Operant conditioning, while a relatively simple task, involves a variety of brain regions, neurotransmitter systems, and receptors in the processes of acquisition, maintenance, and extinction. Thus, it is of little surprise that a variety of pharmacological manipulations have been shown to either enhance or diminish these aspects of operant conditioning behavior. Here, we will focus on several prime examples of drugs or pharmacological manipulations which enhance one or more of these aspects of this behavioral learning process.
Nicotine
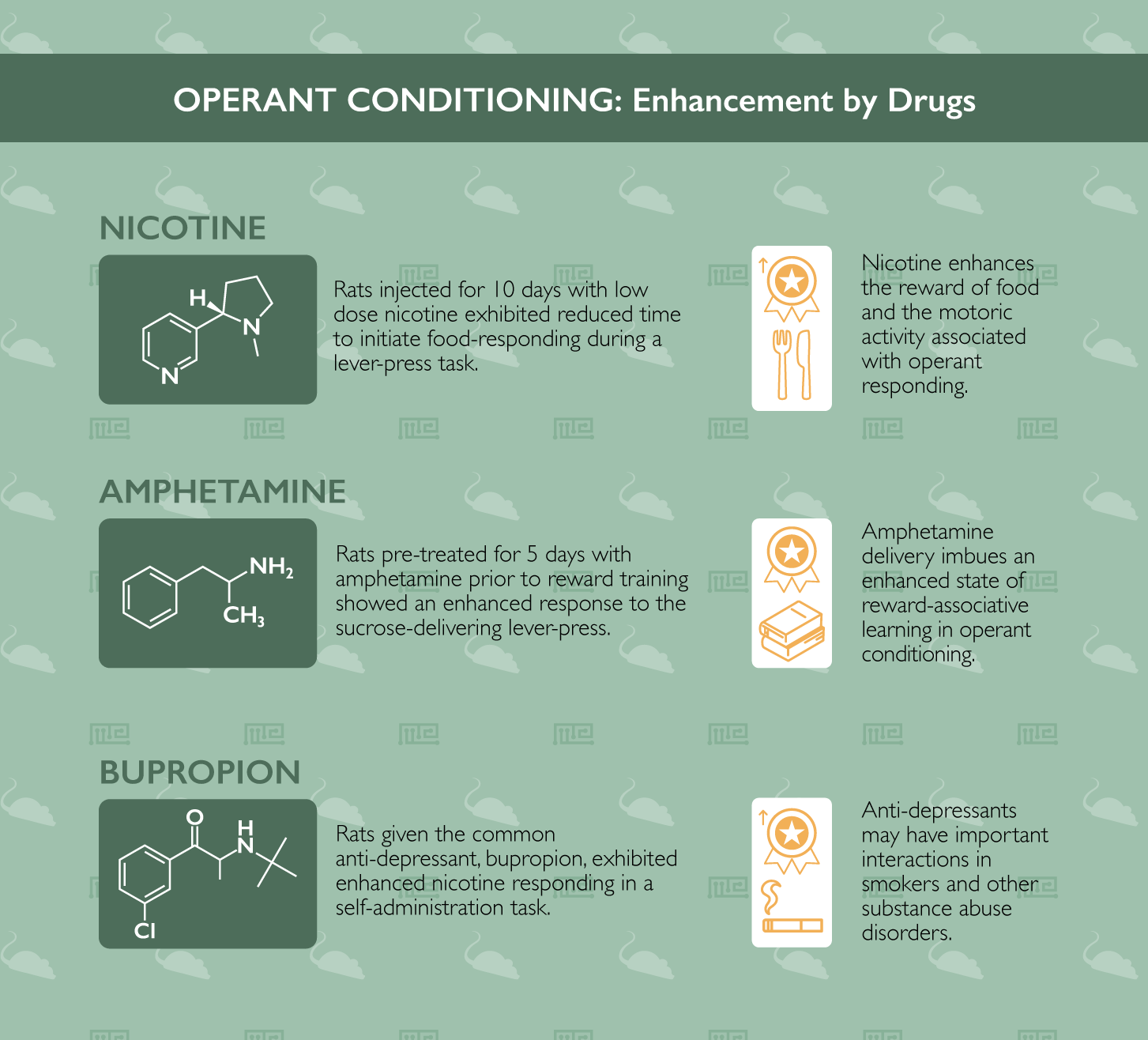
Nicotine has repeatedly been investigated as an enhancer of operant conditioning. For instance, in characterizing operant food-seeking behavior, researchers at Arizona State University in a 2018 publication found that nicotine administration enhances multiple aspects of this conditioning.[1] To study this, rats were injected with nicotine for 10 days (following an initial acute exposure). Next, when the rats performed a lever-press task to receive a food reward, the researchers noted that the low dose nicotine-exposed rats exhibited reduced time to initiate food-responding in each operant bout, as well as increased total responses following higher doses of pre-session nicotine exposure. They interpret these data to indicate that nicotine enhances the reward value of food and, at higher doses, the motoric activity associated with operant responding.
