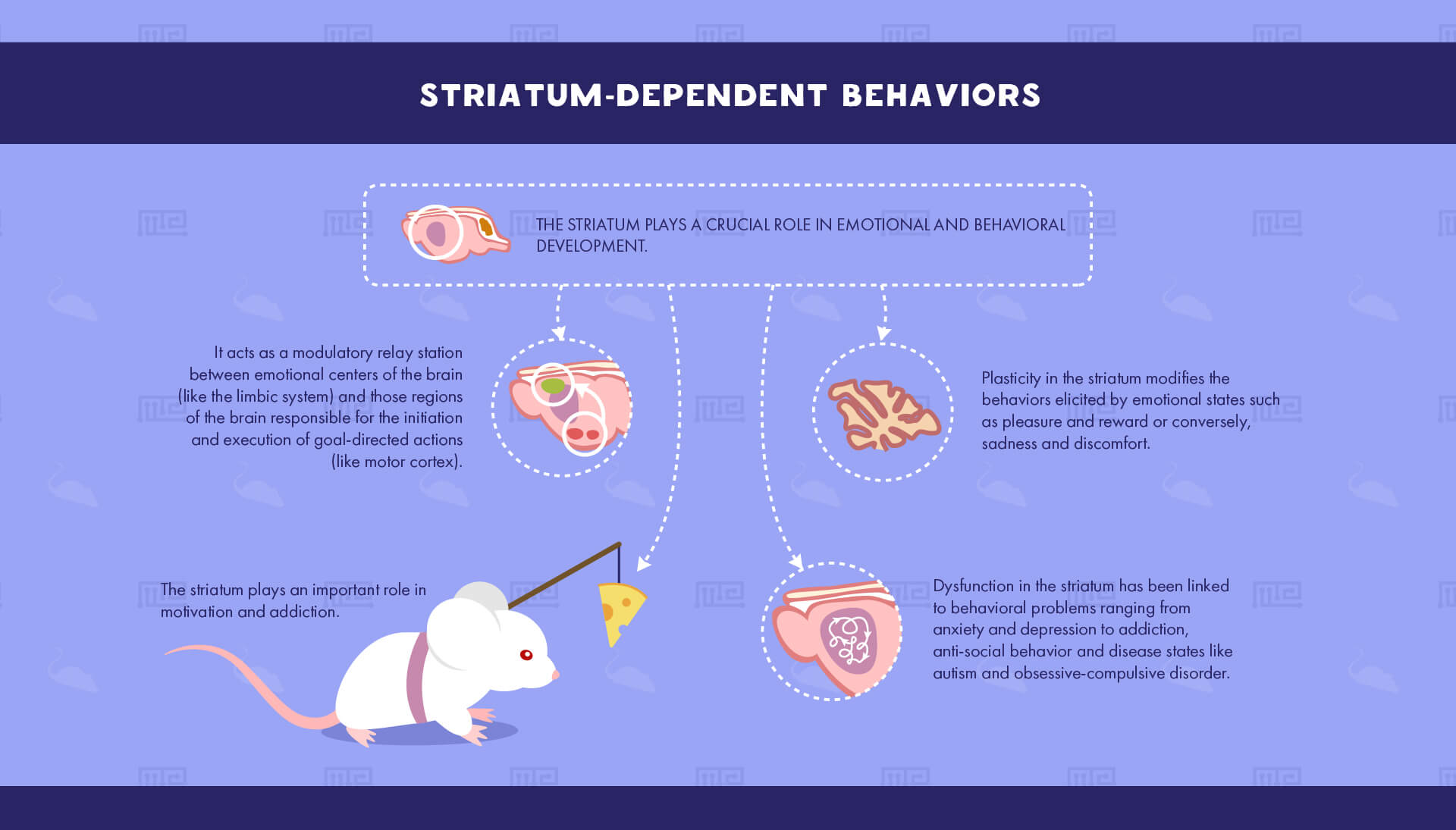
Striatum-Dependent Behaviors
The striatum plays a crucial role in emotional and behavioral development by acting as a modulatory relay station between emotional centers of the brain (such as the limbic system) and those regions of the brain responsible for the initiation and execution of goal-directed actions (such as the substantia nigra and motor cortex). Plasticity in the striatum thereby modifies the behaviors elicited by emotional states such as pleasure and reward or conversely, sadness and discomfort. Dysfunction in the striatum has thus been linked to behavioral problems ranging from anxiety and depression to addiction, anti-social behavior and such disease states as autism and obsessive-compulsive disorder.
