Introduction
Stress and anxiety are unfortunate but wholly familiar aspects of every human life. It is likely that these negative emotional experiences were selected for in evolution, on account of how they goad us to perform important actions and solve urgent problems in a timely manner.
While serving this useful function, anxiety and stress often prove pathological: anxiety disorders such as obsessive-compulsive disorder and generalized anxiety disorder are the most common category of mental illness, with over 270 million cases of such disorders reported worldwide in 2010, affecting more than 1 in every 15 people in some countries.[1]
Since these pathologies have such a high prevalence and lead to marked reductions in productivity and quality of life for their sufferers, it is imperative to investigate their causes and manifestations, as well as develop effective treatments for them. Mouse models, through a number of experimental paradigms, have featured centrally in this endeavor.
In this article, we will first give a brief overview of the meaning of stress and anxiety, and the neurobiological basis of these phenomena, before exploring the most common tests used to assess stress and anxiety in mouse models.
What are Stress & Anxiety?
Stress is an imprecisely defined phenomenon that overlaps to a certain extent with several other related behavioral phenomena, including anxiety and fear. Generally, stress is seen as a pathological or borderline-pathological feeling where the subject of the stress feels as if they are in a sort of “limbo” state in between normal functioning and some kind of breaking point.
An analogy can be drawn with materials: when we say a certain material is “stressed”, we mean that there is a force being applied to it that risks breaking it if the material is unable to resist or recover. Similarly, a subject undergoing physical or emotional stress feels a need to exert resistance, in order to prevent a catastrophic breakdown of vital biological functions, in the face of a hindrance exerted by some stressor (e.g. lack of resources, bad weather, an injury etc).
Although “stress” and “anxiety” are sometimes used interchangeably, and the physical signs of the two phenomena are quite similar, there are key differences: while stress relates to feelings of physical pressure and tension in response to a particular stressor, anxiety describes a more generalized feeling that some unwanted event (whose identity may be completely unknown) might occur in the near future. In practice, it is hard to tease these two apart in experimental paradigms,
While stress generally does not involve fear (although constant fear can induce a feeling of stress), anxiety can be thought of as a kind of fear i.e. the fear that something bad will happen. Not all fear is anxiety; for example, a prey animal may experience a kind of fear (i.e. terror) when being chased by a predator, which is completely distinct from anxiety.
Stress & the Brain
Stress has a central neuroendocrinological component, where the brain interacts with other glands in the body to produce a hormonal response to a stressor. The pituitary gland, a part of the brain underneath the hypothalamus, releases adrenocorticotropic hormone (ACTH) which travels to the adrenal gland in the abdomen and causes the release of steroids from the adrenal cortex.
The release of ACTH from the pituitary gland is regulated by several other parts of the brain. The hypothalamus can stimulate ACTH release via corticotropin-releasing factor (CRF), and the amygdala and hippocampus can release noradrenaline. The adreno-pituitary action causes glucocorticoid levels in the bloodstream to rise, which elicits a number of cognitive and behavioral symptoms.
There are further signaling effects: levels of the inhibitory neurotransmitter GABA fall in the hippocampus, while levels of dopamine in the striatum rise and noradrenaline dampens the desire to eat and sleep. The release of glutamate in the hypothalamus raises heart rate and blood pressure. It becomes difficult to perform tasks requiring good focus and working memory. Anxiety sets in, leading to agitated and uncoordinated movement, and immune function decreases, sometimes to dangerous levels.
