The forced swim test (FST) is one of the most commonly used animal models for assessing depressive behaviors. The forced swim test involves the scoring of active movements such as swimming and climbing vs. passive immobile behavior while swimming in a cylinder from which there is no escape. A wide range of treatments has been shown consistently to reduce the amount of immobility time while increasing active escape behaviors.
- Product Description
- Prices and Sizes
- Modifications Available
- Documentation
4.1 Introduction
4.2 Apparatus and Equipment
4.3 Training Protocol
4.4 Modifications
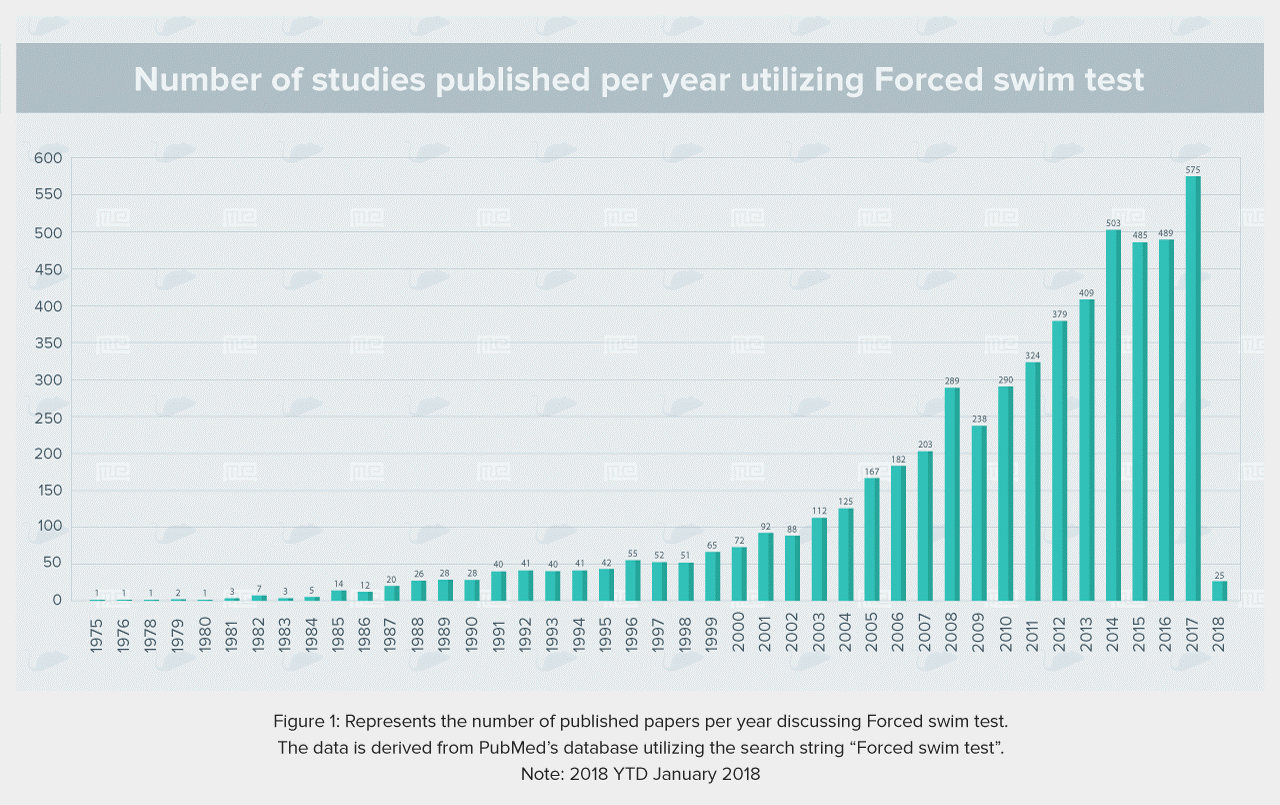
4.5 Sample Data
4.6 Strengths and Limitations
4.7 Summary and Key Points
4.8 References - Product Blueprints
5.1 Product Sizes
5.2 Product Images
Overview
The Forced Swim Test (FST) was developed by Roger D. Porsolt in the late 1970’s as a simple task to be used in the assessment of anti-depressive properties of compounds in small animals. Depression is one of the most prominent mood disorders affecting millions across the globe. The aetiology of depression is yet to be fully understood, and modelling symptoms of depression in animals tend to be difficult. However, many of the behavioral signs can be used as a basis for the measure of the effectiveness of drug treatments and other interventions.
The Forced Swim Test, also known as Behavioral Despair test, models learned helplessness by subjecting the animal to a stressful environment (a cylinder filled with water) with no escape. The animal on the realization that there is no escape from its situation resigns by adopting an immobility posture and makes minimal effort only to keep its head above the water. When treated, the animals show an increase in active escape behaviors. An animal that stops swimming behaviors early in the test is thought to be feeling hopelessness and is comparable to a human experiencing depressive symptoms. The behavioral parameter used to screen antidepressant activities is based on the idea that stressful life events play a crucial role in leading to depression and related behaviors.