Motivation and addiction
As a mediary between emotions and actions, the striatum plays an important role in behaviors associated with reward-learning such as appetitive behaviors (e.g. feeding and drug-taking) as well as responses to negative emotions (e.g. fear). One well-studied aspect of the striatum’s role in behavior is the study of drug addiction. The striatum, and in particular the nucleus accumbens, has been shown to mediate drug-seeking and drug-taking behavior in both humans and rodents for nearly all addictive drugs, from cocaine and heroin to nicotine and alcohol.[4] Experimenters commonly use the conditioned place-preference task, wherein animals exhibit a preference for one area of the maze in which they are exposed to rewarding drugs.[3] Alternatively, self-administration chambers are used to allow animals to take drugs at will. The behaviors of acquiring, maintaining, escalating and extinguishing self-administration of addictive drugs have been extensively shown to require activation of the ventral striatum.[5]
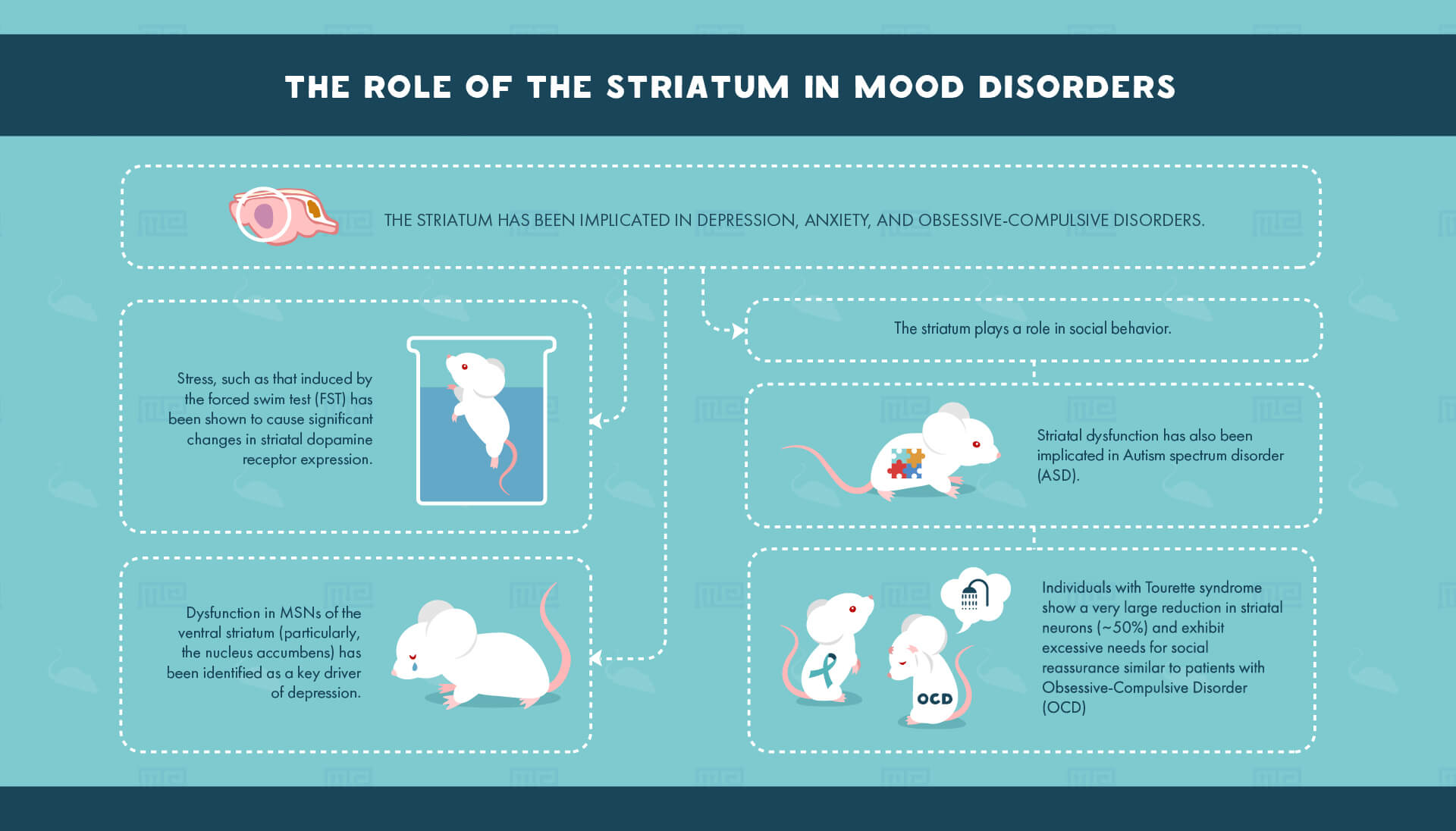
Mood disorders
Furthermore, the striatum has been implicated in depression, anxiety, and obsessive-compulsive disorders. Stress, such as that induced by the forced swim test (FST) has been shown to cause significant changes in striatal dopamine receptor expression.[6] After experiencing the stressful stimuli, animals may be again tested in the FST in order to observe coping strategies. In this case, the researchers found that blockade of a subtype of dopamine receptors in the dorsolateral striatum prevented the acquisition of coping mechanisms seen in normal mice. Thus, the forced swim test may be used both to induce stress as well as examine the behavioral consequences of previously induced stress.
