Product Description
The conditioned place preference chamber is a paradigm widely used to explore the reinforcing effects of natural and stimuli. Combinations of floor and cues are available.
In this variant, subjects are allowed to freely move between a compartment in which they were conditioned with either cues or neutral cues. The different cues provide visual reinforcement.
This dual chamber place preference allows for biased and unbiased conditioned place preference testing. A removable door (not shown) allows isolation into one compartment of the apparatus of the subject. Preference testing is then done by removing the door to allow the mouse to freely explore between the two compartments.
Visual patterned inserts are sandwiched between a clear acrylic interior layer and the outer grey layer, protecting from general wear and tear.
Optional Standing Inserts are supplied for spatial place preference procedures. Please request separately.
Variations



Automated shock plates can be inserted anywhere in the CPP chamber
Automated heat plates can be inserted anywhere in the CPP chamber
Automated feeders/pellet dispensors can be inserted any point in the maze
Automated lickometers can be inserted any point in the maze
Variants
- All
- Accessories
- Activity
- Ant
- Anxiety and Defensive Behavior
- Automated
- Balance
- Bat
- Bees
- Behavior
- BehaviorCloud
- Bird
- Canine
- Classic
- Cognition
- Conductor Software
- CPP
- Custom
- Cuttlefish
- Decision Making
- Drosophila
- Environmental Enrichment
- Fungi
- Historical
- Human
- Insect
- InventionUp
- Labyrinth
- Learning and Memory
- Modifications
- Navigation
- Octopus
- Odor
- Operant
- Operant Builder
- Pain/Analgesia
- Pig
- Planaria
- Plant
- Poeciliid
- primate
- Robotics
- Sheep
- Social Behavior
- Spatial Navigation
- Spotlight
- Tech Transfer
- Trims
- Virtual Reality
- Visual Cues
- Zebrafish
Similar apparatus
Price & Dimensions
Sizing
Mouse
$ 1890
+ Shipping and Handling (approx $150)- Default dimensions: total (cm): Width 46 x Depth 27 x Height 30 (customizable)
- Compartments (cm): Width 20 x Depth 18 x Height 30
- Corridor (cm): Width 20 x Depth 7 x Height 30
- Doors (cm): Width 8 x Height 30
- Default double walls: Grey(Exterior)/Clear (interior) (customizable in color and pattern)
- Acrylic
- Easy clean with 70% Ethanol
- No odors
Rat
$ 2290
+ Shipping and Handling (approx $300)- Default dimensions Total(cm): Width 86 x Depth 47 x Height 40 (customizable)
- Compartments (cm): Width 40 x Depth 34 x Height 40
- Corridor (cm): Width 25 x Depth 13 x Height 40
- Doors (cm): Width 10 x Height 40
- Default double walls: Grey(Exterior)/Clear (interior) (customizable in color and pattern)
- Acrylic
- Easy clean with 70% Ethanol
- No odors
Introduction
The conditioned place preference (CPP) is a widely used behavioral model which can evaluate the motivational properties such as the rewarding and aversive effects of natural substances as well (Suzuki 1999). Furthermore, it has been used to study the neural mechanism underlying the conditioned reward. Traditionally, the rewarding properties were assessed by the conventional self-administration method. In the late 1970s, the CPP paradigm revolutionized the methodological procedure to evaluate the motivational properties to compensate for the methodological and interpretative ambiguities associated with the self-administration model. Since then, CPP has become one of the most frequently used models even surpassing the conventional self-administration method. Most importantly, it has opened avenues for understanding the neural mechanism of rewarding and aversive effects of substances.
In place conditioning tasks, animals are introduced to an apparatus having two distinct chambers, either through a doorway or a smaller connecting chamber. Distinctions are made between these two chambers based on visual and tactile cues, including wall color and floor texture, but in some cases, other elements such as olfactory cues may also be used.
Subsequently, the affective state of the rodents is altered by the administration of a drug, or change in a physiological state. On alternate days, the drug or physiological state is paired with the other environment. After this conditioning trial, the subjects are free to explore all the chambers, and they may depict an increase or decrease in the time spent in the chamber that was previously conditioned with the substance. A conditioned place preference (CPP) is said to occur if the subjects spend considerably more time in the pharmacological-paired compartment than the vehicle-paired compartment. This shift in preference may be attributed to the rewarding properties of the substances or the physiological situation that has evoked an approach response (Schechter & Calcagnetti 1993).