Due to an increase in levels of free radicals (especially the signaling molecule NO) and subsequent oxidative stress, as well as high blood pressure, chronic stress response can cause permanent damage to the brain, including synapse loss, neuronal loss, and stroke. Moreover, the diminished immune response during stress raises the sufferer’s risk of contracting bodily illnesses, which can also negatively impact cognitive function.[2]
Stress Testing in Mice
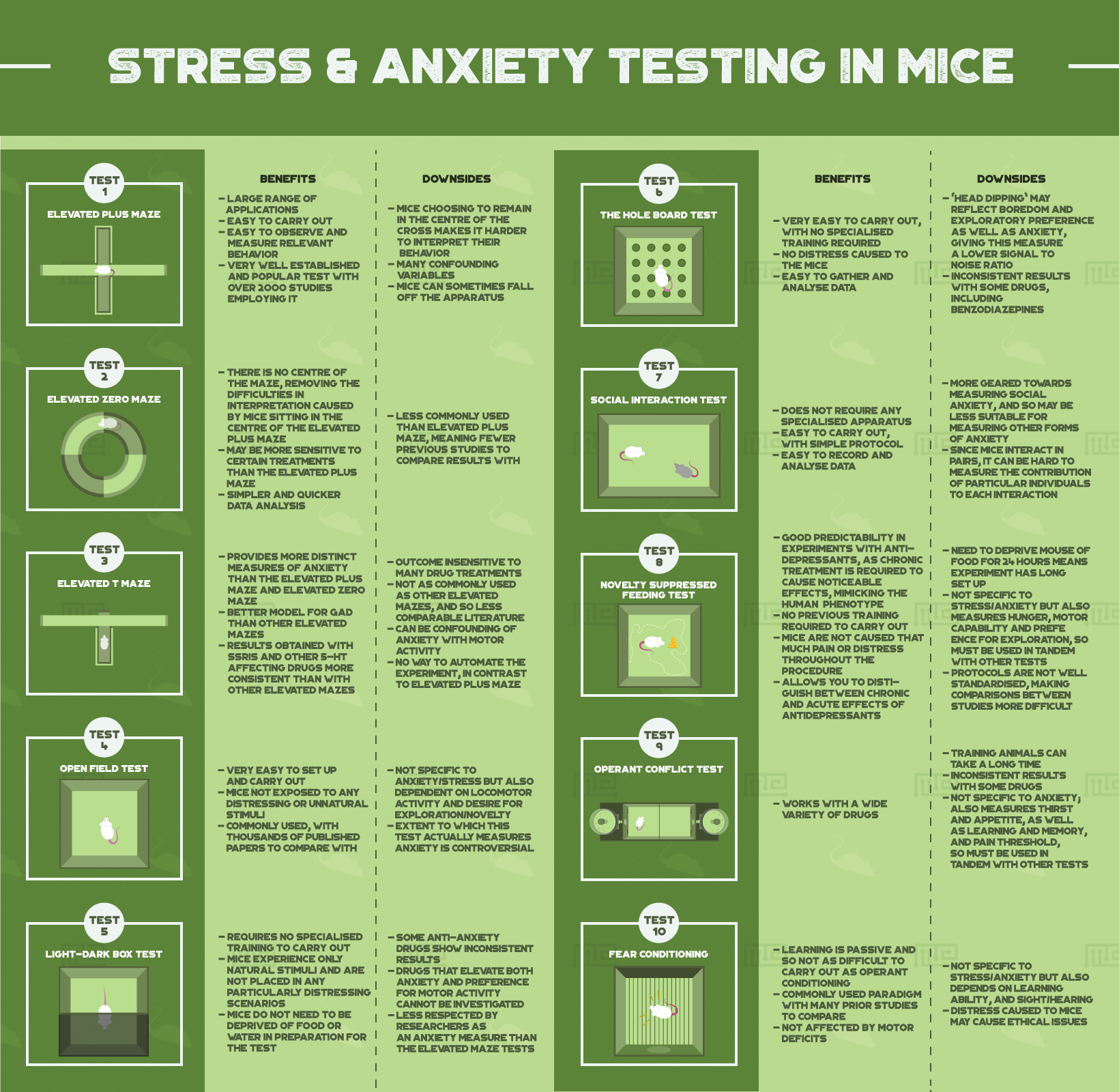
We will now examine the most common experimental paradigms used to measure stress and anxiety levels in mice. We begin by going through the design and experimental process of each test, before considering their benefits and downsides, and important things to consider when administering them.[3]
Elevated Plus Maze
The elevated plus maze is likely the most popular test of anxious behavior used with mouse models. The apparatus consists of a raised maze with four arms in a cross shape; two of the arms are exposed to the open air, and the other two arms are enclosed. Mice naturally prefer to avoid elevated areas for fear of falling, but also like to explore previously unvisited areas; as a result, the mice will spend an anxiety-limited time exploring the open arms (the higher their anxiety and stress, the less time they will spend in the open arms).
