Disabilities occupy a wide spectrum of types and form. Not being able to see, hear, walk, and talk are all heavy burdens to the affected and his or her surroundings. The mentioned disabilities have a root source, traceable to the problems origin. This makes treatment easier for the patient; it makes finding solutions more probable and more likely. What happens when a disability has no traceable origin? What if it has no effective treatment, yet is considered as disabling as the physical loses mentioned above? Obsessive-compulsive disorder (OCD) is one of many psychiatrically disabling conditions that obstruct normal daily life functioning[1]. Despite the severity, high prevalence, and overall costly nature of obsessive-compulsive disorder, effective treatment has yet to emerge that satisfies clinical necessities.
As a result, obsessive-compulsive disorder mouse models are the primary source of experiments that can help shed light onto the complex disorder within the human context. The rest of this article will comprehensively evaluate and investigate what obsessive-compulsive disorder is, its clinical features along with its symptoms, while also illustrating pre-clinical mouse models that have emerged as potential models and their treatment that can, with further study, be replicated in human settings.
Definition and Features
OCD is a mental disorder in which intrusive and unwanted thoughts (obsessions) trigger a repetitive pattern of behavior (compulsion) in the patient. Thus, the disorder is composed of two features; the obsession and the compulsion. The former manifests as repetitive, intrusive thoughts, while the latter consists of repetitive behaviors that obstruct daily life[2]. The obsessions lead to anxiety, and the compulsions are used to reduce the disabling state. Anxiety, being the link between the two features, establish a starting point for pre-clinical research and paves the way in finding a model.
Furthermore, prior to establishing an obsessive-compulsive disorder mouse model, research has established that several regions are affected by the disease. Using functional and structural neuroimaging methods, studies have collectively shown the following regions to be altered in patients with OCD:
- Anterior Cingulate Cortex (ACC)
- Orbitofrontal Cortex (OFC)
- Ventral striatum
These inter-related regions allow researchers the possibility to investigate animal models that can be used potentially as therapeutic treatments within the human context.
Model Types and Assessment of OCD in Mice
Before we talk about the various obsessive-compulsive disorder mouse models, it’s important that we understand how animal models work and how to set them up effectively.
Because we are talking about mimicking one species-related disease onto another one, and given the fact that we are talking about a psychiatric disorder, animal models must follow some certain criteria to make sure they mimic human-context as much as possible. Such criteria are:
- Symptoms in the animal model must be, to some extent, analogous to that of the modeled species, i.e. face validity.
- Treatment of disease induction reverses symptoms in animals, i.e. predictive validity.
- Neural or physiological systems underlying the disease involved in the animal model must be similar to that of the modeled species. i.e. construct validity.
- Animal models must be tested for reliability, i.e. results must be replicated across various laboratories.
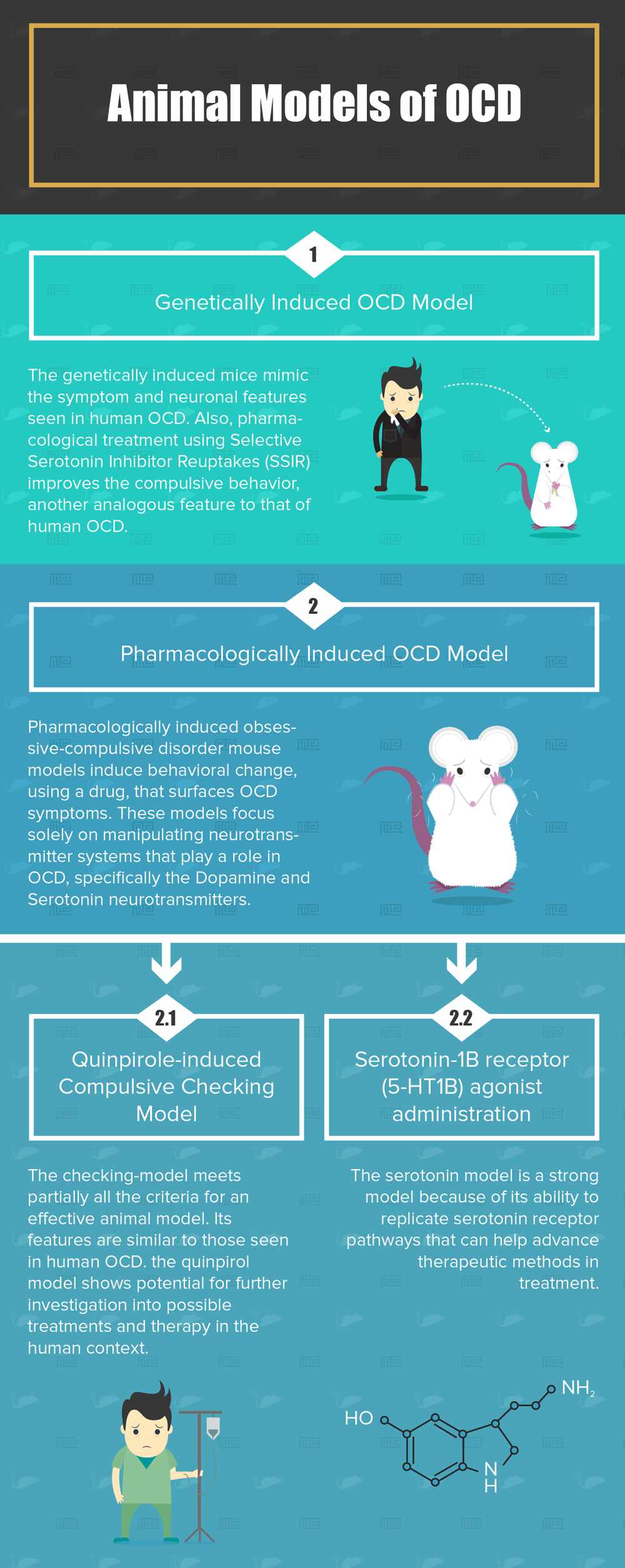
Genetically Induced OCD Model
Because no genetic mutation is evident in human OCD, mouse models in this section are constructed to satisfy the behavioral aspect of the disorder, that is, the compulsive-like behavior. Such results are induced by genetically manipulating a known gene. Here are the current strains belonging to genetic induction:
- Hoxb8lox mutant mice
- SAP90/PSD95 mice
- Slitrk5 KO mice
- Slc1A1 KO mice
When genetic induction occurs in these mouse models, several behavioral changes are witnessed that mimic human OCD[4]. Compulsive behaviors such as excessive self-grooming, digging, and running were all witnessed in these models. Furthermore, neuronal abnormalities that were reported in human OCD were also observed in these mouse models, specifically abnormalities in the OFC and striatum.
