Introduction to Bipolar Disorder
Bipolar disorder, originally termed manic-depressive disorder, is a devastating neuropsychiatric disease, which is characterized by alternating manic and major depressive episodes that may be recurrent over time. Bipolar disorder affects more than 1% of the global population and is a leading cause of disability.[1] Even though patients with bipolar disorder may achieve full remission or have symptom-free periods, it is the early onset of the disease combined with the unpredicted nature of recurrent episodes that reduces patient’s functionality, posing a heavy burden on the patient, their family, and society in total.
According to the latest edition of the Diagnostic and Statistical Manual of Mental Disorders of the American Psychiatric Association (DSM-V), bipolar disorder is neither a disease in the spectrum of psychosis, nor an affective disorder belonging to the spectrum of unipolar depression; instead it is an independent mental disorder that is further sub-classified based on the severity and duration of the manic and depressive episodes. An episode of mania is characterized by persistently elevated energy, euphoria, engagement into risky activities, decreased need for sleep and/or distraction of attention. A typical depressive episode consists of lack of pleasure and energy, excessive feeling of worthlessness, loss of interest for almost all activities and/or sleep disturbances. Patients with bipolar disorder suffer from mood instability, ranging from extreme euphoria to suicidal thoughts, which often results in severe problems during everyday life activities, such as social life and work.[2]
As with all neuropsychiatric disorders, the etiology of bipolar disorder is unknown. It is a multifactorial disease that arises from a combination of genetic and environmental factors, each contributing a small percent to the manifestation of the disease.[3] Gene wide association studies (GWAS) have revealed a number of associated loci, but the majority of identified polymorphisms are low-risk and low-penetrance, making it difficult to draw causative relationships.[4]
Current therapeutic approaches are untargeted and mainly consist of mood stabilizers, such as lithium and valproate, combined with antidepressant or antipsychotic drugs. Unfortunately, these drugs only have an effect on the manic episodes, while leaving the depressive symptoms largely untreated, and may even exacerbate the mood cycling in a number of patients.[5] Therefore, the use of animal models is necessary, in order to develop new therapeutic approaches and delineate the complex pathophysiological mechanisms of bipolar disorder.
Research Techniques for Studying Bipolar Disorder
In vivo Studies Using Animal Models
The use of animal models is vital to expanding our understanding of all aspects of human biology. Although the process of modeling has inherent disadvantages, as it may lead to false conclusions and inaccurate predictions, the ethical limitations of human studies render the use of animals inevitable. We need to be able to test our hypotheses, rather than simply observe biological facts, and animals represent an indispensable tool in this process. Rodents have a leading role in the field of neuropsychiatric disease modeling, as they offer a unique combination of characteristics, such as conserved molecular mechanisms with humans, relatively rapid life-cycle, advanced genetics and a plethora of well-characterized behaviors. In recent years additional models have been used to study bipolar disorder, including the fruit fly Drosophila melanogaster and the zebrafish Danio rerio.[6][7][8][9] However, in the scope of this article, we will focus on mice models of bipolar disease, as they are used in the vast majority of scientific publications studying bipolar disorder.
Using rodents to model neuropsychiatric diseases has proven to be particularly difficult, due to our incomplete understanding of the underlying pathological mechanisms, as well as the inherent mismatch of human and animal behavior. There is no specific gene to knock out or specific environmental trigger to induce the disease in mice. Moreover, animals cannot express the feeling of worthlessness or suicidal thoughts. Thus, it is impossible to reproduce the full aspect of the disease in one animal model. Instead, efforts have focused on modeling specific aspects of the disease, to create rodent models that partially recapitulate features of bipolar disorder. Even though these models do not strictly meet the standard criteria of face, construct and predictive validity, in fact, they have provided critical insight in our understanding of the pathogenic mechanisms and have enriched our arsenal of therapeutic strategies. The most challenging part in modeling of bipolar disorder is the recurrent and spontaneous nature of manic and depressive symptoms.[10] Until today there are no animal models that exhibit spontaneous alternation of behavior. Thus, separate models of mania and depression are being used. As we have already analyzed the rodent models for depression and depression behavioral tests, in this article we will further discuss the models of mania, as well as models that recapitulate other aspects of the disease.