Benefits & Downsides
Benefits
- Large range of applications
- Easy to carry out
- Easy to observe and measure relevant behavior
- Very well established and popular test with over 2000 studies employing it
Downsides
- Mice choosing to remain in the centre of the cross makes it harder to interpret their behavior
- Many confounding variables (see below)
- Mice can sometimes fall off the apparatus[4]
Things to Consider
- If the experimenter is visible to the mice, this can affect their anxiety level, and hence their performance in the maze. It is advisable to record the experiment on video rather than watching personally.
- Other important possible confounding variables include quality of handling, housing conditions, light levels, and prior exposure to stressful stimuli.
- Mice can become acclimated to the maze following repeated experiments, and acclimatisation rates may differ between individuals.
Elevated Zero Maze
The elevated zero maze is very similar in both form and function to the elevated plus maze. Instead of being cross-shaped, this maze is circular with dark enclosed sections and lighter open sections alternating around the circumference. Assessment of anxiety level is made by the same criteria as with the elevated plus maze.[5]
Benefits & Downsides
Benefits
- There is no centre of the maze, removing the difficulties in interpretation caused by mice sitting in the centre of the elevated plus maze
- May be more sensitive to certain treatments (such as benzodiazepines) than the elevated plus maze
- Simpler and quicker data analysis
Downsides
- Less commonly used than elevated plus maze, meaning fewer previous studies to compare results with
Things to Consider
- Confounding variables for the elevated zero maze are the same as for the elevated plus maze.
Elevated T Maze
The elevated T maze test is also similar to the elevated plus maze and elevated zero maze, but is designed to test a slightly broader range of anxious behavioral phenotypes. The T shaped maze has two open arms exposed to the air and one enclosed arm.
The mouse is placed at the extreme end of one of the open arms, and then the time it takes to move away from that arm is measured. The experiment is then repeated, and the change in latency for the mouse to leave the arm is calculated. While the ratio of time the mouse spends in the open vs closed arms provides the same information as with the previous two mazes, the time taken to leave the open arm at the beginning provides an additional measure of “panic level”.[6][7]
Benefits and Downsides
Benefits
- Provides more distinct measures of anxiety than the elevated plus maze and elevated zero maze
- Better model for GAD than other elevated mazes
- Results obtained with SSRIs and other 5-HT affecting drugs more consistent than with other elevated mazes
Downsides
- Outcome insensitive to many drug treatments
- Not as commonly used as other elevated mazes, and so less comparable literature
- Can be confounding of anxiety with motor activity
- No way to automate the experiment, in contrast to elevated plus maze
Things to Consider
- As with the elevated plus maze, experimenters should remain hidden from the mice, to avoid raising their stress levels.
Open Field Test
The open field test is one of the most popular paradigms used with mouse models. The mouse is placed in a rectangular enclosure with high walls and a flat base and then left to roam freely for a certain amount of time. The distance travelled by the mouse, as well as which parts of the enclosure it spends time in, and how long it remains immobile, are measured. Greater movement and more movement away from the centre are interpreted as signs of lower anxiety/stress.[8][9]
Benefits and Downsides
Benefits
- Very easy to set up and carry out
- Mice not exposed to any distressing or unnatural stimuli
- Commonly used, with thousands of published papers to compare with
Downsides
- Not specific to anxiety/stress but also dependent on locomotor activity and desire for exploration/novelty
- Extent to which this test actually measures anxiety is controversial
Things to Consider
- This test is best used in tandem with other tests such as the elevated plus maze, to allow for the effects of anxiety to be teased apart from other variables.
- Boxes with grid lines make the measurement of location easier.
- The apparatus should be lit with a dim light, so that mice can see to navigate, but are also not made uncomfortable by bright lighting.
Light-Dark Box Test
The light-dark box test exploits the natural aversion that mice have to well-lit areas, on account of the risk of predation. Mice are placed in the enclosure, which consists of one lit compartment and one dark enclosed compartment, and left to roam around freely. The amount of time the mice spent in each compartment is then measured and compared, with increased time spent in the dark compartment interpreted as an index of elevated anxiety.
Benefits and Downsides
Benefits
- Requires no specialised training to carry out
- Mice experience only natural stimuli and are not placed in any particularly distressing scenarios
- Mice do not need to be deprived of food or water in preparation for the test
Downsides
- Some anti-anxiety drugs show inconsistent results
- Drugs that elevate both anxiety and preference for motor activity cannot be investigated
- Less respected by researchers as an anxiety measure than the elevated maze tests[8]
Things to Consider
- Mice that have increased novelty-seeking behavior may explore the light compartment more, leading to a false-positive measurement for anxiety.