Similarly, in studying pavlovian conditioning protocol in water-deprived rats for the association between a light/tone conditioned stimulus and a water reward, researchers at the University of Toronto found that acquisition of the initial pairing between the conditioned stimulus (light/tone) and the reward was significantly enhanced by nicotine administration.[2] In fact, rats who were given nicotine during these pairing sessions showed stably higher levels of responding to the now-associated conditioned stimulus (known as a conditioned reinforcer) over multiple subsequent sessions, suggesting a stable and prolonged effect of nicotine on acquisition in simple rewarding behavioral associations.
As is often the case, sexually-divergent effects have been found in the effects of previous nicotine exposure on operant conditioning. For instance, in a 2014 study from Yale University in the United States, rats were pre-treated with daily nicotine during the adolescent period in order to model early-life smoking behavior (adolescence indeed being the period when most chronic smokers experience first exposure).[3][4] Then, at maturity, the rats were tested for a simple Pavlovian conditioning protocol wherein the conditioned stimulus (a small light) was paired with an appetitive unconditioned stimulus (water delivery after brief water deprivation). Interestingly, female rats who were exposed to nicotine during the adolescent period were found to exhibit decreased conditioned stimulus approach during a test session following 10 days of training, while male rats showed enhanced approach under the same conditions. Nicotine exposure during adolescence, however, enhanced the conditioned reinforcement association in both sexes.
Amphetamine
Amphetamine, being an enhancer of dopaminergic transmission which itself is a known mechanism underlying reward prediction and association behavior that underlies operant conditioning, has been thoroughly investigated with regards to its effects on operant conditioning acquisition, maintenance, and extinction.
In one such experiment, researchers from Utah State University published in a 2015 paper in Behavioral Pharmacology that amphetamine administration inhibited the extinction of operant conditioning behavior in a food-reward task.[5] Once rats had sufficiently acquired an operant conditioning behavior for the administration of a food reward, they were treated with either amphetamine or a drug which loosely works in an inverse manner (SCH23390) prior to extinction sessions (in which the conditioned response stimulus is present but the unconditioned stimulus, food, is no longer delivered). As they reported, animals who received a pre-session administration of amphetamine showed significantly diminished rates of extinction attributed to enhanced dopaminergic signaling, while animals pre-treated with SCH23390 showed the opposite effects.
Another example of amphetamine enhancing operant conditioning comes from a 1998 paper from the University of York in the United Kingdom.[6] In their experiment, rats underwent a 5-day pre-treatment with amphetamine, followed by a washout prior to beginning sucrose-reward training. Indeed, compared to their saline pre-treated control counterparts, rats which had been previously exposed to the repeated amphetamine exposures showed a significantly enhanced responding to the sucrose-delivering lever-pressing task in an operant- and Pavlovian-conditioning protocol (light or tone indicating the availability of a lever-press-contingent sucrose delivery). These results, like those presented above, indicate that the dopaminergic stimulation which results from amphetamine delivery imbues an enhanced state of reward-associative learning behavior in operant conditioning tasks in rats. This effect is true even when the amphetamine exposure occurs an entire week prior to the beginning of operant training in a Pavlovian conditioning protocol.
Other drugs
Countless other drugs, hormone modulators, and neurotransmitter level manipulations have been studied in relation to operant conditioning behavior, further underscoring the breadth of brain regions and neurological systems associated with this complex behavior. Such interesting manipulations as increasing levels of the milk-derived oligosaccharide 2’-Fucosyllactose which stimulates central nervous system function and enhances synaptic plasticity have yielded particularly interesting results. Indeed, dietary delivery of 2’-Fucosyllactose enhanced operant conditioning behavior via vagus nerve activation in adult rats.[7]
In a thorough battery of testing to examine FDA-approved drugs which may alter nicotine self-administration in rats previously trained in conditioned responding to the drug, researchers at the University of Toronto identified an important interaction between the common antidepressant, bupropion, and nicotine-motivated operant conditioning.[8] Rats given bupropion exhibited enhanced nicotine responding, highlighting the potential risk to patients prescribed the drug who are struggling with substance abuse disorder. Conversely, two other common drugs (Varenicline and Lorcaserin) were found to reduce nicotine responding in the operant conditioning task. Such a finding indicates potential promise for the use of these drugs as pharmacotherapeutic intervention strategies for nicotine addiction.