The apparatus consists of a transparent Plexiglas cylinder that is filled with water to a height that does not allow the subject to touch its paws or tails to the floor nor does it permit escape by climbing over the walls.
History
2.1 Origin
This test was first used and described by Roger D. Porsolt in the late 1970s in his several publications (Porsolt et al.,1977a, Porsolt et al.,1977b, Porsolt et al.,1978). Porsolt and his colleagues showed for the first time that rodents could be used as models of depressive states and could be sensitive to anti-depressant compounds. When doses of compounds were administered before the forced swim test, Porsolt observed a decrease in behavioral despair and immobility of the animals (Porsolt et al.,1977a). In his later publication, Porsolt compared how different strains of mice behave in this test, with and without the administration of anti-depressive drugs (Porsolt et al.,1978). Since these initial papers, the FST has become a standard test for the evaluation of anti-depressive compounds.
2.2 Developments
In their paper published in 1981, Herman et al. evaluated the effects of various compoundss on the behavior of rats in the Forced Swim Test task. The investigation also studied the relation between the test performance and the inborn level of exploratory and locomotor activities. The investigation results showed that certain compounds prolonged the immobility phase whereas while others shortened it.
Shimazoe et al. suggested that objective measure of immobility can be performed by recording the vibrations of the wall due to escape directed behavior of the subject in the Forced Swim Test and comparing it with the locomotion of those in the activity cage. They modified the FST apparatus by adding detectors to the walls of the cylinders to record the vibrations for subjects tested with different drugs and compounds.
The influence of prenatal protein malnutrition was tested in the FST for females and males by Trzctńska et al. Two different experiments were conducted one of which tested the protein malnourished male and female subjects in the Forced Swim Test at the beginning and the end of a stress regime while the second experiment tested two separate groups of male subjects without the stress regime.
2.3 Recent Developments
A major amino acid from fenugreek seeds was investigated for its possible antidepressant-like effects in olfactory bulbectomized rats (Kalshetti et al.,2015). FST results showed that rats treated with the amino acid showed decreased immobility and increased swimming time.
Bergman et al. investigated if deep brain stimulations could potentially induce antidepressant-like effects in mice. After ventromedial prefrontal cortex deep brain stimulation, the results of the investigation showed that both wild-type and knockout mice showed a decrease in immobility in the Forced Swim Test, suggesting that deep brain stimulation can be a viable option in the treatment of subjects lacking a fully operational serotonin transporter.
Apparatus and Equipment
The Forced Swim Test employs a cylindrical tank of size varying between 25 to 50 cm in height and 10 to 30 cm in diameter, to hold rodents and small primates. The tank is filled with room temperature water to a level that prevents the subject from touching its paws or tail to the floor of the cylinder and prevents escape. Opaque dividers are often used within the tank to allow multiple subjects testing.
Although, live scoring is possible it is recommended to use a video and tracking software such as the Noldus Ethovision XTor ANY-Maze to avoid inaccuracies and variance in scoring. The tanks must be sufficiently lit to improve the quality of the recording, and a white noise generator can be used to mask any noise that may affect the performance of the subject.
Apparatus to Study Anxiety
Training Protocol
The Forced Swim Test assesses the depressive state of the animal in an anti-depressant versus sham control group, by observing their behaviors in the stressful environment. The subject typically exhibits struggling behaviors in an effort to escape the environment, however, after a while it will naturally reach a state of behavioral despair wherein it assumes an immobility posture by making small movements to keep its head above the water. This state of helplessness and immobility is seen to be reduced on treatment with anti-depressants and other compounds.
While outside stress on the animals should be avoided for this test, it requires no or very little prior training of the animals. An initial struggle followed closely by behavioral despair is a natural reaction of the animals placed in the forced swim apparatus.
Generally, a six-minute testing period is used although testing durations can range anywhere from 4 to 20 minutes depending on the mammal species. The entire test is recorded, and generally, a shorter portion of the test is analyzed by measuring the exact durations of specific behaviors. Of the typical six-minute test, usually, the last four minutes are analyzed. During the first two minutes, the animals are typically very active in their behaviors, and their initial movements can mask any potential effects of the treatment. It is only when the control mice reach a state of hopelessness and behavioral despair that the effects of treatment can be observed.
Evaluation of Depressive-like States in Small Mammals
The Forced Swim Test cylinder is filled with clean, room temperature water to an appropriate height. Dividers are put in at this point if there are being used. The subjects are administered the test substances 30 minutes before the test in case of intraperitoneal or subcutaneous injection and 60 minutes for oral administration. Video recording is initiated before placing the subject in the cylinder. The subject is placed in the tank by lowering it into it by holding its tail. Immobility behavior is scored during the last 4 minutes of the test; this includes short periods of slight activity by the animal to remain afloat.
