Depression and altered behavior often result from stressful situations. The brain interprets these situations and decides if they are life-threatening, then reacts by supplying extra energy and boosting the levels of brain chemicals[1]. Sometimes, however, these chemicals can remain elevated for longer periods of time, which ultimately leads to depression. Interestingly, there is increasing evidence that the resulting depression may involve numerous changes in the structures of the brain[2]. Specifically, brain imaging studies on animals have shown that certain areas of the brain may change in size and function in response to depression. Recent researches reveal that creatine, a substance found naturally in muscle cells, may alleviate depression and improve mice behavior.
What is Creatine?
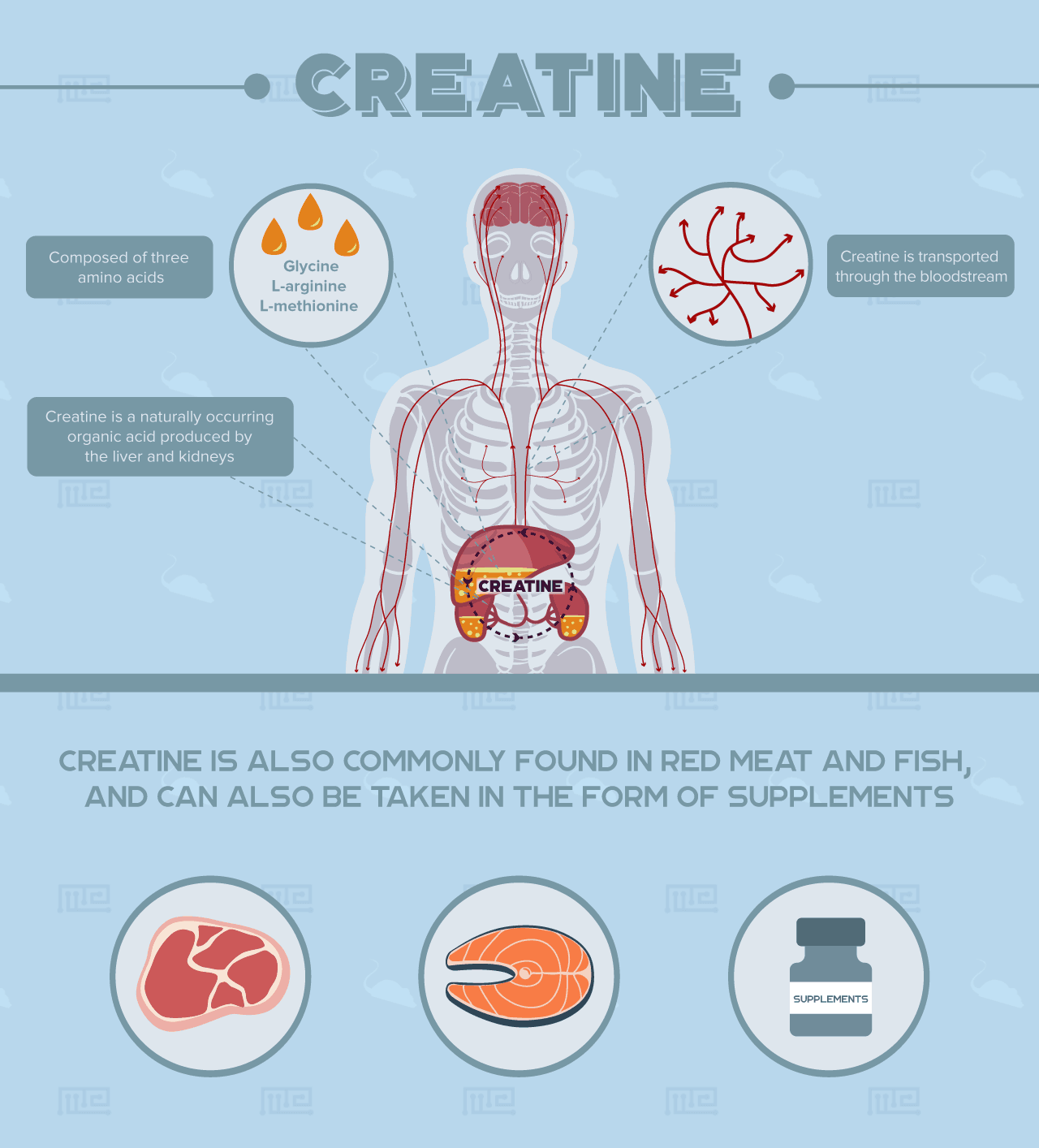
Creatine is a naturally occurring organic acid produced by the liver and kidneys. It is composed of three amino acids including glycine, L-arginine, and L-methionine. It is transported through the bloodstream to provide a readily available source of energy in body parts that demand high amount of energy such as the brain and skeletal muscles. Creatine is commonly found in red meat and fish, and it can also be taken in the form of supplements.
Can Creatine Alter Mice Behavior?
Changes in brain energy metabolism and damage to neurons (nerve cells) are associated with the development of depressive disorders[3]. The role of creatine in mice behavior is well established. Creatine may modify depressive behavior in mice because of its crucial role in
preventing energy exhaustion and death of neurons. In a study by Dedeoglu et al., researchers found that creatine administration in mice model of Huntington’s disease (an inherited condition that causes progressive breakdown of neurons in the brain) significantly extended survival and improved motor performance in both the 6- and 8-week treatment[4]. The researchers used the rotarod performance test in order to assess motor performance of mice. On the other hand, the researchers used criteria for euthanization (mice were euthanized if they were unable to right themselves after being placed on their back and initiate movement after being gently prodded for 20 seconds) to assess survival. Aside from extending survival and improving motor performance, oral creatine supplementation also significantly increased the concentrations of creatine and adenosine triphosphate (an energy-carrying molecule) of mice, which is suggestive of improved brain energy metabolism.