Other Studies
Taking into account the complexity of bipolar disorder, many researchers opt for working directly with patients to unravel the neurobiological substrates of the disease. Widely used approaches involve structural and functional brain connectivity and activity data, using diffusion tensor imaging tractography, fMRI and MEG (magnetoencephalography).[11][12] These studies have identified altered function and connectivity in midbrain structures, such as the amygdala-anterior paralimbic system, as well as the prefrontal cortex. Moreover, several large scales, unbiased experimental approaches, such as transcriptomics, lipidomics, and metabolomics, are currently used to develop biomarkers for the diagnosis of bipolar disorder and the prediction of drug responsiveness.[13][14][15][16]
On the other end of the spectrum, a wealth of studies has employed in vitro systems to study bipolar disorder. As cell cultures represent a simpler model, they allow for strictly controlled experiments, as well as targeted innervations and direct readouts. Cellular models have mainly focused on assessing the involvement of various signaling pathways, apoptosis, ER stress and mitochondria homeostasis in the pathogenic or therapeutic effects of genetic and pharmacological innervations. In vitro systems include both non-neuronal systems such as lymphoblastoid cell lines (LCLs) and neuronal cells derived from patients, such as olfactory neuronal epithelium (ONE) cells, and neurons differentiated from induced pluripotent stem cells (iPSCs).
Drug-induced Models of Mania
Psychostimulant Models
For many years, the gold-standard method to mimic human mania in rodents was by administration of psychostimulant drugs, namely amphetamine. This paradigm is still widely used, with locomotor activity being the readout test, using the open field apparatus.[17][18] However, in recent years criticism has highlighted the disparities of human mania and mouse hyperactivity, as on the one hand mania is an ensemble of symptoms that extend beyond increased activity and on the other hand mania is chronic while drug-induced hyperactivity models are acute, implying the involvement of distinct neurobiological mechanisms.[19]
In an attempt to optimize the amphetamine model, researchers have developed a model with combined administration of amphetamine and anxiolytic drugs, such as benzodiazepines, as well as models of amphetamine sensitization. Both models have some predictive validity, as lithium and valproate have been reported to reduce excessive locomotor activity, but other drugs that showed promising results in preclinical studies proved to have limited efficacy in bipolar disorder patients. Additionally, it is important to notice that models with co-administration of drugs require a large number of control experimental conditions, that may render the study complicated and increase confounding variables.
Ouabain Model
A different approach to model human mania entails the administration of ouabain, an adenosine triphosphate inhibitor. Intracerebroventricular administration of ouabain has a long-lasting effect that is correlated to increased oxidative stress and decreased BDNF levels in the brain, observation that are in accordance with data from bipolar disorder patients. Thus, the face validity of the model is better, compared to the amphetamine-induced hyperactivity. Nonetheless, the main output of the model is locomotor hyperactivity, leading to the same limitations as the psychostimulant models.[20]
Genetic Models of Mania
Based on findings from human genetic studies, researchers have developed a number of genetic models of bipolar disorder that recapitulate specific features of the disease, in order to investigate the neural correlates of the observed behaviors and explore the mechanism of action and efficacy of existing and novel drugs. The most widely used genetic models of bipolar disorder include the circadian rhythm regulator ClockΔ19 (dominant negative) and Clock knockout mice, the GSK-3b overexpressing mouse, the dopamine transporter DAT- Knockdown mouse, and the Ankyrin deficient mouse.[21] Even though these genes have been associated with bipolar disorders, the wide spectrum of their functions raises questions about their specificity to model bipolar disease. Still, years of research employing these models have provided fruitful findings on bipolar disorder and expanded our understanding.
