Dominance hierarchies are a common feature of animal societies. Some individuals will behave more dominantly toward others, and these interactions result in a rank or status order. In some species, social ranks do not change (e.g. in insects such as bees), but most social mammals like humans and other primates have adaptable roles, where one individual can move through many social ranks over a lifetime.
The study of animal sociality is called sociobiology, which E. O. Wilson (1975) defines as “the extension of population biology and evolutionary theory to social organization”. Under this framework, social behavior is understood to ultimately arise through natural selection. Dominance hierarchies impact the survival and reproduction of individuals by regulating access to food, mates, and shelter. As such, the stability of a dominance hierarchy can affect whether a population prospers or collapses.
Why are dominance hierarchies important?
The Nobel laureate, Nikolaas Tinbergen (1963), established that social behaviors arise through a combination of genetic, physiological, and environmental factors. Today, we understand that regions of the brain responsible for recognizing body language and storing social memory control dominance behavior. Indeed, a defining characteristic of humanity is complex hierarchical social structure. As testaments to this fact, our brains have evolved extraordinary amounts of neural circuitry for processing social interactions, and socioeconomic status is a top health indicator.
The relationship between social status, individual health, and social stability makes the scientific understanding of social hierarchy in animals critical to human medicine and psychology. As such, model organisms (lab rodents in particular) have been major assets to scientists studying the neurological basis of social dominance behavior.
