The brain and the body are intimately and ineluctably intertwined; our behaviors are not simply caused by our brain, but are a product of complex interactions between our brain and other organs. Most of us are familiar with how damage to the brain can affect behavior (e.g. a stroke causing a sudden change in personality), but many of us are not so familiar with how diseases of other organs can also have significant behavioral consequences.
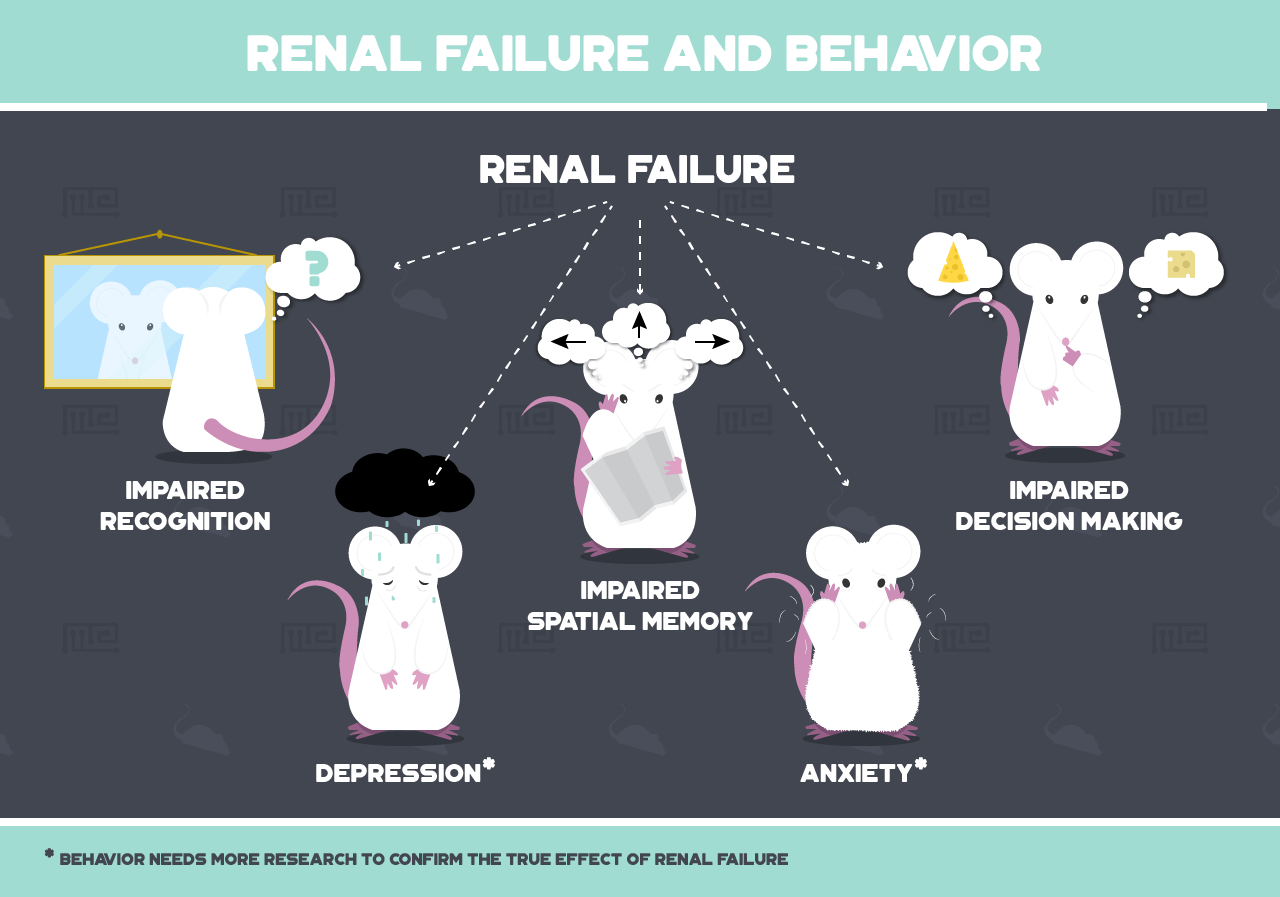
Renal failure, in which the kidneys cease to function, is one bodily disorder that can have such profound behavioral effects. The kidneys filter the blood, removing toxins and regulating levels of vital substances such as sodium and potassium. Mouse models with induced renal failure have been used to study the effects of this disorder on behavior.
Renal Failure and Depression
Studies of behavioral change in mice with induced renal failure have consistently associated depression, or at least symptoms interpreted as depression-like, with the animals’ disease.