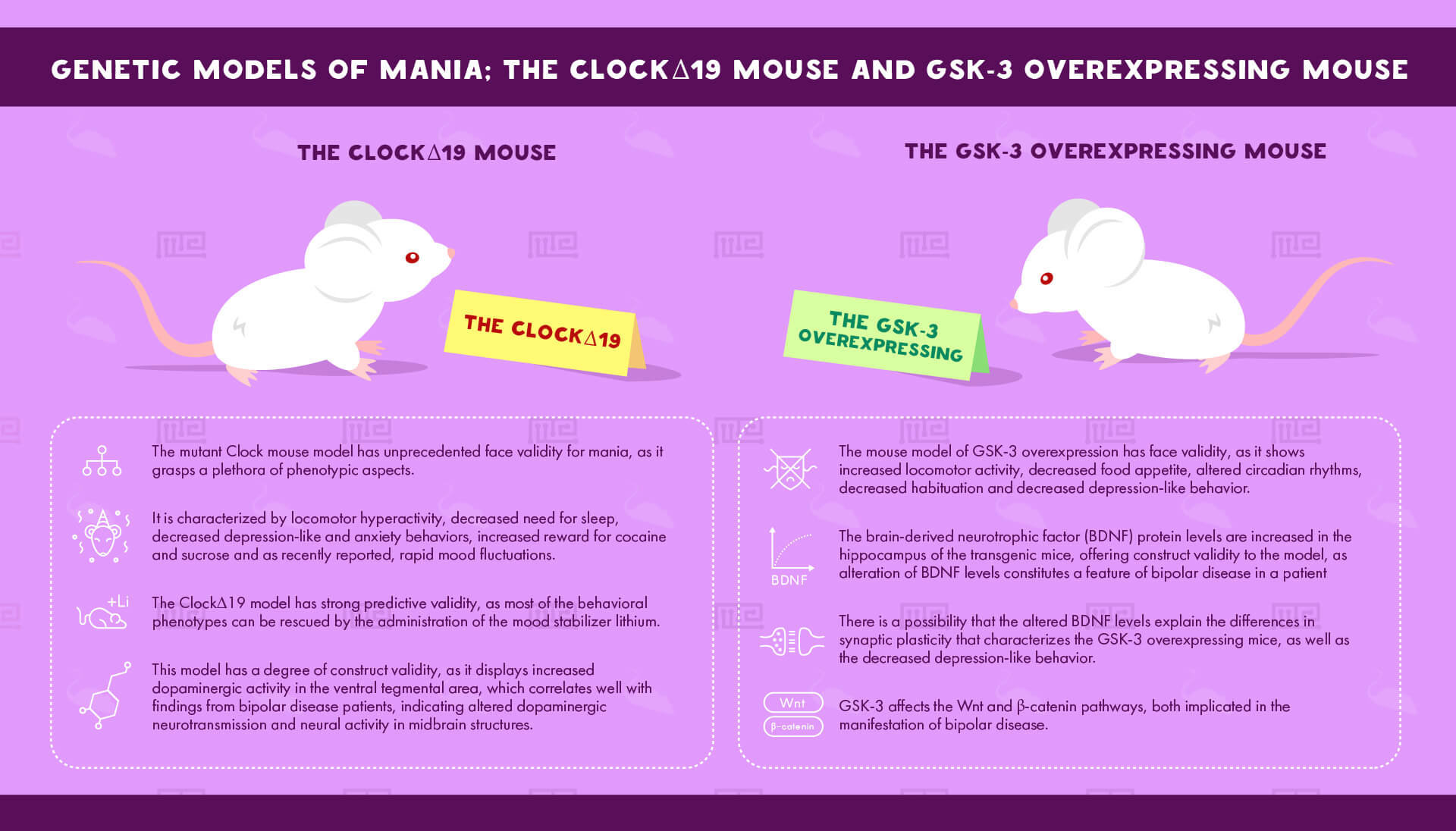
The ClockΔ19 Mouse
Sleep deprivation and alteration of circadian rhythms are typical symptoms of manic episodes of bipolar disease patients. The clock is a central transcriptional activator of molecular rhythms and a critical component of the internal circadian regulatory mechanism. The mutant Clock mouse model has unprecedented face validity for mania, as it grasps a plethora of phenotypic aspects. It is characterized by locomotor hyperactivity, decreased need for sleep, decreased depression-like and anxiety behaviors, increased reward for cocaine and sucrose and as recently reported rapid mood fluctuations. Moreover, the ClockΔ19 model has strong predictive validity, as most of the behavioral phenotypes can be rescued by the administration of the mood stabilizer lithium. In addition, this model has a degree of construct validity, as it displays increased dopaminergic activity in the ventral tegmental area, which correlates well with findings from bipolar disease patients indicating altered dopaminergic neurotransmission and neural activity in midbrain structures. Moreover, Clock polymorphisms have been indicated to participate in bipolar disorder pathogenesis by human genetic studies.[22][23]
The GSK-3 Overexpressing Mouse
Interest on the glycogen synthase kinase 3beta (GSK-3beta) GSK-3 piqued when the mood stabilizer lithium was found to inhibit GSK-3, as a part of its molecular way of action. The mouse model of GSK-3 overexpression has face validity, as it shows increased locomotor activity, decreased food appetite, altered circadian rhythms, decreased habituation and decreased depression-like behavior. Moreover, the brain-derived neurotrophic factor (BDNF) protein levels are increased in the hippocampus of the transgenic mice, offering construct validity to the model, as alteration of BDNF