Similarly, a study by Brustovetsky et al. found that creatine exerts neuroprotective effects[5]. The researchers evaluated the ability of creatine to prevent death of cultured mice neurons exposed to either glutamate or 3-nitropropionic acid (3NP). The results of the study showed that pre-treatment of creatine for 2-5 days protected mice neurons against glutamate and 3NP toxicity.
Because creatine supplementation in mice is known to improve brain energy metabolism and protect neurons against damage or cellular injury, studies have been conducted to further assess the effects of creatine on mice behavior. For instance, Allen et al. examined the effects of long-term effect of creatine supplementation on Sprague-Dawley rats using the forced swim test (FST)[6]. Also known as the behavioral despair test, FST involves placing a rat or mouse inside a cylinder filled with water in order to assess the mobility and depression-like behavior of the animal. Thirty male and thirty-six female Sprague-Dawley rats were fed with a creatine diet for 5 weeks prior to the test. The results of the study revealed that creatine supplementation altered depression-like behavior during FST in female rats only. This suggests that creatine has a beneficial effect on depression-like behavior in rats in a sex-dependent manner.
In another study, Cunha et al. investigated the effect of oral creatine on male and female mice behavior[7]. The researchers used the tail suspension test in order to determine antidepressant activity. This test is based on the observation that if a mouse is placed under inescapable stress, then it will become immobile. Interestingly, researchers observed that creatine reduced the immobility time in the tail suspension test without affecting the locomotor activity of the mice, indicating that creatine does have an antidepressant-like effect.
How Creatine Exerts its Antidepressant Effect?
Creatine has been proposed to exert therapeutic effects in the management of depression-like behavior in mice, but the cell signaling pathways involved in its antidepressant effects are not well-known. In order to determine the specific mechanisms involved in the antidepressant effects of creatine, Cunha et al. investigated the involvement of PI3K/Akt pathway, an intracellular signaling pathway directly related to cell cycle[8]. The researchers treated mice with creatine at a dose of 1 mg/kg orally and they were subjected to undergo a tail suspension test to determine antidepressant activity. They observed that the acute treatment of mice with creatine was able to increase the activities of the PI3K/Akt pathway. The researchers therefore concluded that the antidepressant-like effect of creatine in the tail suspension test depends on the activation of the PI3K/Akt pathway.
Interestingly, evidence suggests that brain intracellular signaling systems including the AKT/GSK3 pathway have been found to be altered in patients with major depression and psychiatric illnesses[9]. Because creatine supplementation has been found to improve the activities of the PI3K/Akt pathway in mouse models of depression, there’s a strong possibility that creatine supplementation may also exert therapeutic benefits in people suffering from depressive symptoms.
Conclusion
Vital for optimum brain structure and performance, creatine is critical for a healthy mood and reducing mental distress in mice. Because of its positive effects on certain brain signaling pathways in mouse models, oral creatine supplementation may also help improve depressive symptoms in humans.
References
- Hitze B, Hubold C, van Dyken R. How the selfish brain organizes its supply and demand. Frontiers in neuroenergetics. 2010; 2:7.
- Gabbay V, Hess DA, Liu S, Babb JS, Klein RG, Gonen O (2007). Lateralized caudate metabolic abnormalities in adolescent major depressive disorder: a proton MR spectroscopy study. Am J Psychiatry 164: 1881–1889.
- Fuchs E, Cze´h B, Kole M, Michaelis T, Lucassen P (2004). Alterations of neuroplasticity in depression: the hippocampus and beyond. Eur Neuropsychopharmacol 14: S481–S490.
- Dedeoglu A, Kubilus JK, Yang L, et al. Creatine therapy provides neuroprotection after onset of clinical symptoms in Huntington’s disease transgenic mice. Journal of neurochemistry. 2003;85(6):1359-1367.
- Brustovetsky N, Brustovetsky T, Dubinsky J (2001). On the mechanisms of neuroprotection by creatine and phosphocreatine. J Neurochem 76: 425–434.
- Allen PJ, D’Anci KE, Kanarek RB, Renshaw PF. Chronic creatine supplementation alters depression-like behavior in rodents in a sex-dependent manner. Neuropsychopharmacology: official publication of the American College of Neuropsychopharmacology. 2010; 35(2):534-46.
- Cunha MP, Machado DG, Capra JC, Jacinto J, Bettio LE, Rodrigues AL. Antidepressant-like effect of creatine in mice involves dopaminergic activation. Journal of psychopharmacology (Oxford, England). 2012; 26(11):1489-501.
- Cunha MP, Budni J, Ludka FK. Involvement of PI3K/Akt Signaling Pathway and Its Downstream Intracellular Targets in the Antidepressant-Like Effect of Creatine. Molecular neurobiology. 2016; 53(5):2954-2968.
- Kim JY, Duan X, Liu CY, et al. DISC1 regulates new neuron development in the adult brain via modulation of AKT-mTOR signaling through KIAA1212. Neuron. 2009;63(6):761–773.