In addition to the FST, the elevated plus maze (EPM) is frequently used to assess models of stress and anxiety. For example, following social defeat stress, in which a dominant animal is placed on one side of the resident–intruder maze in order to induce a stress response in an animal on the other side, the defeated animal will show changes in their behavior in the elevated plus maze.[7] In particular, mice were found to spend more time in the closed arms of the EPM, which they perceive as safe, and less time in the open arms in which they feel more vulnerable. Mice within this study also showed an increased preference for sucrose as well as a decreased social behavior in a sociability chamber.
Similarly to stress and anxiety, dysfunction in MSNs of the ventral striatum (in particular, the nucleus accumbens) has been identified as a key driver of depression.[8] Frequently, potential treatments for depression are often investigated using the tail suspension test. Animals exhibiting depressive symptoms show increased time spent immobile when suspended by their tail, as opposed to time spent struggling to escape. One such recent study investigating the potential for Δ,[3]2-Hydroxybakuchiol as a therapeutic treatment for depression found that mice treated with the drug exhibited reduced immobility time in the tail suspension test, as well as in the FST.[9]
Social behavior
Furthermore, certain aspects of social behavior require the involvement of the striatum. For instance, to better understand the role of the neuropeptide oxytocin in the striatum, researchers treated mice with varying concentrations of oxytocin before testing them for sociability.[10] They found that the oxytocin-treated mice spent more time interacting with novel social partners. These same mice additionally showed alterations in more basic, non-socially dependent memory functions, as exhibited by changes in their responses in the novel object recognition task.
Autism Spectrum Disorder
Finally, striatal dysfunction has been implicated in Autism spectrum disorder[11] (ASD). ASD is characterized by certain compulsive behaviors and given the role of the striatum in promoting emotionally driven actions it is of little surprise that both children and adults with ASD show altered function in the striatum.[12] One model for autism-like symptomology in mice is the Shank3 knockout model wherein mice are bred lacking the Shank3 protein which normally functions as an anchor tying receptors and ion channels to the post-synaptic density in addition to guiding synapse formation during development. Using a three-chamber sociability task, researchers have found that Shank3 knockout mice exhibit significantly reduced time spent interacting with social partners, in addition to a reduced discrimination between novel and previously known social partners.[13] These social deficits were directly linked to changes in the striatum, where Shank3 is primarily expressed, including reductions in synaptic density proteins and both morphological and electrophysiological abnormalities in striatal MSNs.
Tourette’s syndrome and Obsessive Compulsive Disorder
Interestingly, while very little attention has been paid to the role that the cholinergic neurons of the striatum play in social behavior, recent data show that specific loss of this small population of striatal neurons leads to social dysfunction.[14] Individuals with Tourette syndrome show a very large reduction in these neurons (~50%) and exhibit excessive needs for social reassurance similar to patients with Obsessive-Compulsive Disorder (OCD). This is proposed to be due to an inability for the basal ganglia to acknowledge the salience of initial social contact, which is normally sufficient and therefore allows an individual to focus on other salient subjects. Using a hole-board test, as well as a sociability chamber, researchers showed that mice with a loss of these cholinergic interneurons spent excessive time exploring already-explored objects and social partners, indicating an inability to task-switch after initial exploration (much like what is seen in patients with Tourette’s Syndrome or OCD).