levels constitutes a feature of bipolar disease in patients. It is possible that the altered BDNF levels explain the differences in synaptic plasticity that characterizes the GSK-3 overexpressing mice, as well as the decreased depression-like behavior. Also, GSK-3 affects the Wnt and β-catenin pathways, both implicated in the manifestation of bipolar disease.
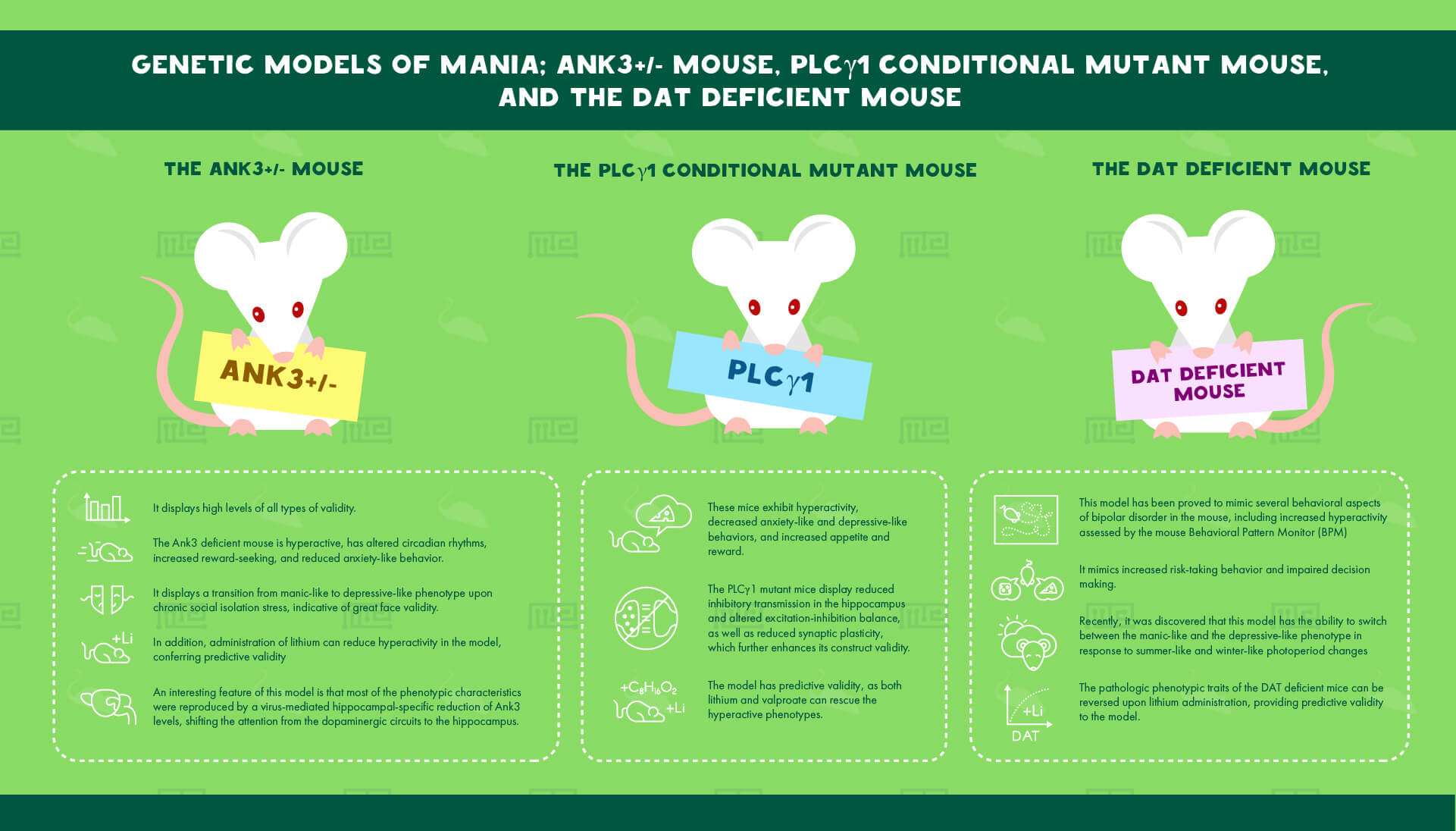
The Ank3+/- Mouse
Ankyrin G is a scaffold protein, encoded by Ankyrin 3 (ANK3), and has been identified by genetic studies as an important risk gene for bipolar disorder. Researchers have created a conditional brain-specific Ank3 heterozygous mouse, that displays high levels of all types of validity. In terms of behavioral assessment, the Ank3 deficient mouse is hyperactive, has altered circadian rhythms, increased reward-seeking, reduced anxiety-like behavior and most importantly displays a transition from manic-like to depressive-like phenotype upon chronic social isolation stress, indicative of great face validity. In addition, administration of lithium can reduce hyperactivity in the model, conferring predictive validity. A really interesting feature of this model is that most of the phenotypic characteristics were reproduced by a virus-mediated hippocampal-specific reduction of Ank3 levels, shifting the attention from the dopaminergic circuits to the hippocampus. Furthermore, the model has predictive validity, as lithium administration was able to rescue the increased-hyperactivity and reduced-anxiety behaviors.[25]
The PLCγ1 Conditional Mutant Mouse
Genetic studies have highlighted the implication of altered phospholipase C (PLC) signaling in bipolar disorder and numerous regulators of PLC such as diacylglycerol (DAG) and PLC itself, have emerged as risk-related genes and loci. Recently, conditional mutant mice of PLCγ1 was