Description
The MazeEngineers Zebrafish T maze allows for choice experiments. Arms are baited with colors or cues and zebrafish are allowed to make choices in experiments. Enrichment boxes can be added to allow for stimuli to drive these choices. Multiple sizing and shapes available. Drain provided upon request alacarte and can be lifted from the ground or table to allow for easy drainage. Please inquire for more details.
Price & Dimensions
T Maze (Cross)
$ 1390
Per Month- Length: 70cm
- Width: 50cm
- Height: 10cm
T Maze (Symmetrical)
$ 1390
Per Month- Length: 50cm
- Width: 50cm
- Height: 10cm
Documentation
Introduction
The T-maze is an operant task widely used in multiple species. In zebrafish, the T-maze has been used to study color discrimination (Colwill et al., 2005, Avdesh et al., 2012), memory (Braida et al., 2014, Echevarria et al., 2016), locomotion and exploratory behavior (Peitsaro et al., 2003, Vignet et al., 2013), and place preference (Swain et al., 2004). Previous reports have shown that zebrafish exhibit long-term retention of spatial alternation (Williams et al., 2002) and active avoidance learning (Pradel et al., 1999) to color discrimination learning. Therefore, zebrafish display rapid and reliable conditioning well-suited for neurobehavioral tests.
The test requires a Plexiglas T-maze with variable configurations and sizes. Diverse protocols can be followed, including assessment of visual discrimination using colored or patterned sleeves in a designated goal zone or enrichment chamber in the arm of the maze. The investigator can use positive reinforcement with food rewards or environmental enrichment in the goal zone.
Experimental protocols typically entail measuring time to reach the goal zone or variation of percentage of correct choices to assess learning, or discrimination between pairs of visual stimuli. In the latter case, extinction and reversal protocols should follow an initial acquisition period to eliminate possible bias (Colwill et al., 2005, Avdesh et al., 2012). Training and testing duration, number and frequency of sessions, and number of trials per session can be modified to fit experimental goals.
The T-maze allows for rapid and reliable toxicology or pharmacological assays, as well as tests of the influence of genetic manipulation or genetic background. T-maze results in zebrafish should be regarded as complimentary to rodent models (Levin and Cerutti, 2009). The use of zebrafish enables the high-throughput study of neurodevelopment processes with little cost of experimentation and with the possibility of superior extrapolation due to being a vertebrate. Furthermore, the projections and function of discrete neural systems can be assessed.
Apparatus and Equipment
The equipment required to perform this test in zebrafish primarily entails a transparent Plexiglas T-maze. We recommend using a maze design either in cross (70 cm x 50 cm x 10 cm) or symmetrical (50 cm x 50 cm x 10 cm), although the dimensions can be modified (Darland and Dowling, 2001, Colwill et al., 2005). Removable opaque Plexiglas doors should be used to block off the arms of the maze from goal and start zones.
Colored or patterned (horizontal vs. vertical) sleeves can be inserted on the walls of the maze along the arms or only in the goal boxes. Choice of colors should take into consideration results from Avdesh et al. (2012) showing aversion to blue and preference of green and red over yellow. The maze should be filled up to a height of approximately 8-10 cm with either tank water (deionized water and sea salts; Braida et al., 2014) or a mixture of filtered tap water pretreated with conditioner and tank water (Colwill et al., 2005). Water temperature should be maintained at around 25.5 ºC-28.5 ºC (Colwill et al., 2005, Braida et al., 2014). Colwill et al. (2005) used a 25-W heater positioned on the floor of the maze. Home tanks should be provided with constant filtration and aeration. A stopwatch should also be used to time trials. If the study includes rewards, stainless steel tweezers should be used to administer food into the appropriate goal zone.
Training Protocol
T-maze protocols can test variation of latency to reach the goal zone comparing different genotypes or the effect of a drug, or color/pattern discrimination including acquisition, extinction and reversal periods. Discrimination reversals are recommended – i.e., a crossover design in which the location of the reward and/or side of color (green and red) or pattern (horizontal and vertical stripes) is counterbalanced. Consistent with other neurobehavioral approaches, experimenter-blind testing is critical (Colwill et al., 2005).
Training
Darland and Dowling (2001) allowed the fish 5 min to freely explore the maze in initial habituation. Conversely, Braida et al. (2014) subjected the fish to two habituation trials of 1 hr every day for 3 days, which served to reduce procedural novelty stress and handling stress. On the other hand, Colwill et al. (2005) performed two sessions with two trials each of exposure to the food rewards in the absence of colored sleeves. Multiple fish can undergo these trials simultaneously (Braida et al., 2014). The size of the groups should be gradually decreased afterwards, until testing is individual (within 3-4 days). The total number, frequency and number of trials of individual training sessions can be adjusted. Colwill et al. (2005) performed 4 trials per session, with 2 trials having the green sleeve on one arm and the purple sleeve on the other arm, whereas, on the other two trials, sleeves location was switched. Braida et al. (2014) performed two individual training trials at an interval of 24 hr. If performing color or pattern discrimination, half the fish should have the goal box to the left and the other half to the right, with subsequent reversal. Each fish should be placed in the start box for 2-5 min with its door closed until being allowed to explore the maze. The door should be immediately closed after the fish leaves the start box (Colwill et al., 2005). Ten minutes can be defined as limit to reach the goal box (Braida et al., 2014). If using food reward, it should be delivered upon a correct choice is made. Fish should be netted 30 sec after choice is made and returned to start box or to home tank at the end of the session. The experimenter should record whether or not the fish ate on correct training trials.