A study conducted in 2014 in Oman[1] looked at mice with renal failure induced by a high dose of the platinum-based anti-cancer drug cisplatin. Behavioral changes observed in this study could not have been caused by direct effects of cisplatin on the brain, since platinum cannot pass through the protective layer of cells around the outside of the brain[2].
Depression-like behavior was discovered via a forced swimming test and a tail suspension test. In forced swimming, a mouse is dropped into a cylinder full of water and left to swim for several minutes; the mice with renal failure showed more immobility (sitting still in the water), which was interpreted as a sign of depression. In the tail suspension test, where each mouse is stuck to a board by its tail and struggles to get free, the mice with renal failure again exhibited significantly more immobility.
Similar findings emerged from a study conducted in India in 2016[3]. The mice used in this study had chronic kidney disease (CKD) (a precursor to renal failure) induced by a diet rich in adenine. These mice were also subjected to a forced swimming test, and showed a greater tendency to remain still in the water than control mice.
The mice in both studies also exhibited motor problems. In the 2014 study from Oman, mice with renal failure fell more quickly during a rotarod test and wire-gripping test, which the experimenters interpreted as a deficit in neuromuscular coordination. Also, the tendency of the mice with CKD in the Indian study to remain immobile in water for longer was interpreted by the experimenters as an abnormality in motor behavior.
The authors of both these studies interpreted the reduced propensity of their mouse models to move around as a sign of depression. However, since the mice in both studies also exhibited motor problems, it is possible that their increased immobility could be accounted for by motor issues and not by depression.
Renal Failure and Motor Activity/Coordination
The 2014 study carried out in Oman[1] revealed a variety of deficits in motor activity and neuromuscular coordination in the mice with induced renal failure. As mentioned above, more rapid falling of these mice during the Rotarod test was interpreted as their exhibiting deficiencies in neuromuscular coordination. These mice also performed worse in a simple grip-strength test requiring them to hang from a wire. These coordination abnormalities were associated with an overall reduction in general motor activity as measured by simple displacement of the mice over time in two dimensions.
However, the French study from the same year does not corroborate the Omanian findings on neuromuscular coordination, reporting no difference in time on the rotarod for mice with chronic renal failure compared to control mice[4]. Perhaps the use of cisplatin to induce renal failure in the Omanian study resulted in motor side effects not present in the French study (where renal failure was induced by ApoE knockout[4]).
A 2011 study[9] where renal failure in mice was induced by adenine showed decreased motility of the mice during a forced swimming test, as well as less time remaining on a rotarod compared to healthy controls, and less movement in general. Thus, the evidence on the effects of kidney failure on motor activity and neuromuscular coordination in mice is mixed, but leans more towards an associated deficit in both of these behaviors.
Renal Failure and Anxiety
Studies on mice with renal failure have shown conflicting results with regard to anxiety. In the study conducted in India in 2016[3], the researchers interpreted the mice as showing greater anxiety. They assessed anxiety using a light-dark box test, where animals choose whether to sit in a dark or light portion of a box. The mice with CKD spent significantly less time in the dark portion of the box, suggesting that they are more anxious.
However, a study carried out in 2014 in France[4] apparently showed the opposite result. The mice with induced renal failure in this study underwent the same kind of assessment in a light-dark box, and did not show a marked preference for the lighter side, suggesting that they were not more anxious. This contradicts evidence from humans that renal failure increases anxiety[5], and the researchers suggest that this may be due to mice not suffering from the same self-esteem issues as human patients.
Renal Failure and Recognition
The French study from 2014[4] also suggested that mice with renal failure have an impaired ability to recognize objects they have seen before. This recognition ability was assessed by a Y-maze test, in which mice with genetically induced renal failure faired noticeably worse.
Renal Failure and Spatial Learning
A paper published in 1999[6], looking at mice with acute partial renal failure induced by removing some of their nephrons (the cloud-shaped structures in the kidney that allow it to filter blood), reported that the mice showed problems with spatial learning in the Morris water maze. The Morris water maze is a simple task in which mice learn the position of a raised platform that allows them to escape from a pool of deep water, and so escape more quickly each time they enter the maze. A failure to improve escape times as quickly as control mice, as seen in this study, suggests issues with spatial learning.
A 2013 study carried out in Japan[7] corroborated these findings: mice with renal failure induced by surgical intervention showed significantly worse performance than control mice on the radial arm water maze test (a hybrid of the radial arm maze and Morris water maze tests) suggesting spatial memory deficiency. Worse performance than controls in the normal radial arm maze test (where mice have to remember the location of food) has been observed with CKD mice [8].
