Product Description
A rodent Y-maze is a type of behavioral test used to assess spatial working memory and reference memory in small animals such as rats and mice. The Y-maze consists of three arms that form a “Y” shape, and each arm is usually made of clear plastic. The floor of the maze is usually made of a smooth and non-slip material such as metal or glass. The test is typically conducted by placing the animal at the end of one arm and allowing it to explore the maze. The animal’s movements are then tracked, and different measures such as the number of arm entries, the time spent in each arm, and the number of errors made can be used to assess the animal’s memory and spatial navigation abilities. The Y-maze test is a simple and inexpensive way to assess spatial working memory and reference memory in rodents and has been widely used in neuroscience research.
The Y maze is similar to the T maze, except with three arms at 120 degrees to each other. The rodent or mouse starts at the end of one arm, then chooses between the other two. Spontaneous alternation is measured to demonstrate learning.
Maze Engineers offers the Y Maze Test.
Backlights

Stand

Escape Tubes

Rat (8cm diameter): $400
Cue Lights

Food Wells
Used for Mouse or Rat
Dimensions: To fit
Cost $100
Water Sealant
Used for Mouse or Rat
Dimensions: N/A
Cost $100
Doors
Used for Mouse or Rat
Dimensions: To fit
Cost $100
The Y-Maze is a modified version of a T-Maze. Unlike the T-Maze that has sharp turn angles, the Y-Maze provides a more natural turn of the arms (120 degrees in the Y-maze compared to 90 degrees in the T-Maze). The natural tendency of rodents to explore new environments serves as the basis of the Y-Maze task. This exploratory task involves many parts of the subject’s brain including the hippocampus, septum, basal forebrain and pre-frontal cortex. Many types of research involving neurological diseases focus on the hippocampus damage to it is believed to affect cognition and spatial learning. Thus Y-Mazes are part of the widely used behavioral tasks in the assessment of spatial learning and memory.
The Y-Maze, just like the T-Maze, offers only two choices to the subjects. In a baited Y-Maze task, once the subject has retrieved the reward from one of the arms, it is expected to visit the next arm on the next trial. This simple behavioral task tests cognitive function based on the ability of the subject to remember spatial locations requiring hippocampal-dependent reference memory. Administration of certain compounds or a disease model can affect this ability.
Spence and Lippitt (1946) used a simple Y-Maze as an experimental test for the sign-gestalt theory of trial and learning that was suggested by Tolman. The Y-Maze goal arms were baited with water in one arm, and food rewards in the other. Subjects that were either thirst or food-deprived were used to put to the test Tolman’s idea of learning. Dember and Fowler (1959) studied alternation behavior using Forced Alternation and Spontaneous Alternation tasks and suggested their findings in the T-Maze can also be replicated on a Y-Maze. Since then the Y-Maze has been used in studying various aspects of spatial learning, memory, and cognition.
The Y-Maze is usually a symmetrical capital ‘Y’ shaped maze with the arms spaced at an angle of 120 degrees. This construction makes the Y-Maze ideal for the Continuous Alternation protocol. Modifications such as guillotine doors and escape tunnels can easily be added to the maze. The Y-Maze can also be adapted for investigation of spatial learning specifics in different animals and disease or intervention models.
Origin
In 1946, Spence and Lippitt made use of a simple Y-Maze that had one arm baited with water and the other arm baited with food. Their experiment was an investigation into the ongoing debate among psychologists regarding Thorndike’s law of effect and Tolman’s theory of learning. Spence and Lippitt’s experiment attempted to translate the theoretical representation of simple trial-and-error learning as suggested by Tolman. In their experiment, rats were divided into two groups and given 12 days’ experience of thirst motivation in a simple Y-Maze. The maze had one path that led to water and the other to an empty box for one group and food in the case of the other group. Their experiment’s result was in disagreement with the Tolman theory as both the groups chose the arm baited with water.
In the late 1950s, Dember and Fowler researched the alternation behavior in animals. Their series of published papers described spontaneous vs. free alternation behaviors and the influence of rewards and reinforcement on these tasks (Dember and Fowler 1959, Fowler et al.,1959a, Fowler et al.,1959b). Since then, the Y-Maze and T-Maze have been developed as relatively simple and have been widely used for assessing spatial memory.