Testing
During acquisition of discrimination, the investigator should record a) the latency to reach the goal box and stay for at least 20 sec; and b) the color/pattern associated with the choice. Number of correct trials can also be used. Similarly to the training protocol, the fish should be returned to their home tank after each session. At the end of the acquisition protocol, the investigator should perform a test trial. However, depending on the study’s aim multiple trials can be performed in specific time-points to assess learning.
If assessing discrimination between two stimuli, extinction tests should follow. Colwill et al. (2005) performed 7 tests similar to the ones described in acquisition (with the same results being recorded), but with no food rewards. Preference for the side that had the food reward is expected to decrease gradually (Colwill et al., 2005). Reversal protocol should subsequently be performed, so that the choice previously incorrect is now rewarded. Colwill et al. (2005) performed the first reversal training session the day after the final session of extinction. Procedure and results recorded were as above. As described for acquisition, a final test trial should be done.
This protocol enables the assessment of whether the zebrafish show color discrimination in the choice of the arm with the goal box, whether that choice declines when food rewards are discontinued in extinction, and whether that preference reverses when the reinforcement is switched to the other color/pattern in discrimination reversal.
Modifications
Alternatively to color or pattern discrimination, the experimenter can opt for reinforced choice by providing an enrichment chamber containing deep water, artificial grass, and marbles (Darland and Dowling, 2001).
The T-maze test, has been extensively used in research, including in studies with fly (Jiang et al., 2016), mouse and rat (Lalonde, 2002).
Sample Data
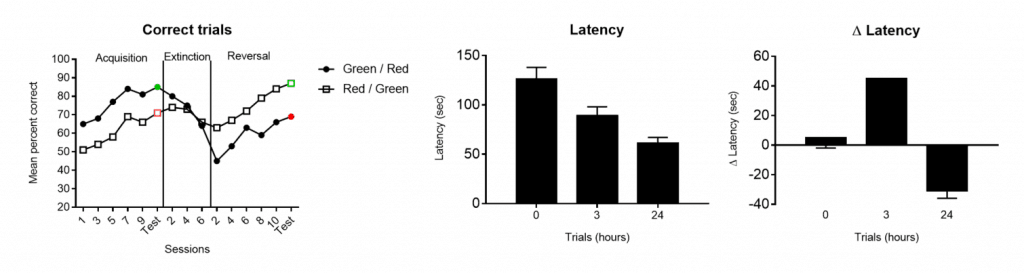
Percentage of correct trials (A) latency to reach the goal zone (B) and/or change in latency relative to initial trial (C) are the most commonly shown results when performing the T-maze test. Data can be shown as variation across test days including extinction and reversal periods in tests of visual discrimination (A) or as group averages on multiple trials (B and C) to evaluate the effect of learning or of a drug/toxic compound. Results in B and C are shown as mean ± standard error of the mean:
Strengths & Limitations
Strengths
The T-maze is a high-throughput test that allows for rapid screening of toxic and therapeutic candidates (Peitsaro et al., 2003, Swain et al., 2004, Echevarria et al., 2016), as well as assessing the influence of genetic manipulation or different genetic backgrounds (Darland and Dowling, 2001).
The use of zebrafish enables the study of neurodevelopment processes while affording superior extrapolation as a vertebrate. Furthermore, the projections and function of discrete neural systems (their proliferation, differentiation and migration) can be assessed. In addition, experimental throughput is significantly increased while the cost of experimentation is decreased in comparison to mammals, as fish are easily bred in great numbers, develop rapidly and do not require a large housing space.
Limitations
In the T-maze, caution should be taken with color choice, as it can influence results (Colwill et al., 2005, Avdesh et al., 2012). In addition, inter-strain variability has been shown (Vignet et al., 2013).
In comparison to mammals, neurobehavioral studies in fish do not enable the unraveling of the role of frontal cortical and hippocampal structures in learning and memory,
Summary and Key Points
- The T-maze in zebrafish is a high-throughput assay that enables rapid screening of toxic and therapeutic candidates, as well as testing the influence of genetic manipulation or diverse genetic backgrounds.
- This test offers the possibility of studying color discrimination, memory, locomotion, exploratory behavior and place preference.
- The equipment required consists of a T-maze with variable configurations. Colored or patterned sleeves can be used to assess visual discrimination; reinforcement can be included using either food rewards in goal boxes or a reservoir with environmental enrichment
- The experimental protocol typically consists of a training period where the fish should learn the location of the rewarded/enriched zone, followed by test trials. If assessing discrimination between colors or patterns, the acquisition period should be followed by extinction and reversal periods in a counterbalanced design.
- Results are typically reported as percentage of correct trials, latency to find goal zone/enrichment chamber with or without assessment of visual discrimination, or change in latency relative to initial trial in order to assess learning.