Not surprising, given the known rewarding nature of µ-opioid activation, researchers at the University of California, San Francisco, recently found that administration of a µ-opioid receptor agonist (DAMGO), significantly enhanced alcohol self-administration in an operant conditioning task.[9] Indeed, animals were pre-treated with DAMGO in a brain region previously characterized as a mediator of reward behavior, the nucleus accumbens. Following this administration, animals who received DAMGO (as compared to their saline-infused counterparts), showed enhanced numbers of operant responses for seeking alcohol self-administration, suggesting that µ-opioid receptor activation in the nucleus accumbens selectively enhances alcohol self-administration.
Conclusions
Clearly, operant conditioning is a behavior which is malleable to the effects of additional motivating and reinforcing drugs. Nicotine, amphetamines, and opioids are prime examples due to their known interactions with the brain’s reward circuitry. They are also important with regards to translational value since comorbidity of drug use and other compulsive behaviors with polysubstance abuse is a common factor in the diagnosis of drug abuse disorders,[10] such as those for which operant conditioning is a common animal model. Indeed, nicotine is amongst the most common additional drugs used by those suffering from substance abuse disorders and its interactions with compulsive behaviors modeled by operant conditioning is therefore of particular relevance.[11] Conversely, there exists an array of drugs which reduce operant responding behavior, indicating significant promise for therapeutic intervention in substance abuse disorder patients. This research is outlined in the article, Operant Conditioning: Inhibition by Drugs.
References
- Romero, K., Daniels, C. W., Gipson, C. D., & Sanabria, F. (2018). Suppressive and enhancing effects of nicotine on food-seeking behavior. Behavioural Brain Research, 339, 130–139.
- Guy, E. G., & Fletcher, P. J. (2013). Nicotine-induced enhancement of responding for conditioned reinforcement in rats: role of prior nicotine exposure and α4β2 nicotinic receptors. Psychopharmacology, 225(2), 429–440.
- Patton, G. C., Carlin, J. B., Coffey, C., Wolfe, R., Hibbert, M., & Bowes, G. (1998). Depression, anxiety, and smoking initiation: a prospective study over 3 years. American Journal of Public Health, 88(10), 1518–1522.
- Quick, S. L., Olausson, P., Addy, N. A., & Taylor, J. R. (2014). Repeated nicotine exposure during adolescence alters reward-related learning in male and female rats. Behavioural Brain Research, 261, 171–176.
- Quick, S. L., & Shahan, T. A. (2015). The impact of D-amphetamine and SCH23390 on behavioral momentum of food seeking and reinstatement in rats. Behavioural Pharmacology, 26(3), 249–259.
- Harmer, C. J., & Phillips, G. D. (1998). Enhanced appetitive conditioning following repeated pretreatment with d-amphetamine. Behavioural Pharmacology, 9(4), 299–308.
- Vazquez, E., Barranco, A., Ramirez, M., Gruart, A., Delgado-Garcia, J. M., Jimenez, M. L., … Rueda, R. (2016). Dietary 2’-Fucosyllactose Enhances Operant Conditioning and Long-Term Potentiation via Gut-Brain Communication through the Vagus Nerve in Rodents. PloS One, 11(11), e0166070.
- Guy, E. G., Fisher, D. C., Higgins, G. A., & Fletcher, P. J. (2014). Examination of the effects of varenicline, bupropion, lorcaserin, or naltrexone on responding for conditioned reinforcement in nicotine-exposed rats. Behavioural Pharmacology, 25(8), 775–783.
- Richard, J. M., & Fields, H. L. (2016). Mu-opioid receptor activation in the medial shell of nucleus accumbens promotes alcohol consumption, self-administration and cue-induced reinstatement. Neuropharmacology, 108, 14–23.
- Liu, Y., Williamson, V., Setlow, B., Cottler, L. B., & Knackstedt, L. A. (2018). The importance of considering polysubstance use: lessons from cocaine research. Drug and Alcohol Dependence, 192, 16–28.
- Kohut, S. J. (2017). Interactions between nicotine and drugs of abuse: a review of preclinical findings. The American Journal of Drug and Alcohol Abuse, 43(2), 155–170.