Summary
Clearly, the role of the striatum as an intermediary between affective states and the execution of emotionally-driven actions implicates it in a wide variety of behaviors. As such, striatal dysfunction is implicated in many normal, as well as aberrant behaviors from social interactions and stress to disease states such as ASD and Tourette’s Syndrome which can be analyzed with a variety of tests.
Reference
- Fix, James D. (2008). “Basal Ganglia and the Striatal Motor System”. Neuroanatomy (Board Review Series) (4th ed.). Baltimore: Wulters Kluwer & Lippincott Wiliams & Wilkins. 274–281
- Lanciego, José L.; Luquin, Natasha; Obeso, José A. (22 January 2017). “Functional Neuroanatomy of the Basal Ganglia”. Cold Spring Harbor Perspectives in Medicine. 2 (12): a009621.
- Tzschentke, T. M. (2007), REVIEW ON CPP: Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addiction Biology, 12: 227-462.
- Scofield, M. D., Heinsbroek, J. A., Gipson, C. D., Kupchik, Y. M., Spencer, S., Smith, A. C., Roberts-Wolfe, D., … Kalivas, P. W. (2016). The Nucleus Accumbens: Mechanisms of Addiction across Drug Classes Reflect the Importance of Glutamate Homeostasis. Pharmacological reviews, 68(3), 816-71.
- McFarland, K., Kalivas, P. W. ‘(2001). ‘The Circuitry Mediating Cocaine-Induced Reinstatement of Drug-Seeking Behavior’’ Journal of Neuroscience, 21 (21) 8655-8663.
- Campus, P., Canterini, S., Orsini, C., Fiorenza, M. T., Puglisi-Allegra, S., & Cabib, S. (2017). Stress-Induced Reduction of Dorsal Striatal D2 Dopamine Receptors Prevents Retention of a Newly Acquired Adaptive Coping Strategy. Frontiers in pharmacology, 8, 621.
- Iñiguez, S. D., Riggs, L. M., Nieto, S. J., Dayrit, G., Zamora, N. N., Shawhan, K. L., Cruz, B., Warren, B. L. (2014). Social defeat stress induces a depression-like phenotype in adolescent male c57BL/6 mice. Stress (Amsterdam, Netherlands), 17(3), 247-55.
- Krishnan, V., & Nestler, E. J. (2008). The molecular neurobiology of depression. Nature, 455(7215), 894-902.
- Zhao, G., Guo, L. H., Huang, W., & Hu, J. L. (2018). Δ3,2-Hydroxybakuchiol Attenuates Depression in Multiple Rodent Models Possibly by Inhibition of Monoamine Transporters in Brain. Evidence-based complementary and alternative medicine : eCAM, 2018, 1325141.
- Zhang, X., Li, Q., Zhang, M., Lam, S., Sham, P. C., Bu, B., Chua, S. E., Wang, W., McAlonan, G. M. (2015). The Effect of Oxytocin on Social and Non-Social Behaviour and Striatal Protein Expression in C57BL/6N Mice. PloS one, 10(12), e0145638.
- Langen, M., Schnack, H. G., Nederveen, H., Bos, D., Lahuis, B. E., de Jonge, M. V., … Durston, S. (2009). Changes in the Developmental Trajectories of Striatum in Autism. Biological Psychiatry, 66(4), 327–333.
- Mundy, P. (2017). A review of joint attention and social-cognitive brain systems in typical development and autism spectrum disorder. European Journal of Neuroscience, 47(6), 497-514.
- Peça, J., Feliciano, C., Ting, J. T., Wang, W., Wells, M. F., Venkatraman, T. N., Lascola, C. D., Fu, Z., … Feng, G. (2011). Shank3 mutant mice display autistic-like behaviours and striatal dysfunction. Nature, 472(7344), 437-42.
- Martos, Y. V., Braz, B. Y., Beccaria, J. P., Murer, M. G., & Belforte, J. E. (2017). Compulsive Social Behavior Emerges after Selective Ablation of Striatal Cholinergic Interneurons. The Journal of Neuroscience, 37(11), 2849.