The significance of this model is that it sticks to the criteria mentioned above. The genetically induced mice mimic the symptom and neuronal features seen in human OCD, and pharmacological treatment using selective serotonin inhibitor reuptakes (SSIR) improve the compulsive behavior, another analogous feature to that of human OCD.
Pharmacologically Induced OCD Model
Contrary to the genetic models, pharmacologically induced obsessive-compulsive disorder mouse models induce behavioral change, using a drug, that surfaces OCD symptoms. These models focus solely on manipulating neurotransmitter systems that play a role in OCD, specifically the Dopamine and Serotonin neurotransmitters.
There are two main pharmacologically induced models.
- Quinpirole-induced Compulsive Checking Model targets Dopamine receptors
- Serotonin-1B receptor (5-HT1B) agonist administration is based on Serotonin receptors.
The significance of these models illustrates that there is a link between the fact that OCD is evident in multiple brain areas, and because of this conclusion, research has supplemented this by establishing a correlation between OCD and dopamine plus serotonin, in which the respective receptors are found throughout several brain regions.
Quinpirole-induced Compulsive Checking Model
This model begins by treating the experimental group of mice with chronic injection of D2/D3 dopamine agonist quinpirol. The observed altered behaviors in this model were as such[3]:
- Mice preferred certain locations to others.
- At these specific locations, the mice presented with specific, routine actions, repeatedly.
- Compulsive performances were witnessed at specific locations.
- Mice participate in operant response in the absence of reward.
The checking-model meets partially all the criteria for an effective animal model, and because the features above are similar to those seen in human OCD, the quinpirol model shows potential for further investigation into possible treatments and therapy in the human context.
Serotonin-1B receptor (5-HT1B) agonist administration
Experimental Balb/cJ mice, when treated with serotonin receptor agonist displays OCD like behaviors. During further investigation, researchers localized serotonin receptors within the OFC, a major neurological region that is altered in human OCD. Studies have established that the serotonin model is a strong model because of its ability to replicate serotonin receptor pathways that can help advance therapeutic methods in treatment.
Because of OCD’s behavioral nature, researchers are able to test and measure the magnitude of symptoms using behavioral assessments. Two main behavioral tests that were used to study symptoms throughout the various models above are the elevated plus maze and elevated zero maze.
These mazes are important for assessment because they enable researchers to find potential therapeutic treatments. SAP90/PSD95 mice, when genetically induced, portray OCD-like behaviors that are analogs in human OCD. One of these symptoms, anxiety, is a major contributor to the compulsion feature of the disorder. When testing the experimental mice for anxiety using the elevated plus maze, the results concluded that anxiety had developed in the genetically induced mice[1].
The significance of this finding, and others like it, offer potential new ways of treating the compulsion part of OCD in humans. When the experimental mice were treated with fluoxetine, the behavioral alterations were reversed, which is a sign that the same might be reproducible in human OCD.
The potentiality of animal models is clearly valid, given the above research. The ability to replicate repetitive behaviors in mice is a milestone closer to establishing a therapeutic method in the clinical setting.
Despite the importance of the models above, each and every one of the models is limited, either by not meeting the animal model criteria fully or by missing some sort of crucial aspect of human OCD. Despite that, with further investigation and research, pre-clinical studies can ensure progression into the ways OCD is treated.
Reference
- Ahmari, S. E. (2016). Using mice to model Obsessive Compulsive Disorder: From genes to circuits. Neuroscience,321, 121-137. doi:10.1016/j.neuroscience.2015.11.009
- Pittenger, C., Kelmendi, B., Bloch, M., Krystal, J. H., & Coric, V. (2005). Clinical Treatment of Obsessive Compulsive Disorder. Psychiatry (Edgmont), 2(11), 34–43.
- Alonso, P., López-Solà, C., Real, E., Segalàs, C., & Menchón, J. M. (2015). Animal models of obsessive–compulsive disorder: utility and limitations. Neuropsychiatric Disease and Treatment, 11, 1939–1955. http://doi.org/10.2147/NDT.S62785
- Wang, L., Simpson, H. B., & Dulawa, S. C. (2009). Assessing the validity of current mouse genetic models of obsessive–compulsive disorder. Behavioural Pharmacology, 20(2), 119–133. http://doi.org/10.1097/FBP.0b013e32832a80ad