On the other hand, if the subject spends a considerably longer time in the vehicle-paired compartment rather than the pharmacological-treated compartment, then this is considered a conditioned place aversion (CPA). This reduced time spent in the pharmacological-conditioned chamber may be attributed to the aversive properties of the substances or the physiological situation that has evoked an avoidance response.
In CPP, the primary stimulus (drug) serves as an unconditioned stimulus (UCS). When it is paired with a secondary stimulus (visual, tactile, or olfactory cue) that acts as a conditioned stimulus (CS), an approach or avoidance behavior for the paired environment is elicited. Pharmacological-induced CPP is based on the principle that when a primary reinforcer is paired with the secondary stimulus, the conditioned stimulus attains secondary reinforcing properties, which are apparently established due to a Pavlovian contingency. Interestingly, the CPP paradigm is not only restricted to substances, but the approach or aversive behavior can also be established using food (Bechara et.al 1992; Swerdiow et.al 1983), copulation (Miller et.al 1987), or water (Agmo et.al 1992) as primary reinforcers.
Important/ Interesting facts
2.1. Interesting facts
The first-ever study regarding place preference was conducted to examine the drive-reduction hypothesis of learning in rodents, and Beach 1957 utilized a Y-choice maze for the procedure; an apparatus different from the modern-day CPP apparatus (see section 3 for currently used CPP apparatus).
2.2. Closely Related Task
2.3. Major Contributions made towards the task/apparatus
Many researchers have made significant contributions toward the CPP task, and a variety of different CPP apparatus have been used for studying the rewarding properties of drugs.
Some of the prominent contributors are:
- BEACH H.D– Conducted first study discussing place preference NELLO A. ROSSI and LARRY D. REID– Introduced the concept of drug-paired conditioning.
- MARTIN D. SCHECHTER– Over 25 published papers. Also reviewed were the trends in conditioned place preference in detail.
- Christopher L Cunningham– Over 75 published studies discussing the conditioning place preference task.
2.4. Applications in other laboratory animals
The CPP model is highly applicable, and a number of other laboratory animals besides rodents have been tested for the rewarding effects of different substances. For example, pharmacologically-induced CPP has been demonstrated in chickens (Hughes et al. 1995). Chicken’s highly evolved visual system is sensitive to color and fine detail. Hughes et al. 1995 used visual stimuli as the CS to establish the CPP. Although originally designed for rodents, presently CPP behavior can also be evaluated in zebrafish by utilizing the Zebrafish Place Preference Test.
Apparatus and Equipment
3.1 Apparatus Design
Traditionally, there are two types of CPP, namely the two-chamber design or the three-chamber design. The two-chamber design comprises two discrete compartments separated by a sliding door. In the three-chamber design, two large compartments are connected by a small central compartment usually called “the neutral chamber”. In contrast to the two-chamber design, the three-chamber design consists of two doors present at the entry of the two large chambers from the central chamber. However, sliding doors can also be used instead of guillotine doors. The sliding doors can be lowered or raised to gain access to each compartment. Each side chamber has characteristic visual and tactile cues. However, the locomotor activity of the subjects can also be recorded with the help of a video tracker such as Noldus Ethovision XT or ANY-Maze.
3.1.1. Two-chamber design
During the conditioning period for the two-chamber design, the subject is introduced into one of the compartments after the incorporation of the substance, and it is confined there for a specific period of conditioning with the doors shut. On the test day, the subject is allowed to explore both compartments freely, and the time spent in each compartment is recorded. This data is compared with the baseline preference for the chambers to determine the final place preference (Cunningham et al. 2006).
3.1.2. Three-chamber design
In the three-chamber design, the subject is introduced into the neutral chamber post-treatment and is allowed to access only one chamber during the conditioning period. On the test day, the subject is again placed into the neutral chamber and allowed to explore all the chambers freely for the determination of final place preference (Smith et al. 2016).