In 1961, Hellyer and Straughan tested the hypothesis that alternate behavior is learned by first pre-training food and water-deprived rats on a rewarded straight runway or tabletop and then testing them on a baited Y-Maze. The resulting analysis showed that animals trained on the tabletop alternated significantly more than those trained on the straight runway. The data also revealed that the animals trained on the tabletop exhibit a relatively high percentage of alternation at the outset but alternate progressively less with trials. The researchers, based on the results, concluded that the effects of preliminary training were temporary.
Developments
The Y-Maze also serves as a simple tool in understanding the role of different parts of the brain in spatial learning. In 1986, Cazala tested the effects of electrical stimulation of certain brain areas of mice and subjected them to a spatial discrimination task in a Y-Maze. Each subject received one electrode implanted in the following structures: the medial hypothalamus, the medial lemniscus, the lateral tegmentum, the reticular formation, the dorsal part of the mesencephalic central gray area and the lateral hypothalamus. In the Y-Maze trials, one-half of the animals had to learn to self-stimulate by interrupting the photocell beam situated in the right arm, whereas the other half had to go to the left arm. The results showed that the animals were able to discriminate between the reinforced arm and the non-reinforced arm of the Y-maze to self-administer the stimulation.
The effects of chronic stress on spatial memory and cognition were investigated by Conard et al. in a Y-Maze that had extra-maze visual cues only. The results of the test revealed that chronic stress impaired spatial memory in the subjects. Interestingly, however, stressed females showed an improved performance in the latter minutes of the maze in contrast to the stressed males who continued to visit the arms at random. Further investigation of chronic stress by Conard and Wright (2005) showed that chronically stressed rats performed well in the intra-maze cue version of the Y-Maze. The subjects spent a similar amount of time exploring both the arms in the traditional Y-Maze while in the cued Y-Maze the rats spent more time exploring the novel arm. This behavior supports the hypothesis that chronic stress leaves novelty-seeking behavior intact while impairing spatial memory.
Recent Developments
The role of cerebellar cortex in conditioned goal-directed behavior was studied by Burguière and colleagues (2010) using a water Y-Maze. They tested heterozygous transgenic L7-PKCi mice in a stimulus-dependent water Y-maze conditioning task resulting in the conclusion that the mutant mice were able to acquire the stimulus-response association though they showed reduced optimization of motor performance. This result suggested that PKC-dependent process in cerebellar Purkinje cells is required for optimization of motor responses.
A 2012 study by Bai et al., showed the anticipatory activity in rat’s medial prefrontal cortex using a Y-Maze task. The study aimed to explore the encoding, by the neuronal ensemble, of a working memory event. A similar study was also conducted by Yang et al. using a two-choice spatial delayed alternation task in a Y maze.
Yin et al. studied the effects of paradoxical sleep deprivation on the Y-Maze task performance. Using Forced Alternation, they evaluated the short-term spatial working memory of the subjects. Although initially, the mice showed no impairment, after 7 days of deprivation, working memory impairment was observed.
The apparatus is a 3-arm maze shaped like a capital ‘Y’ with each arm spaced at an angle of 120 degrees. The arm lengths range between 30 to 50 cm and are usually symmetrical, although asymmetrical mazes are also used. The pathway widths are approximately 10 cm. The maze is usually used as an enclosed maze with approximately 30 cm high walls and the entire maze is elevated to a height of 50 cm from the floor. The two-goal arms may contain a well with a food reward; alternatively, the goal arms may contain an exit tube that allows the animal to escape the maze (Deacon 2013). Generally, there are guillotine doors at the entrance of each arm that can be used to confine the animal to a specific arm or prevent the animal from entering one of the goal arms. The maze is usually a dark color to prevent the animals from feeling extra anxiety while performing the task. Intra-maze cues can also be used to help animals distinguish and remember goal arms.
Fully automated Y-Mazes are also available which detect the location of the subject within the maze and control the opening and closing of the arm doors as a trigger response. The automated maze is also capable of detecting food rewards.
The Y-Maze can also be adapted to include the benefits of water mazes and used for rodent models (Burguière et al.,2010, Deacon 2013). The Y-Maze has also seen adaptations as an aquatic model (Natt et al.,2017, Abreu et al.,2017) and for use with arthropods ( Jaworski et al.,2017, Simonnet et al.,2014).
To avoid shadows in the maze, the Y-Maze should be well lit from above. Proper lighting also ensures that the subject is able to see the food rewards. Tracking software such as Noldus Ethovision XT, mounted above the maze assist with live scoring, tracking and recording the subject and its movements within the maze. The apparatus must be cleaned thoroughly before and after each trial to limit influence from any residual stimuli from previous trials.