On completion of the 6 minutes of the session, the recording is stopped, and the subject is removed from the tank by their tails, dried using towels and placed under a heat lamp before returning them to their home cage.
In the event of a subject failing to maintain swimming and floating behaviors it is removed from the tank and excluded from the experiment.
Modifications
Since its initial development and use by Porsolt in the late 1970’s, the Forced Swim Test has seen few modifications in the apparatus and the protocols.
In their 1987 paper, Shimazoe et al. suggested using detectors placed on the walls of the cylinder to detect and record vibrations arising from the movements of the subject within the task. They suggested that it provided an objective measure of the immobility seen when the subject reaches a state of behavioral despair.
Another improvement was suggested by Lucki (1997). They increased the depth of the water in the apparatus and used the time-sampling technique to score the animals’ behaviors. This allowed for categorizing the behaviors into two types: climbing and swimming. Thus enabling a convenient method for quantifying escape behavior. This also allowed researchers to observe a difference in behaviors caused by serotonin re-uptake inhibitors.
Data Analysis
The data obtained from the forced swim test is generally visualized by showing the time an animal spent performing different behaviors. The amount of time an animal spent swimming, climbing, mobile, or immobile can be obtained by analyzing the experiment recording with a stopwatch. Alternatively, a time-sampling method of scoring can be used where the experiment is divided into five-second intervals, and the animal’s activity during that interval is recorded. A general definition of mobility for this test includes any behavior other than those necessary to balance the body and keep the head above water (Cryan & Mombereau 2004). The time spent mobile and immobile can be easily graphed and compared across the intervention and sham control groups.
Using graphs to compare the amount of time different treatment groups spend immobile or mobile allows for easy visualization of the effect of the anti-depressant. Animals treated with saline should show increased immobility and decreased mobile movements. In contrast, those treated with an anti-depressant should show decreased immobility and increased swimming and climbing. Control animals treated with a stimulus of motor activity, such as caffeine, should show increased activity for the entire duration of the test. Generally, animal cohorts of 10-15 animals are sufficient to obtain p-values of <0.05 using a 1- or 2-way ANOVA, Dunnett’s post hoc, or Bonferroni’s post hoc tests (Can et al.,2012, Costa et al.,2013).
Translational Research
In the development of new therapeutics for stress-related disorders, the rough-and-tumble play has been shown to have great potential as suggested by the investigation conducted by Burgdorf et al. Their experiment’s data demonstrated a positive effect of rough-and-tumble in inducing resilience to stress in the subjects. In the Forced Swim Test, the subjects that were allowed 3 minutes of rough-and-tumble play for 3 days showed reversed stress-induced effects of chronic unpredictable stress.
Yuen et al. analyzed the quantitative translation of Forced Swim Test data to human efficacious does by using dose-response and time-course FST experiments. They used four common marketed antidepressants and compared the data to human doses. Based on their assessment they recommended that a concentration approach should be used in the prediction of clinically efficacious doses of new potential antidepressant agents.
Strengths and Limitations
The Forced Swim Test has shown significant effectiveness in validating the efficiency of antidepressant compounds as evidenced by the numerous published studies. The large collection of data compounded with the ease, reliability, and speed with which the test can be performed make it a suitable tool for drug discovery and research comparisons over a range of subjects and drugs/compounds used. The task does not require any pre-training and can be done for multiple subjects at one time by using dividers.
However, even a singular test places significant stress on the subject, which, over repeated trials may influence the resulting behaviors. The test models and observes a particular type of stress-induced behavior, which is believed to be related to depressive human feelings, but it is important to remember that these behaviors are caused by extreme stress (Petit-Demouliere et al., 2005).
While the forced swim test forces animals into a state of hopelessness comparable to human depressive feelings, it is important to note that this test is not equivalent to the entire spectrum of human depressive states. It remains unclear how similar human depression and the neurobiological mechanisms of behaviors seen in animal models are (Bourin et al., 2001). The observable outcome of this test is one-dimensional, and while it can determine if a compound has anti-depressive properties, it cannot determine the mechanisms by which that compound acts. The exception to this is seen in rats, where the difference between various compounds can be observed (Detke et al., 1995). It is also possible that the tested compounds may affect the overall activity levels of the animals, which could lead to unreliable results of the Forced Swim Test. It is therefore important to control the overall activity levels using an additional test such as the Open-Field test.
Summary
- The Forced Swimming Test was developed by Roger D. Porsolt in the late 1970’s as a simple task to assess and evaluate the anti-depressive properties of compounds in small animals.
- The task is also known as Behavior Despair or Learned Helplessness task as the subjects become immobilised denoting hopelessness.
- The task uses a cylindrical tank filled with water such that when the subject is placed in it, it is unable to reach the floor or escape the tank.
- Immobility includes any behavior necessary to balance the body and keep the head above water.
- The time-sampling technique is categorizing the behaviors into two types: climbing and swimming. This technique allows a convenient method for quantifying escape behavior.
- When treated with certain compounds there is a decrease in immobility behavior.