3.1.3. Two-chamber Versus Three-chamber Design
The added chamber (neutral chamber) in the three-chamber design is the only physical difference between both designs. This neutral chamber has its advantages and drawbacks as well. Its biggest advantage is the ease it provides to the experimenters, which permits them to place the subjects in the center of the apparatus; a major problem with the two-chamber design as it is difficult to place the subject in the exact center of the apparatus. Conversely, the neutral chamber gives interpretational difficulties when the subjects make an association with the neutral chamber; they may show preference or aversion for the neutral chamber instead of the other two chambers. The two-chamber design is preferred in this regard as it excludes this pitfall by leaving the subject with two possible choices for the association.
3.2 Mazeengineers Apparatus Designs
Need Custom CPP variation? – Mazeengineers got you covered! We can make any custom CPP variation. If you want to replicate an apparatus or plan to start from scratch, Mazeengineers is always there to handle your science. All we need from you is an overview and dimensions, and the rest will be taken care of.
Documentation/ Scientific Research
4.1 History
4.1.1. Origin
The first documented work on place preference was way back in 1957. Two papers were published that year discussing place preference by Beach et al. He trained rats in a Y-choice discrimination box and associated one of the boxes with the treatment of reward. Although this study did not explicitly stress the rewarding properties of drugs and instrumental responses involved, it surely had some conceptual similarities to the current versions of place conditioning methods. However, the study assessed evident place preference.
On the other hand, Garcia et.al 1957 investigated that avoidance behavior can be produced by irradiation where conditioning criteria involve consumption. Their apparatus consisted of a straight alley enclosed by an acrylic lid. They divided the alley into distinct compartments with the help of an acrylic divider. Each compartment was distinct in terms of visual and tactile cues. One compartment was painted flat black and had a grid floor, while the other was painted flat white and had a mesh wire floor. The alley’s base was balanced on a fulcrum, and a micro-switch was located beneath each end for data collection.
These published works were followed by limited research on place conditioning as only nine papers were published during the span of 1958-1974. The currently used apparatus and procedure were established by Kumar in 1972, followed by Ross and Reid in 1976. Kumar modified the procedure by introducing the subjects to the testing chamber right after substance incorporation. Beach and Kumar administered a high dose of daily pain treatment to the subjects before the CPP test, and the same high dose was also employed for CPP induction, the subject-conditioned responses may have been elicited by the relief of withdrawal rather than the rewarding effects of the substance under study.
In 1976, Rossi and Reid developed the concept of pharmacologically-paired conditioning to measure pharacologically-induced affective state (approach or avoidance behavior) as a measure of the rewarding properties of substances. They not only introduced the concept of pharmacologically-paired conditioning, but they were able to induce a conditioned response in rats at low doses of pain treatment productively. They used an alley that was divided into three distinct compartments each separated by the middle compartment having doors on each side. The middle compartment was gray, had a solid floor, and served as a starting point for the animals during the testing procedure. On one side of the middle compartment was a black compartment with a wire mesh floor, which they referred to as the black side. On the other side of the middle compartment was a white compartment with a wire mesh floor, which they referred to as the white side. They claimed that the CPP paradigm could successfully access the substance’s affective consequences and rewarding properties.
4.1.2. Developments
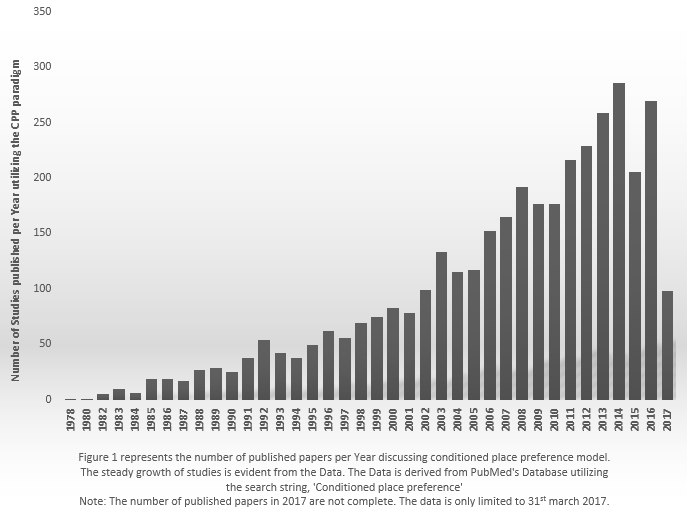
Since then many studies probed the rewarding properties of many agents, and even natural reinforces as well. According to PubMed’s database, 3693 search results are obtained starting from 1978 to the present by utilizing the search string, “Conditioned place preference”, and the number is increasing (see Figure 1).