Training Protocol
The Y-Maze has been widely utilized in assessing spatial memory and learning in animals, usually in control versus disease model or intervention group. The task involves observation of the ability of the subject to remember the previously visited arm. The task takes advantage of the natural explorative nature of rodents and the idea that they tend to use an optimal search strategy to obtain food with minimal effort.
The performance in the task weakens or decreases for subjects with hippocampal damage as seen in neurodegenerative diseases or as a result of natural aging.
Several protocols exist to be used with the Y-Maze, though the two most commonly used protocols are the Rewarded Alternation task and Spontaneous Alternation task. For the Rewarded alternation task, the experimenter decides which arm is the “correct choice” whereas the Spontaneous Alternation task uses the subject’s natural explorative drive and allows it to choose which arm to explore first. Spontaneous alternation task has been shown to be more successful in the assessment of subjects with hippocampal lesions as these animals tend to develop a side preference (Deacon and Rawlins 2006). The task is also used to quantify cognitive deficits in transgenic strains of mice and evaluate effects of compounds on cognition.
Another protocol used with the Y-Maze is the Delayed Alternation task. The Delayed Alternation task allows assessing spatial working memory by first allowing the subject to explore a baited arm of its choice and removing the subject once the choice is made and limiting it to the start box of the base arm using a door. This trial is followed by a formal trial, after a predetermined delay between the two trials, wherein the subject is reintroduced to the maze by opening the start arm door and is expected to choose the arm that it did not choose in the previous trial.
Pre-training for the Y-Maze
Rodents tend to be wary of eating anything new. Thus it is important to familiarize the subject with the food rewards used, prior to the testing, within a familiar environment (housing cage). Food rewards can be solid (such as sweetened breakfast cereal) or liquid (such as chocolate milk.) Generally, liquid rewards are used in case of compond testing as some compounds may make eating solid food an unpleasant experience for the subject. Subjects are usually maintained at about 85 to 95% of their free-feeding weight throughout the training and testing phases.
For the food-deprived task, the food-scavenging behavior is encouraged by depriving the subject of food the night prior to the testing. Subjects may be familiarized to the maze in pairs (usually cage-mates) to reduce the anxiety in a novel space during the initial phase of exploration. The acclimation is usually done individually, although, this may require more time than paired familiarization. The maze is scattered with food rewards, and the subject is placed in the start arm and allowed to explore the maze freely. The food rewards are replaced on consumption. The familiarization procedure is repeated four times with at least ten minutes between each exposure. The acclimation period usually lasts 1 to 2 days. Next, one of the arms is blocked, and the open goal arm is baited with a food reward. During this forced trial the subject is placed in the start arm and forced to visit the open arm. This trial is repeated by randomly varying the closed arm, for an equal number of trials for each arm, until the subject has been familiarized with the task.
Evaluation of Spatial Memory using Rewarded Alternation in the Y-Maze
The choice trial begins by baiting both the arms and allowing the subject to visit only one arm and consume the food reward. This procedure is done by pre-selecting the correct choice and blocking it with a door. Now the unvisited arm becomes the correct choice in the next trial. For the immediate trial both the arms are open and only the correct choice arm is baited. The subject is expected to visit the correct choice arm. If it makes the right choice, it is allowed to consume the food reward, and if it chooses the wrong arm, it is allowed to see that the food well is empty and then it is removed from the maze.
Each trial lasts approximately for 2 minutes. The procedure is repeated for each of the animals on a ten trial per day basis for up to twelve testing days. The correct choice arm is randomly varied throughout the testing sessions.
Evaluation of Spatial Learning using Spontaneous Alternation in the Y-Maze
The Spontaneous Alternation task is based on the novelty of the maze. Thus the task is performed without prior familiarization of the subjects to the maze. The subject is given a free choice, during the trial, to choose either one of the baited arms unlike in the Rewarded Alternation task where one of the arms is blocked. Once the subject has made its choice, it is confined to that goal arm by closing the respective door for 30 seconds. The subject is removed from the goal arm after it has consumed the food reward and the doors in the maze are opened. The subject is then once again placed in the start arm and is expected to alternate its choice from its previous selection. If the subject visits the unvisited arm, it is allowed to consume the food reward or else, in case it visits the already visited arm it is allowed to see that the food well is empty before it is removed from the maze.
Each trial lasts no longer than 2 minutes, and the procedure is repeated for each of the animals on a ten trial per day basis for up to twelve testing days.