- A false positive measure can also be caused by a preference in some mice for increased locomotor activity.
- Controls should be chosen carefully, as a basal preference for exploration varies between mouse strains.
The Hole Board Test
In the hole board test, mice are placed into a square enclosure with a flat floor that has a number of holes. Mice engage in a “head dipping” exploratory behavior that will lead them to lower their head into the holes and investigate them. Mice who explore the holes less are interpreted as having elevated anxiety levels. There is also a modified hole board test which combines the normal hole board with the open field test.[10][11]
Benefits and Downsides
Benefits
- Very easy to carry out, with no specialised training required
- No distress caused to the mice
- Easy to gather and analyse data
Downsides
- “Head dipping” may reflect boredom and exploratory preference as well as anxiety, giving this measure a lower signal to noise ratio
- Inconsistent results with some drugs, including benzodiazepines
Things to Consider
- Mice repeatedly exposed to the enclosure will become acclimated to it, and will no longer alter their behavior in response to benzodiazepine administration.
Social Interaction Test
The social interaction test focuses specifically on social anxiety i.e. anxiety felt towards interaction with other mice. Mice are placed in an enclosure with each-other and left to roam freely. The amount of time each mouse spends engaging in social interaction is then measured. Less time spent engaging in this behavior is seen as a sign of higher anxiety levels.
Benefits and Downsides
Benefits
- Does not require any specialised apparatus
- Easy to carry out, with simple protocol
- Easy to record and analyse data
Downsides
- More geared towards measuring social anxiety, and so may be less suitable for measuring other forms of anxiety
- Since mice interact in pairs, it can be hard to measure the contribution of particular individuals to each interaction
Things to Consider
- It would be best to use a more docile strain in this test, to avoid the risk of mice attacking and injuring one another.
Novelty Suppressed Feeding Test
Mice show a lower preference for food consumption when stressed. In the novelty suppressed feeding test, animals are not given food for 24 hours to induce hunger and then placed in a brightly lit box with a piece of food on a platform in the middle. The latency of the mouse to reach the platform is measured, with a longer latency interpreted as a sign of elevated stress levels.[12]
Benefits and Downsides
Benefits
- Good predictability in experiments with antidepressants, as chronic treatment is required to cause noticeable effects, mimicking the human phenotype
- No previous training required to carry out
- Mice are not caused that much pain or distress throughout the procedure
- Allows you to distinguish between chronic and acute effects of antidepressants
Downsides
- Need to deprive mouse of food for 24 hours means experiment has long set up
- Not specific to stress/anxiety but also measures hunger, motor capability and preference for exploration, so must be used in tandem with other tests
- Protocols are not well standardised, making comparisons between studies more difficult
Things to Consider
- It is good to set a standard maximum time limit on the latency measurement for each mouse before beginning the experiment, in case some mice simply refuse to eat indefinitely.
Operant Conflict Test
The operant conflict test involves operant conditioning, where an animal is trained to complete a task in order to gain a reward. In this case, the mouse is trained to push a lever and receive a sweet drink. The lever push is accompanied by a light or tone, and then in later iterations by an electric shock.[13][14]
The mouse is faced by a conflict between obtaining the reward and getting shocked, and how often it opts to endure the shock indicates its anxiety level, with fewer lever presses interpreted as showing higher anxiety. A variant of this test (the Vogel conflict test) removes the lever and simply requires the mouse to find a water bottle in a metal enclosure.
Benefits and Downsides
Benefits
- Works with a wide variety of drugs
Downsides
- Training animals can take a long time
- Inconsistent results with some drugs
- Not specific to anxiety; also measures thirst and appetite, as well as learning and memory, and pain threshold, so must be used in tandem with other tests
Things to Consider
- If the electric shock delivered is not strong enough, the animals may be able to ignore it, and so it will not serve as a suitable aversive stimulus.
- Type of housing (group housing or individual housing) can be a confounding variable.