The three-chamber design is the most extensively used type of CPP; other apparatuses vary from this design by having a different number of compartments (e.g., two or four compartments) and shapes. The two-compartment apparatus requires a forced choice, whereas a three-compartment area offers a central choice area between the experimental chambers. There are also paradigms designed to assess place preference within an open field or allow for the association of the interoceptive effects of drugs with a unique environment.
Training Protocol/ How to use the apparatus
4.2.1. Experimental Design
The two major experimental designs used to condition animals under the influence of substances or natural reinforcers are called the Biased and Unbiased experimental designs. (Schechter & Calcagnetti 1993)
4.2.2. Biased Design
In the biased procedure, subjects are allowed to explore the CPP chamber for several days to determine their baseline place preference between the two compartments without the administration of substances. Rodents have the inherent ability to prefer dark over light, so they will choose a compartment with less light every time. This preference, if significant, may confound the interpretational results. However, this problem can be alleviated by balancing the stimuli between the two compartments. For instance, by fluctuating the intensity of external light reaching the two compartments, or by changing other sensory modalities that would diminish the subject’s approach such as a dab of vinegar in a dark environment.
Using this biased design, the subjects choose a less-preferred compartment and a more-preferred compartment. For the conditioning procedure, the animals are paired with one of the environments; a substance thought to have rewarding properties is paired with the less-preferred environment, and the control treatment is paired with the more-preferred environment. This conditioning process is repeated for some days with multiple pairings of drugs with the less-preferred side and an equal number of pairings of control treatment with the more-preferred side. In order to determine the final place preference, the subjects (on a control treatment) are allowed to explore all the compartments once again freely.
On the test day, if the subjects spend more time in the previously less-preferred environment during their baseline preference test, it can be inferred that the agent produces rewarding properties sufficient to elicit an approach response, thus depicting a place preference.
4.2.3. Unbiased design
In the unbiased design, the subjects are trained to associate an environment with one or the other discrete state, i.e., after either agent or control treatment disregarding their initial preference for the stimulus cues present in one or the other environment. In their next session, they are trained to associate the stimulus cues of a second environment with the other condition. The design inherent in this procedure is to balance the number of subjects that receive one treatment in the first environmental pairing with the number of subjects that receive the second treatment in that first pairing. Following this design, the same subjects receive the opposite treatment on the second day of training.
The process comprises two days that can be repeated a number of times after which the final place preference is determined by permitting the subjects to explore both environments freely. As the agent and control treatments are randomized between each of the two contexts, the choice of a preferred context during testing does not specify a bias at the time of baseline testing, which is usually seen in the biased design.
In addition, a third method can be employed, saline-injected control subjects are used to determine CPP. In this method, one group of subjects is conditioned to one of the two environments, while the control group is given saline in both environments. CPP is then determined as the difference in duration spent in the drug-paired environment for conditioned animals compared to the saline-injected controls on the test day. The pre-testing phase for baseline place preference prior to conditioning may or may not be employed for this method. (Bardo et al., 1995)
4.2.4. Biased Versus Unbiased Design
The researchers have the choice to make the chambers either biased or unbiased depending upon their needs and ease. The biased design can be taken as a design in which the subjects prefer one chamber over the other during habituation. Conversely, in the unbiased design, the subjects prefer each chamber equally during adaptation. In general, the unbiased design is more advantageous as it permits researchers to determine CPP for the chamber paired with drug treatment on the test day. Furthermore, the data can be analyzed with great ease as the researcher simply notes and records differences in chamber preference as compared to the animal’s initial level of equal preference for both chambers during adaptation. (Lucke-Wold, 2011)
Detailed Procedure of Conditioned Place Preference
4.2.2.1. Pre-training for CPP
The subjects are allowed to access all compartments of the apparatus for several days in order to eliminate the novelty-seeking behavior as it can be a confounding variable. CPP task, whether biased or unbiased follows the timeline of the pre-test, Conditioning, and Testing Phase. The pre-test may be separated from conditioning by 1-3 days; however, conditioning and the post-test day usually take place on consecutive days.