Modifications
Similar to the T-maze, Y-Maze has also seen modifications and adaptations to meet the needs of various cognitive neuroscience research to allow studying different aspects of spatial learning and alternate behavior.
A simple modification to the maze can be done using discriminative stimuli such as patterns or objects to which the subject must respond to, in order to obtain a reward. For example, Wright and Conrad (2005) used large enamel-painted rocks as intra-maze cues. Such cues ease the task difficulty and assist the animals in remembering which goal arms they have visited. The subjects can be trained to only choose the arm with the specific rock or cue.
Other variants of the Y-Maze include the water Y-Maze that is a hybrid between the Morris Water Maze and the traditional Y-Maze. Deacon (2013) used a shallow paddling pool Y-Maze as a relatively non-aversive environment for testing mice. The maze included false escape tubes with only one true exit. Another water Y-Maze variant was used by Burguière et al.,2010) to test the role of the cerebellar cortex in conditioned goal-directed behavior. The maze arms were filled with water and used two different conditioned stimuli: a light or a sound. A wrong turn resulted in air puff which acted as an aversive stimulus.
Aquatic models of the Y-Maze also exist for research using fish. Aoki et al. used an automated aquatic Y-Maze that used computer controlled visual cues on an LCD screen placed under the Y-Maze tank. The zebrafish were trained to avoid one arm by pairing the floor color with an electric shock. Aquatic Y-Mazes have also been in studying shoals (Ward et al.,2011). Drosophila Y-Mazes also exist and allow evaluation of chemosensory responses and other behaviors of drosophila.
Multiple Y-Mazes (Botwinick et al.,1963, Ainge et al.,2007) provide a multi-choice complex maze for assessing memory performance and spatial learning. Elevated Y-Mazes are often employed for testing anxiety or stress related performance of animals. Fully automated Y-Mazes are also available which detect the location of the subject within the maze and control the opening and closing of the arm doors as a trigger response. The automated maze is also capable of detecting food rewards.
The Y-Maze can easily be adapted and modified to be used with birds, insects and other animals for assessment of spatial working memory and cognitive abilities. The ease of construction and the simplicity of the apparatus make it one of the popular behavioral assays.
Data Analysis
The data obtained from the Y-Maze is generally very straightforward and consists of the number of correct (the animal enters the opposite arm on the second run) vs. incorrect (the animal enters the same arm previously entered on the previous run) arm entries in each trial. The time it takes the animal to retrieve the food reward can also be recorded. As the animal learns that entering a new arm results in a food reward, the number of incorrect arm entries should decrease. The percentages of correct arm choices can be graphed and compared across a sham control group and a disease model/intervention group.
Using graphs to compare arm entries between different disease or treatment groups allows easy visualization of the effect on spatial memory and learning. Animals in the controls groups should show significant improvements in their correct arm choices. Animals as disease models of neurodegenerative disorders, for example, should show a much slower learning curve with more incorrect choices, even after several trials. Generally, animal cohorts of 10-30 animals are sufficient to obtain p-values of <0.05 using ANOVA (Wright & Conrad 2005, Conrad et al.,2003).
Strengths and Limitations
The Y-Maze allows for easily reproducible results due to its simplicity as compared to other behavioral tasks that test spatial learning and memory. Symmetrical Y-Mazes allow for continuous alternation tasks and usually require minimal training and testing time. Unlike the Morris Water Maze Test, there is minimal stress placed on the subject in a conventional Y-Maze. However, the Y-Maze can be adapted for stress and anxiety related task (Water Y-Maze and Elevated Y-Maze). The Y-Maze is a highly adaptable and modifiable task that allows research into different aspects of spatial learning and cognition. The absence of significant stressors and familiarization with the maze prior to testing enable better observations of working memory in the animals as they perform in the maze.
Although simple to construct and easy to use, the Y-Maze has its own limitations. A simple Y-Maze is a single-point two-choice maze thus it has a higher rate of success possibility as the probability of the subject choosing the correct arm is 50% by default. The subjects may also use strategies other than spatial learning based on the spatial and non-spatial cues. Odor trails too may affect the quality of research if olfactory cues are not an intended part of the investigation.
For the task to deliver appropriate results, the subject’s exploratory drive must be maintained throughout. Extensive handling and overtraining of the subjects may place undue stress on them, affecting their performance on the maze. It is also important to maintain minimum variability in the amount of reward used during the training and testing lest the subject should suffer from ‘contrast effects’ wherein its motivation decreases due to receiving less than expected reward (Deacon & Rawlins 2006). In case of Y-Mazes that make use of guillotine doors, it is essential that doors are closed carefully and not dropped too close to the subject to avoid startling it as this experience may be stressful for the subject and cause it to avoid that arm in subsequent trials.