Fear Conditioning
In contrast to the operant conflict test, standard fear conditioning is a form of Pavlovian or classical conditioning where the mouse is trained to associate a neutral stimulus, such as a light or a noise, with an aversive stimulus such as an electric shock. Receiving the neutral stimulus on its own will then cause the mouse to display startle and freeze behaviors, which will be more pronounced in mice with elevated anxiety and stress.[15]
Benefits and Downsides
Benefits
- Learning is passive and so not as difficult to carry out as operant conditioning
- Commonly used paradigm with many prior studies to compare
- Not affected by motor deficits
Downsides
- Not specific to stress/anxiety but also depends on learning ability, and sight/hearing
- Distress caused to mice may cause ethical issues
Things to Consider
- All mice must be healthy and calm before the experiment is started.
- Housing of the mice and performance of all experiments must take place in a quiet room (ideally soundproofed).
- Male mice may fight when housed, especially if a less docile strain is used, and so many researchers prefer to test female mice only.
Conclusion
Anxiety disorders are serious conditions that affect millions of people worldwide. Stress testing in mice has proven key to the development of effective treatments for these disorders. Taking note of the information provided here should allow researchers to choose the right paradigms for their mouse anxiety and stress experiments.
References
- J. Baxter, T. Vos, K. M. Scott, A. J. Ferrari and H. A. Whiteford. The global burden of anxiety disorders in 2010. Psychological Medicine. January 2014, pp 1 – 12.
- Kumar A, Rinwa P, Kaur G, Machawal L. Stress: Neurobiology, consequences and management. J Pharm Bioall Sci. 2013;5:91‐
- Alline C. Campos, Manoela V. Fogac ̧ Daniele C. Aguiar, Francisco S. Guimarã̃es. Animal models of anxiety disorders and stress. Revista Brasileira de Psiquiatria. 2013;35:S101–S111.
- Walf, A. A., & Frye, C. A. 2007. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nature Protocols, 2(2), 322–328.
- Braun, A. A., Skelton, M. R., Vorhees, C. V., & Williams, M. T. 2011. Comparison of the elevated plus and elevated zero mazes in treated and untreated male Sprague-Dawley rats: Effects of anxiolytic and anxiogenic agents. Pharmacology, Biochemistry, and Behavior, 97(3), 406–415.
- Frederico G. Graeff, Cristina Ferreira Netto, He ́lio Zangrossi Jr. The elevated T-maze as an experimental model of anxiety. Neuroscience and Biobehavioral Reviews. 23. 1998. 237–246,
- Pinheiro, Simone H., Zangrossi-Jr., Hélio, Del-Ben, Cristina M., & Graeff, Frederico G. 2007. Elevated mazes as animal models of anxiety: effects of serotonergic agents. Anais da Academia Brasileira de Ciências, 79(1), 71-85.
- Ennaceur, A. 2013. Tests of unconditioned anxiety — Pitfalls and disappointments. Physiology & Behavior. 135: 55–71.
- Sturman, O; Germain, P-L; Bohacek, J. Exploratory rearing: a context- and stress-sensitive behavior recorded in the open-field test. 2018. Stress. 1–10.
- Takeda, H.; Tsuji, M.; Matsumiya, T. 1998. Changes in head-dipping behavior in the hole-board test reflect the anxiogenic and/or anxiolytic state in mice. European Journal of Pharmacology. 350 (1): 21–29.
- Labots, M., Van Lith, H. A., Ohl, F., & Arndt, S. S. 2015. The Modified Hole Board – Measuring Behavior, Cognition and Social Interaction in Mice and Rats. Journal of Visualized Experiments : JoVE, (98), 52529.
- Blasco-Serra, González-Soler EM, Cervera-Ferri, Teruel-Martí, Valverde-Navarro. A standardization of the Novelty-Suppressed Feeding Test protocol in rats. Neurosci Lett. 2017 Sep 29;658:73-78.
- Millan, Mark J. 2003. The neurobiology and control of anxious states. Progress in Neurobiology. 70: 83–244.
- Millan, Mark; Brocco, Mauricette. 2003. The Vogel conflict test: procedural aspects, g-aminobutyric acid, glutamate and monoamines. European Journal of Pharmacology. 463: 67–96.
- Curzon P, Rustay NR, Browman KE. Cued and Contextual Fear Conditioning for Rodents. In: Buccafusco JJ, editor. Methods of Behavior Analysis in Neuroscience. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2009. Chapter 2.