Move Aside! The Tube Dominance Test
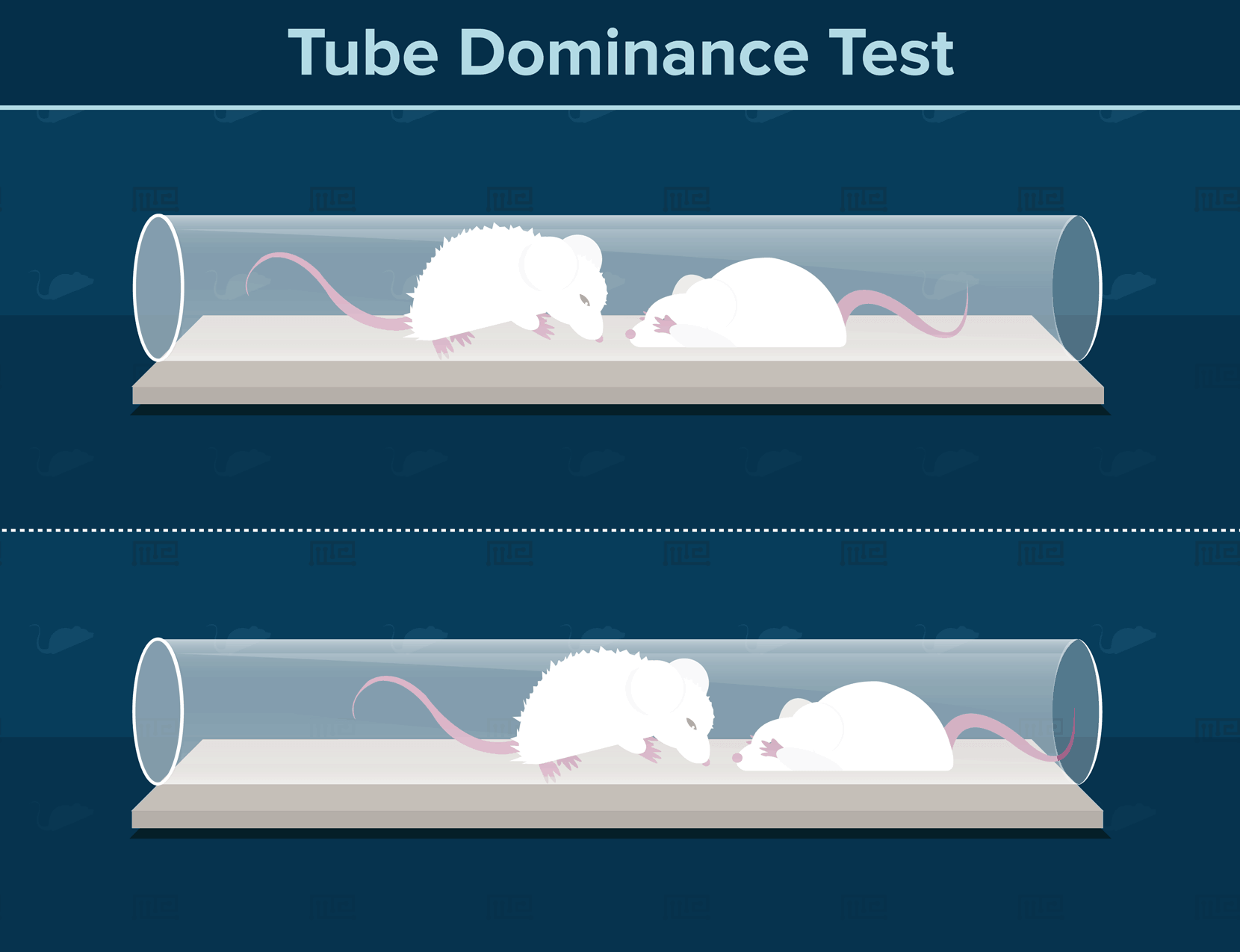
Perhaps the most basic paradigm for establishing social hierarchy in mice is the tube dominance test[3]. If you have ever walked through a narrow hallway or aisle, and considered whether a person coming in the opposite direction will move aside for you, then you know the basic principles of this social dominance test. Mice are initially trained to walk through tubes narrow enough to allow only a single individual to pass through. The experimenter then encourages two mice to enter a tube from opposite ends, such that one individual must retreat for the other mice to continue. The more dominant individual will consistently force the more subordinate individual to retreat in a series of trials. This test of social dominance can be applied to successive pairs of mouse individuals in a laboratory population, to determine the overall social hierarchy.
Tube Test Winners Display Other Dominance Behaviors
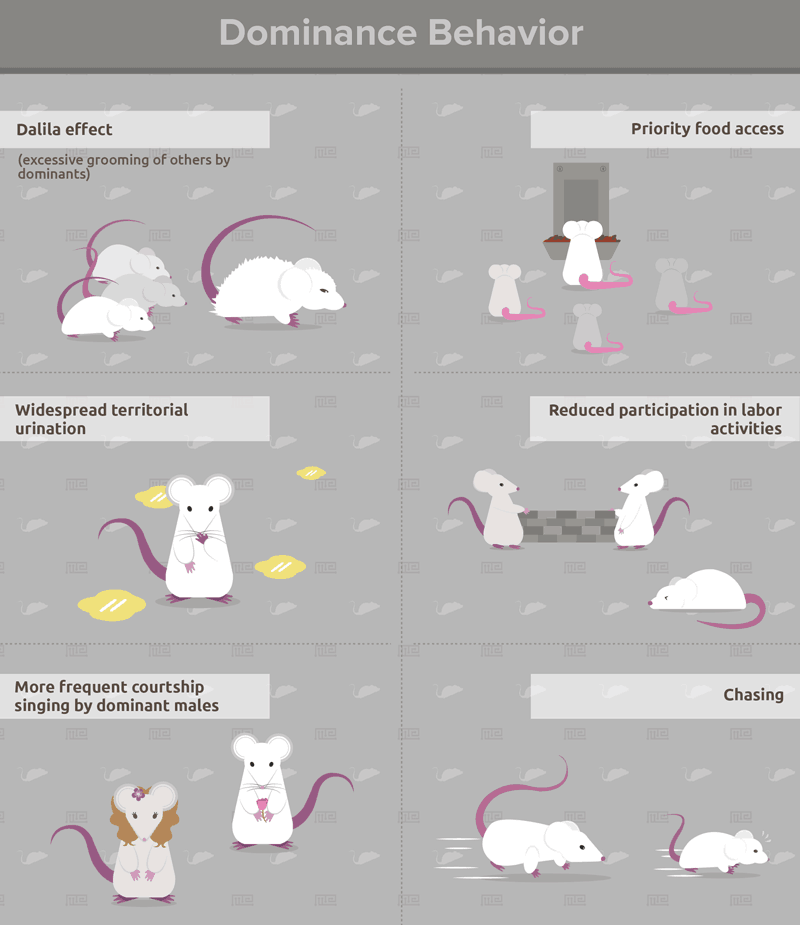
Individuals with long winning streaks in the tube test exhibit behaviors that typify dominance in mice. These include the Dalila effect (excessive grooming of others by dominants), priority food access, widespread territorial urination, reduced participation in labor activities, more frequent courtship singing by dominant males, and other agonistic behaviors such as chasing[4]. Whether the tube test is performed before or after other dominance tests (see below) does not affect these correlations[5]. As such, the tube test is unlikely to impose an artificial hierarchy, which may be an issue with other paradigms such as social defeat. Overall, the tube dominance test is a highly reliable predictor of dominance behavior in lab mice.