4.2.2.2. Pre-Test/Habituation Phase
The subjects are allowed to explore all the compartments freely. During the pre-testing phase, baseline place preference is assessed and recorded. Baseline data is determined in terms of the average amount of time spent in each compartment over 3–5 days.
This procedure is the same for both biased and unbiased designs. The main aim of this phase is to determine the baseline place preference so that it can be exploited later. In the case of a biased design, the baseline preference will serve to determine the less and more preferred side. Conversely, for unbiased design, this baseline preference data will be used to compare results obtained after the final place preference.
4.2.2.3. Conditioning Phase
4.2.2.3.1. Conditioning for Unbiased Design
On the first day of the conditioning phase, the subject should receive treatment and then be placed into a specific compartment for 30 minutes. On the next day, the subject should receive control treatment and be placed into the opposite compartment for 30 minutes. This alternate scheme of treatment and control treatment should be repeated for eight days. The locomotor activity is recorded with the help of the photocells.
4.2.2.3.2. Conditioning for Biased Design
The subjects are initially divided into equal groups. One group should be conditioned on their non-preferred side (as determined by the baseline place preference) whereas the other group is randomly assigned to the side in which their respective conditioning should take place. All subjects from both groups are then randomly divided into groups that either receive drug or control treatment. The conditioning period for biased design is usually repeated for four days. On the conditioning day, each subject is injected with its respective treatment and confined into the pre-determined chamber for a period of 30 minutes. After the conditioning procedure, the subjects are safely removed from the conditioning chamber and placed into their home cages.
4.2.2.4. Testing Phase
In the testing phase, the final place preference is recorded. The subjects should be placed in the central chamber after the administration of control treatment and allowed to explore freely for 15 to 30 minutes. This testing procedure is repeated on three consecutive days. The final place preference is recorded as a mean of the locomotor activity and the time spent in each compartment following the three testing days.
The time spent in each compartment can either be recorded manually or automatically. For the automatic collection of data, two prominent techniques i.e. the use of microswitches, or a video tracking device are employed. The latter technique involves the placement of the CPP apparatus on a fulcrum in such a fashion that the weight of a rat in one end compartment causes the micro switch to close, thus resulting in automatic time calculation. Whereas the former technique simply involves a video tracker such as Noldus Ethovision XT or ANY-maze to collect data. Furthermore, the base of the chambers is embedded with a photocell beam for automated data collection (see our automated CPP chamber)
Strength and Limitations
4.6.1. Strengths
- The most promising advantage of the CPP paradigm is its ability to assess both the preference and aversion of the substance within a single test.
- The substance may have effects beyond the rewarding/aversive domain that may influence the time spent by the subject in the previously paired side.
- This problem is resolved by the CPP model as it determines the baseline and final place preference in an animal that is drug-free.
- The motivational properties of the different substances can be assessed by minimal quantities (low doses) when compared to other behavioral methods that assess motivational properties. This capacity makes CPP one of the most sensitive behavioral tests.
- While there are superior methods of assessing factors that contribute to addiction, namely self-administration, CPP is a simple and much more accessible approach to measuring reward function.
- It is amenable to several manipulations and could be used to test both subject’s place preference and aversion to certain substances. There is also less stress on the subject as treatment is administered.
- One of the greatest advantages of the CPP is that it is well-suited for evaluating locomotor activity concurrently with the reward. Furthermore, it also helps to investigate the neuronal circuits involved in reward.
- It is still one of the very simple and dynamic models that have been developed in an attempt to understand the complexities of addictions clearly.
- CPP is preferred over the self-administration experiments in terms of the dose-effect curves. CPP characteristically yields a monophasic dose-effect curve, whereas self-administration experiments yield inverted U-shaped dose-effect curves. The CPP task is highly preferred because it is completed within a short period of time.
- The likelihood exists that a CPP or CPA can be shown in as meagre as one pairing, and when different pairings are utilized, these pairings can be directed either more than once or twice a day without diminishing the overall strength of conditioning.
