Description
Activity wheels can be used in neuroscience to assess the activity of the circadian system. Circadian rhythms are important biological processes that allow the adaptation of physiological systems to cyclic environmental changes. Gene expression, hormone levels, and many behaviors are all known to be regulated in a circadian pattern. Although these rhythms are usually robust, there are a number of genetic, pathological, and pharmacological factors that can affect them. The physiological consequences of these instabilities can potentially disturb many bodily functions and processes. A number of neurological, neurosensory, and neurobehavioral disorders have associated circadian rhythm disturbances (e.g. Roybal et al, 2007).
The apparatus used for this test is a cage with running wheels, equipped with a system to monitor their rotation. They are connected to software to record wheel running activity, allowing them to assess the animals’ activity. Animals must be single housed in a test cage to allow the assessment of their activity on the wheel.
MazeEngineers offers activity wheels with home cages. Inquire for more information
Features
Key Components
- Handheld device with capacitive touch screen to configure and record activity
- Rodent cage (1000 cm² ) with 1 running wheel per cage
- Wheel: Motorized or manual rotation
- Wheel diameter: mouse 20 cm and rat 40 cm
- Water bottle and food holder
- USB cable
Parameter Settings
- Test time: 0 – 9000 minutes
- Auto start and stop schedule setup
- Rotation direction: clockwise or counter clock
- Data model: count by distance or count by a circle
- Count by distance – using m for distance and m/s for speed
- Count by a circle – using r for distance and r/min for speed
Data Measurements
- Wheel Revolution – the number or revolution of the wheel by the animal
- Distance – animal run using the running wheel
- Duration – the time in minutes the animal used the running wheel
Data Store and Upload
- The handheld device stores up to 200 data sets
- Easy to browse the stored data
- Upload data to PC using a USB cable
Isolation Chamber
- Dual visible and IR light bulb.
- Sound (Hz) Detector
- Speaker Frequency: (100-40,000 Hz)
- Speaker Intensity 1-150 dB)
Optional accessories
- Shock or air puff
Conductor Integration
- Take advantage of Neuralynx, Ethovision Integration, SMS and Email integration with the Conductor Science Software. No I/O Boxes Required
Passive Activity (optional)
- The motor drives the wheel and forces animal to run
- Speed: 0.1 – 99.9 r / min with step 0.1 r / min
- Acceleration: 0.1 – 100 r / min 2
Price & Dimensions
Activity Wheel with Home Cage
$ 2990
Per Month- Wheel diameter: 20cm
- Cage
- Overall Dimensions: 13.9”L x 9.25″W x 7.875”H (35.3 23.5 x 20 cm)
- Weight: 5.8 lbs.
- Wheel Diameter: 5.0” ID (12.7 cm)
- Run Distance: 0.40 meters/revolution
- Wheel Drag: < 3 grams
- Camera Holder: Ideal with DMK 22AUC03. Please inquire with us about your camera model and we can customize the holder for you.
- Color: Grey
Documentation
Introduction
The use of activity wheels for circadian rhythms is based on the fact that when rodents have free access to a running wheel in their home cage, voluntary use of the wheel will depend on the time of day (Pittendrigh & Daan, 1976). Running-wheel activity is the most commonly used and reliable method for assessing the status of the master circadian clock, the suprachiasmatic nucleus (SCN) of the hypothalamus (Verwey et al, 2013).
Activity wheels allow for a non-invasive and simple assessment of circadian rhythms that can be used to study the effects of a specific treatment, of the knockout of a specific gene, or can be used in genetic screening to identify new factors influencing circadian activity (e.g. Godinho et al, 2007).
The test apparatus consists of cages with running wheels, equipped with a system to monitor the animals’ running activity, and a controlled tight-light chamber, which allows the control of light/dark cycles and the maintenance of the animals in an effectively dark environment.
Apparatus & Equipment
The apparatus used for this test is a cage with running wheels, equipped with a system to monitor their rotation. They are connected to software to record wheel running activity, allowing them to assess the animals’ activity. Animals must be single housed in a test cage to allow the assessment of their activity on the wheel.
Given that circadian activity is primarily controlled by light, it is fundamental that the wheel-running cage is placed in a light-tight 12h-light/12-dark environment. A controlled tight-light chamber, which allows the control of light/dark cycles and the maintenance of the animals in an effectively dark environment, can also be also included in the apparatus. The chamber includes an internal light source that can be controlled by an external timer. The walls of the chamber are matte black to prevent reflection artifacts that create changeable illumination. A light monitor is also included in the chamber, allowing to check the lighting conditions without opening the chamber and disturbing the animals. The chamber is properly ventilated.
Training Protocol
The purpose of the activity wheels for circadian rhythms is to evaluate the responses of the circadian system in rodents in a control vs. disease model/intervention group, by assessing their spontaneous running activity in different light patterns (Banks & Nolan, 2011).
When rodents have free access to a running wheel in their home cage, voluntary use of the wheel will depend on the time of day, according to their circadian rhythms. Assessing their spontaneous running activity allows the assessment of circadian rhythm disturbances potentially associated with diseases or interventions.