Nevertheless, certain factors such as whether individuals have had prior exposure to the test, and whether the test population has had time to establish a stable hierarchy, must be controlled when performing the tube test. Other confounding variables may also be present, particularly when manipulating genetic background or other treatments. For this reason, it is generally advisable to evaluate dominance in mice using multiple sociability test paradigms[4].
Other Dominance Test
The tube dominance test corresponds to the tendency for dominant individuals in wild rodent populations to win social conflicts, but there are also paradigms for other social behaviors in mice associated with dominance.
A visible burrow system (VBS) can be used to study dominance by controlling food resources. This monitoring system consists of a large surface area connected by tunnels to black Plexiglas chambers so as to mimic a burrow environment. A group of mice under experimental observation will be given limited food resources. The more dominant individuals in the hierarchy will gain weight because they have first access[6].
Participation as a ‘parasite’ rather than a ‘worker’ in a labor division test is also a high status trait. This test is run in a modified Skinner box with two individuals such that one individual must activate a mechanism to release food or water to the other. The more dominant mouse will tend to refrain from labor, acting as a parasite to the more subordinate partner[7].
Resident-intruder paradigms test for social aggression. In this test, an unfamiliar individual (intruder) is introduced to the habitat of a larger individual (resident), and the aggressive behavior of the resident is quantified. More dominant individuals will tend to be more aggressive, though not always. Since mice have social plasticity, aggression is often suppressed under stable social group settings (Mooney et al. 2014)

A similar set-up is used in the social defeat paradigm. In this case, the focus is on the intruder mouse, which has been subjected to several rounds of social defeat by the larger resident. Typically, the defeated individual will be more inactive and socially avoidant (characteristic signs of depression), but higher ranked individuals will show increased resilience to these effects[8].
Controlled laboratory settings can also be used to quantify other dominance behaviors such as which individuals groom or trim the whiskers of others excessively (Dalila effect)[9]. The dominance rank of male mice can be assessed by quantifying courtship vocalizations when exposed to a female[10], or by the amount of urine deposited following aggressive encounters with other males[11].

All of these other paradigms for testing social dominance correlate well with performance in the tube dominance test[4].
The Prefrontal Cortex is the Primary Regulator of Social Dominance
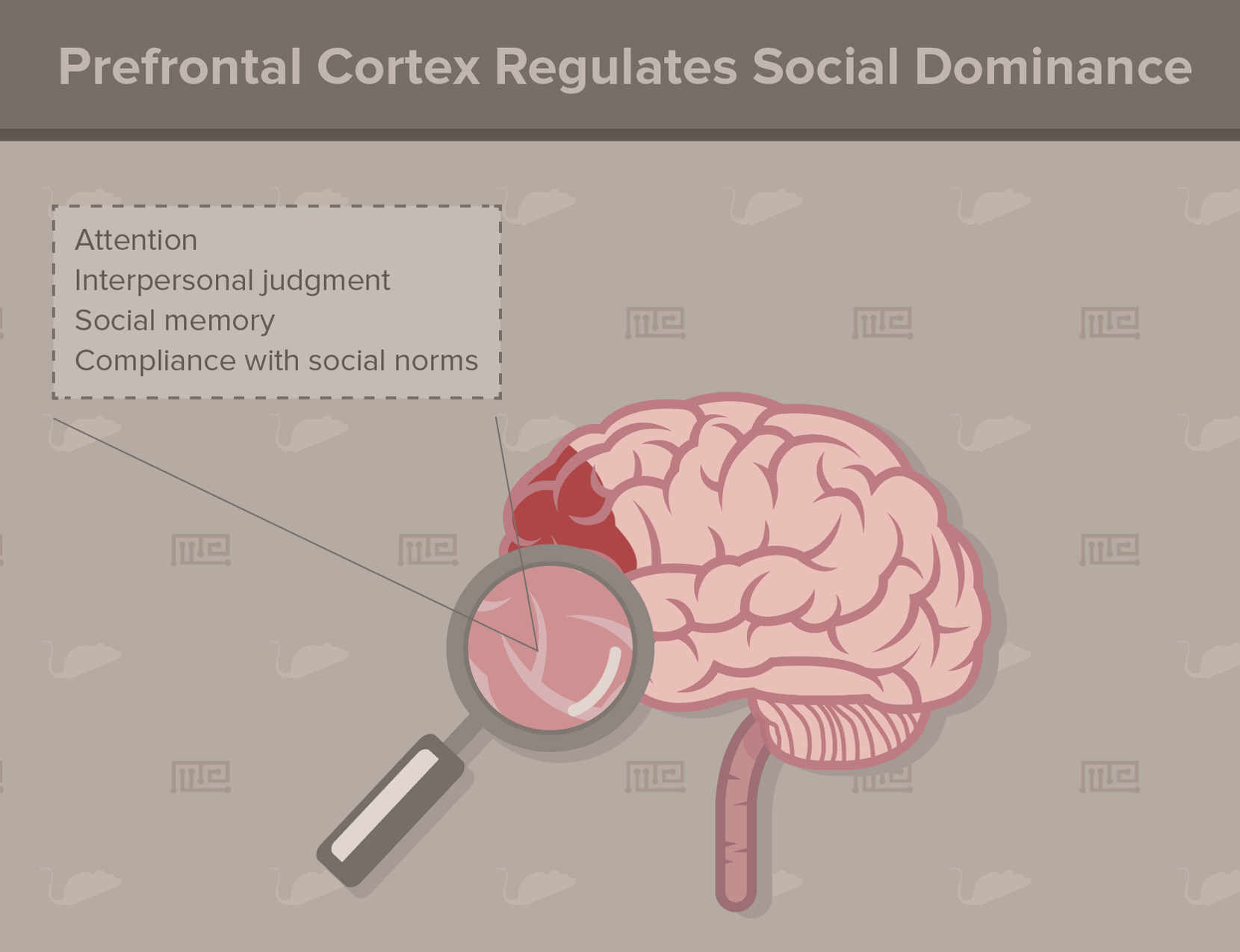