- The equipment needed to regulate the CPP model has turned out to be exceptionally refined with devices that not just permit automation of the time spent on a specific side, but additionally, the capacity to quantify the actions of subjects while it is physically present in a specific environment with the help of video trackers such as Noldus Ethovision XT or ANY-Maze
- The applications of the CPP model overshadow the equipment expenses as the results are highly reproducible. The subjects can be tested in a drug-free state, where there is no disruption of their behavior, additionally, the impacts of pharmacological antagonists can be determined without disruptions.
- Incorporating the antagonist amid conditioning and just later testing for potential impacts of the antagonist upon the agonist used to condition the subject, allows a reliable measure of the agonist-antagonist associations to be resolved.
- Furthermore, this task can be adjusted to evaluate the rewarding properties of different physiological states, e.g., access to a sexually responsive mate or to another juvenile rodent as a playmate. The CPP test can also assist to determine the effects on physiological states.
- CPP gives a reliable measure of the rewarding and aversive properties of substances and other physiological treatments. This notion is further strengthened as the CPP’s verdict about the rewarding and aversive properties is also confirmed by other behavioral models that measure rewarding and aversive properties as well.
- Substances that have seemed to yield place preference in the CPP task have also shown rewarding properties in other behavioral models as well. In this way, substances, and physiological treatments, for example, access to sustenance, are able to produce CPP and have rewarding properties in operant behavioral models.
- On the other hand, agents such as lithium chloride, and physiological treatments such as irradiation depict CPA, and these treatments elicit avoidance behavior in other behavioral paradigms as well.
4.6.2. Limitations
- There has been a lingering criticism of CPP regarding what exactly is being evaluated by the CPP task.
- The likelihood exists that subjects are not depicting a drug-environmental paired preference, but rather are influenced by the locomotor activating and sedative actions of the substances.
- For example, the increased activity as manifested by the stimulant drug may change the interoceptive state of the subject in a specific environment in order to enhance the exploratory activity and, accordingly, uplift its acquaintance with the substace-paired environment.
- Conversely, after control treatment in a particular environment, the subject may associate less exploration with this environment.
- On the test day, the subjects will be well-suited to enter a place that they have explored comprehensively. A confounding variable associated with the CPP test dwells in the likelihood that the signs of the conditioning environment stay novel to the subject because of the drug effects.
- The subject may spend more time on the combined side during testing because of the novelty of encountering that side in a substace-free state.
- Shortfalls in performance produced by drug-state reliance might be viewed as another constraint innate in the CPP test. The animals are trained in one state in that they encounter the substance effects in a specific context and, later, they are tested in an alternate state (substance-free).
- The likelihood exists that the failure to express a preference for the substance-combined environment is innate in the way that training and testing happen in different interoceptive states.
- Another likely mechanism underlying the CPP response could be the anxiolytic properties of the substance as opposed to the rewarding effects of substances.
- So, if a substance decreases subjects’ neophobia to the least preferred side, it will then be slanted to spend more time on that side.
- The likelihood exists that the substance may be producing an avoidance behavior for the favored environment, while the substance meddles with the subject’s capacity to recollect its introduction to the substance-conditioned environment.
Summary and Key Points
- The conditioned place preference (CPP) is a widely used behavioral model which can evaluate the motivational properties such as the rewarding and aversive effects of substances.
- A conditioned place preference (CPP) is said to occur if the animals spend considerably more time in the paired compartment than in the vehicle-paired compartment.
- Rossi and Reid developed the concept of paired conditioning to measure substance-induced affective state (approach or avoidance behavior) as a measure of the rewarding properties of substances. In CPP, the primary stimulus serves as an unconditioned stimulus (UCS).
- When it is paired with a secondary stimulus (visual or tactile cue) that acts as a conditioned stimulus (CSS), an approaching or aversive behavior for the paired environment is elicited CPP continues to be very popular because of its numerous applications.
- Classically, the CPP apparatus is of two types, namely the two-chamber design and the three-chamber design. The two major experimental designs used to condition animals under the influence of substances or natural reinforcers are called the Biased and Unbiased experimental designs.
- CPP task, whether biased or unbiased follows the timeline of the pre-test, conditioning, and testing Phase.