Mouse models of Alzheimer’s disease with comorbid CKD have been observed[9] to exhibit diminished performance in the Morris water maze. In this case, the experimenters interpreted the reduction in spatial memory capacity as an acceleration of the typical cognitive decline associated with Alzheimer’s. It is worth bearing in mind that wild-type CKD mice in this study (without the mutation conferring Alzheimer’s like pathology) do not show worse performance in the Morris water maze, suggesting the cognitive decline results from the combined effect of AD and CKD rather than CKD alone.
Renal Failure and Decision Making
The study carried out in France in 2014[4] also showed that mice with renal failure may have altered or impaired decision making. The researchers studied decision making with an elevated maze where the mouse is placed in the center and has to decide which way to move. The mice with renal failure sat in the center for longer, suggesting they may take longer to make decisions.
Conclusion
A number of studies using mouse models of renal failure have shown that this disease may have highly significant effects on behavior, both in model organisms and in humans. Renal failure (and its precursor CKD) in mice has been associated with depression, anxiety, deficits in spatial learning, recognition and motor functions, and delayed decision making. This mirrors some of the effects seen in human patients[6][11]. It adds to the growing body of knowledge obtained from mouse models about how bodily disorders can affect behavior and the brain, and will help us to further understand how such disorders can affect human psychology.
References
- B. H. Ali, A. Ramkumar, T. T. Madanagopal, M. I. Waly, M. Tageldin, S. Al-abri, M. Fahim, J. Yasin, A. Nemmar. Motor and Behavioral Changes in Mice With Cisplatin-Induced Acute Renal Failure. Physiol. Res. 63: 35-45, 2014.
- Bernocchi G, Bottone Mg, Piccolini Vm, Dal Bo V, Santin G, De Pascali Sa, Migoni D, Fanizzi Fp. Developing central nervous system and vulnerability to platinum compounds. Chemother Res Pract 2011: 315418, 2011.
- Mazumder MK, Giri A, Kumar S, Borah A. A highly reproducible mice model of chronic kidney disease: Evidences of behavioural abnormalities and blood-brain barrier disruption. Life Sci. 2016. Sep 15; 161:27-36.
- Jean-Marc Chillon, François Brazier, Philippe Bouquer and Ziad A. Massy. Neurological Disorders in a Murine Model of Chronic Renal Failure. Toxins 2014, 6, 180-193.
- A. De Sousa. Psychiatric issues in renal failure and dialysis. Indian J Nephrol. 2008 Apr; 18(2): 47–50.
- M. Al Banchaabouch, R. D’Hooge, B. Marescau, P. P. De Deyn. Behavioural Deficits During the Acute Phase of Mild Renal Failure in Mice. Metabolic Brain Disease. September 1999, Volume 14, Issue 3, pp 173–187.
- Kiichiro Fujisaki Kazuhiko Tsuruya Mayumi Yamato Jiro Toyonaga Hideko Noguchi Toshiaki Nakano Masatomo Taniguchi Masanori Tokumoto Hideki Hirakata Takanari KitaCerebral oxidative stress induces spatial working memory dysfunction in uremic mice: neuroprotective effect of tempol. Nephrology Dialysis Transplantation, Volume 29, Issue 3, 1 March 2014, Pages 529–538.
- Haruyama N, Fujisaki K, Yamato M, Eriguchi M, Noguchi H, Torisu K, Tsuruya K, Kitazono. Improvement in spatial memory dysfunction by telmisartan through reduction of brain angiotensin II and oxidative stress in experimental uremic mice. Life Sci. 2014 Sep 15;113(1-2):55-9.
- Takashi Nakagawa, Yu Hasegawa, Ken Uekawa, Shokei Kim-Mitsuyama. Chronic kidney disease accelerates cognitive impairment in a mouse model of Alzheimer’s disease, through angiotensin II. Experimental Gerontology. 87 (2017) 108–112.
- Badreldin H Ali, Amal Ziada*, Isehaq Al Husseni, Sumaya Beegam, Abderrahim Nemmar. Motor and behavioral changes in rats with adenine-induced chronic renal failure: influence of acacia gum treatment. 2011. Experimental Biology & Medicine. Volume: 236 issue: 1, page(s): 107-112.
- Oyekçin DG1, Gülpek D, Sahin EM, Mete L. Depression, anxiety, body image, sexual functioning, and dyadic adjustment associated with dialysis type in chronic renal failure. Int J Psychiatry Med. 2012;43(3):227-41.