As with all mazes that measure aspects of learning and memory, it is important to remember that many different processes play into the behavior in the maze. In many cases, the Y-Maze is used in conjunction with other mazes to study disease models or transgenic animals and gain a fuller understanding of spatial learning and memory.
Summary
- Y-Maze is a modification of the T-Maze, in which the arms are placed at a natural angle (120 degrees) unlike the sharp 90-degree turn of the T-Maze arms.
- The natural turns make the Y-Maze ideal for Continuous Alternation task.
- The task exploits the innate explorative nature of rodents and subjects them to tasks that require them to alternate between the goal arms to retrieve food rewards.
- Commonly used protocols with the Y-Maze are Forced Alternation task and Spontaneous Alternation task.
- The T-Maze can be easily adapted to investigate the different aspects of spatial learning and for different subjects.
- By using guillotine doors, the Y-Maze can be easily adapted for Delayed Alternation protocol.
- The Y-Maze has been extensively used in the study of hippocampal functions, age-related cognitive decline and anxiety.
- The Y-Maze is also utilized in understanding the effects of compounds and in understanding underlying pathology of diseases on spatial learning and memory.
- Subjects of the diseased model show a much slower learning curve in comparison to the control group in Y-Maze tasks.
Mouse Y Maze (CM)
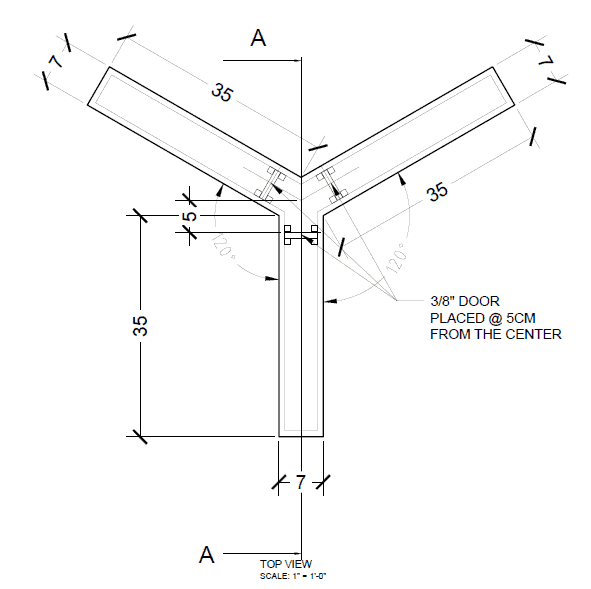
Mouse Y Maze (CM)
- Single Arm Width: 5
- Single Arm Length: 35
- Single Arm Height: 20
Rat Y Maze (CM)
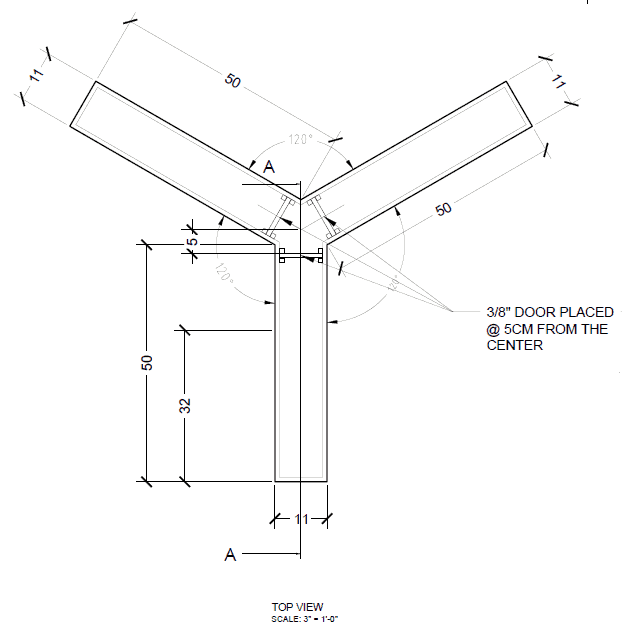
Rat Y Maze (CM)
- Single Arm Width: 10
- Single Arm Length: 50
- Single Arm Height: 30
Request a quote
"*" indicates required fields