For animals with a dominance hierarchy, the prefrontal cortex (PFC) predominantly mediates social status recognition[12]. According to studies in humans, other primates, and rodents, the PFC controls attention, interpersonal judgment, social memory, and compliance with social norms. In some primates, even the size of the rostral PFC correlates with social rank. Brain lesions in this region (such as from injury or disease) impair social hierarchy processing. In rats, lesions introduced to the PFC increase subordinate behaviors, indicating a lowered social rank.
A couple of elegant studies have directly manipulated the expression of key neuronal receptors in the mouse PFC to solidify the causal link between animal social hierarchy and this brain region.
Knockout and rescue of cholinergic receptor function in the medial PFC correspondingly reduces and restores motivation for social interaction[13]. Wang and colleagues (2011) were able to control synaptic plasticity in the medial PFC through virally expressed glutamate receptor subunits and signaling proteins. Remarkably, the resulting changes to dominance rank (as indicated by the tube test and male courtship singing) were bidirectional.
In other words, changes in synaptic activity correspond to changes in social rank. This correlation indicates that social dominance behaviors are directly tied to synaptic plasticity in the PFC. Experimental manipulation of synaptic activity is also a close approximation of the natural changes underlying social plasticity in mice.
Other brain regions that likely affect social dominance have downstream connections to the PFC. For example, the basolateral nucleus of the amygdala (BLA) is responsible for emotional learning and memory, and is physically larger in people with more complicated social networks. The Dorsal Raphe Nucleus (DRN), as the primary site of serotonergic signaling, regulates aggression, while the hypothalamus and periaqueductal gray (PAG) may regulate other agnostic behaviors. Social reward pathways may also activate in response to winning social conflicts, and so could serve as a dominance hierarchy reinforcing mechanism.
Social Status and Psychosocial Stress
Though many of the key mechanisms underlying social hierarchy in animals have been established using lab rodent models, much about how social dominance impacts health remains unclear.
For example, socioeconomic status (SES) is a human approximation of social rank that has a strong inverse correlation with chronic stress and mortality[14], but the neurological mechanisms underlying this relationship are understudied[15]. Behavioral interventions for managing psychosocial stress, such as physical activity or social integration, are not easily standardized across individuals. They also do not address SES factors associated with income, education, or occupation. On the other hand, pharmaceutical treatments are often expensive and have adverse side effects. As such, clinical and therapeutic treatment of chronic stress will likely be improved with further study of the neurological and genetic mechanisms underlying dominance behavior[4].