There are several versions of protocols to be used with activity wheels, varying mainly in the number of days during which the animals’ activity is monitored.
Pre-training for the activity wheel
Given that this test is used to assess circadian rhythms, all light/dark-affecting variables should be controlled. Animals should be habituated to the specific 12h-light/12-dark cycle to be used, ideally matching that of the animal house to reduce the amount of time it will take an animal to adjust to the circadian chamber.
Since social isolation is a stress factor, animals should also habituate to being single-housed.
Evaluation of circadian rhythms using activity wheels
1: Light/dark cycle
Record activity data from the animals for 7 to 14 days.
During this time, perform daily visual checks and top off food and water as necessary, but minimizing disruption of their behavior.
Change cages on the final day of the light/dark cycle and again once animals have been switched to a constant light environment.
2: Constant conditions
Dark
Reset the lighting timer to leave the animals in a state of constant darkness (at the end of a light cycle, do not interrupt it).
Record activity data from the animals for 10 to 14 days.
Perform daily checks. To minimize disruptions to activity, use infrared cameras or red-filtered light sources to visually inspect the mice.
Light
Reset the lighting timer to leave the animals in a state of constant light. Change cages.
Record activity data from the animals for 10 to 14 days.
Perform daily checks on the mice with minimum disruption to their routines.
This basic protocol allows for a simple assessment of the animals’ daily activity. With the use of specialized software, more complex evaluations can be performed. These include, for example, the daily patterns of running-wheel activity; the circadian period or tau (τ), a measure of an animal’s internal circadian clock; the duration the active and rest phases of the circadian rhythm; the amplitude, which is the difference between the peak and the mean value of a rhythm.
Modifications
A number of modifications have been introduced to the basic protocol when using activity wheels for circadian systems.
Exposing nocturnal animals to light pulses during the dark phase will result in acute suppression of activity, a phenomenon known as masking. The masking effect can be calculated by comparing the animals’ activity during each hour of the light pulse to the activity at the same time when there is no light pulse (LeGates & Altimus, 2011).
Exposing animals to cycles with a period differing from that of the usual 24-hour period (T-cycle, 11:11 or 13:13 hour light/dark cycles, for example) allows for the measurement of the animals’ entraining ability, i.e., the ability to align their circadian system to an external rhythm. Another possibility is to study the animals’ ability to re-entrain; this involves shifting the light cycle by several hours (advance 6 hours, for example), forcing the circadian clock to adjust to a new light cycle (LeGates & Altimus, 2011).
Other measurements can be performed by adjusting the light/dark patterns (see Jud et al 2005).
Sample Data
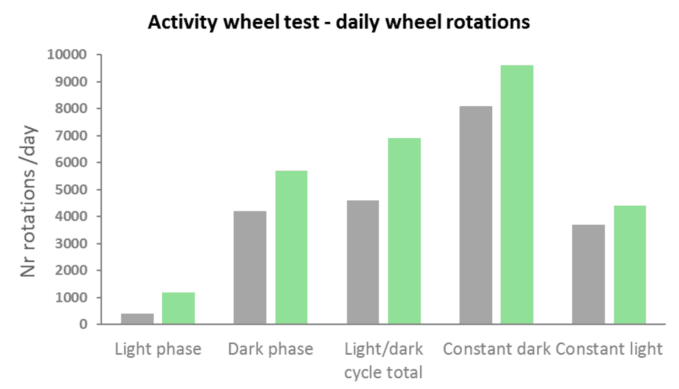
Wheel running activity can be presented by graphing the number of wheel rotations per day. This is the simplest measurement, providing an indication of the activity of an animal, and allowing to assess hypo or hyperactivity.
With specialized software, more complex evaluations can be performed. Circadian activity rhythms are frequently presented as a specialized graph known as actogram. Actograms provide a graphic illustration of the daily patterns of running-wheel activity, in which each horizontal line represents 1 day of activity, while vertical lines plotted within the horizontal lines show wheel-running activity within time bins (e.g. 5 min) of that day. The height of the vertical lines represents the level of activity. A number of additional measurements such as the circadian period, a measure of an animal’s internal circadian clock, or the duration of the active phase, for example, can also be made (see Jud et al 2005).
Strengths and Limitations
The main strength of the activity wheels for circadian rhythms is its simplicity – a simple apparatus connected to an analysis software allows for a reliable evaluation of the circadian system of rodents.
The major limitation is the obvious sensitivity to the lighting conditions. Any uncontrolled source of light can easily disrupt the circadian assessment performed with this test. This must be kept in mind when using the activity wheels for circadian rhythms.
Summary and Key Points
- Activity wheels can be used to assess the activity of the circadian system.
- This test assesses the animals’ activity level associated with circadian rhythms, their ability to entrain to changes in those rhythms, and a number of characteristics of the circadian system.
- This test can be adapted in order to collect data regarding different aspects of the circadian system.
- Animals in disease models or undergoing interventions that can affect their circadian system will show changes in their circadian activity patterns.
- Activity wheels can be used to study the effect of diseases and treatments on the circadian system, or for genetic screening of molecules that can modulate the circadian system, for example.
Request a quote
"*" indicates required fields