Animal models continue to be promising systems for studying causal links between brain function and stress-related aspects of mental wellness (e.g. anxiety, depression, addiction). In particular, further research is required on how neurological plasticity can be controlled to alter brain function, both through behavioral and pharmaceutical intervention[15]. Taking the example from Wang and colleagues (2011) above, if social rank in mice can be changed by manipulating synaptic plasticity, does this correspond to a measurable change in anxiety, depression, or susceptibility to disease?
The health applications of social dominance research even extend to other (non-human) animals. As human societies continue to expand, conservation biologists must determine how to mitigate human-caused disruption of social structure in endangered species[16]. The humble laboratory mouse serves as a vital model not only for our own psychosocial health, but also for many of the most vulnerable and socially complex animal species, from great apes to great whales, with which we share our planet.
References
- Wilson, E. O. 1975. Sociobiology: The New Synthesis, Belknap Press of Harvard University Press
- Tinbergen, N. (1963). On aims and methods of ethology. Z. Tierpsychol. 20, 410-433. doi:10.1111/j.1439-0310.1963.tb01161.x
- Lindzey, G., Winston, H., Manosevitz, M. (1961). Social dominance in inbred mouse strains. Nature. 191, 474-476. doi:10.1038/191474a0
- Wang, F., Kessels H. W., and Hu, H. (2014). The mouse that roared: neural mechanisms of social hierarchy. Trends Neurosci. 37, 674-682. doi:10.1016/j.tins.2014.07.005
- Wang, F., Zhu, J., Zhu, H., Zhang, Q., Lin, Z., Hu, H. (2011). Bidirectional control of social hierarchy by synaptic efficacy in medial prefrontal cortex. Science. 334, 693-697. doi:10.1126/science.1209951
- Arakawa, H., Blanchard, D. C., Blanchard, R. J. (2007). Colony formation of C57BL/6J mice in visible burrow system: identification of eusocial behaviors in a background strain for genetic animal models of autism. Bhav. Brain Res. 176, 27-39. doi:10.1016/j.bbr.2006.07.027
- Masur, J. and Benedito, M. A. (1974). Genetic selection of winner and loser rats in a competitive situation. Nature. 249, 284. PMID:4833250
- Mooney, S. J., Peragine, D. E., Hathaway, G. A., Holmes, M. M. (2014). A game of thrones: Neural plasticity in mammalian social hierarchies. Soc. Neurosci. 9, 108-117. doi:10.1080/17470919.2014.882862
- Strozik, E. and Festing, M. F. (1981). Whisker trimming in mice. Lab. Anim. 15, 309-312. doi:10.1258/002367781780953040
- Nyby, J, Dizinno, G. A., Whitney, G. (1976). Social status and ultrasonic vocalizations of male mice. Behav. Biol. 18, 285-289. PMID:999582
- Desjardins, C. Maruniak, J. A., Bronson, F. H. (1973). Social rank in house mice: differentiation revealed by ultraviolet visualization of urinary marking pattens. Science. 182, 939-941. PMID:4745598
- Goldman-Rakic, P. S. (1996). The prefrontal landscape: Implications of functional architecture for understanding human mentation and the central executive. Philos. Trans. R. Soc. Lond. B: Biol. Sci. 351, 1445-1453. doi:1098/rstb.1996.0129
- Avale, M. E., Chabout, J., Pons, S., Serreau, P., De Chaumont, F., Olivo-Marin, J. C. et al. (2011). Prefrontal nicotinic receptors control novel social interaction between mice. FASEB J. 25, 2145-2155. doi:10.1096/fj.10-178558
- McEwen, B. S. and Gianaros, P. J. (2010). Central role of the brain in stress and adaptation: Links to socioeconomic status, health, and disease. Ann. N. Y. Acad. Sci. 1186, 190-222. doi:10.1111/j.1749-6632.2009.05331.x
- Marmot, M. G., Smith, G. D., Stansfeld, S., Patel, C., North, F., Head, J., White, I., Brunner, E., Feeney, A. (1991). Health inequalities among British Civil-Servants: the Whitehall II Study. Lancet. 337, 1387-1393. PMID:1674771
- Whitehead, H., Rendell, L., Osborne, R. W., Wursig, B. (2004). Culture and conservation of non-humans with reference to whales and dolphins: review and new directions. Biol.Conserv. 120, 427-437. doi:10.1016/j.biocon.2004.03.017