constructed, bearing a deletion of PLCγ1 gene in the excitatory neurons in the forebrain, instructed by the CAMKII promoter. These mice exhibit hyperactivity, decreased anxiety-like and depressive-like behaviors, and increased appetite and reward. The PLCγ1 mutant mice display reduced inhibitory transmission in the hippocampus and altered excitation-inhibition balance, as well as reduced synaptic plasticity, that may be due to altered BDNF- TrkB levels, further enhancing its construct validity. The model has also predictive validity, as both lithium and valproate can rescue the hyperactive phenotypes.[26]
The DAT Deficient Mouse
Based on genetic, molecular and imaging data indicating lower dopamine transporter (DAT) levels in the pathophysiology of bipolar disorder, researchers have developed a DAT knockdown mouse model to study bipolar disorder. This model has been proved to mimic several behavioral aspects of bipolar disorder, such as increased hyperactivity assessed by the mouse Behavioral Pattern Monitor (BPM), impaired decision making, increased risk-taking behavior and most recently the ability to switch between the manic-like and the depressive-like phenotype, in response to summer-like and winter-like photoperiod changes. This finding resembles the seasonally-induced switching between mania and depression in bipolar disease patients, with mania associated with summer and depression with winter and further increases the face validity of the model. Finally, the pathologic phenotypic traits of the DAT deficient mice can be reversed upon lithium administration, providing predictive validity to the model.[27][28]
The genetic models of bipolar disorders are characterized mainly by three downsides. First of all, because bipolar disorder is not a single-gene disease, targeting a single protein in a genetic model is not enough to recapitulate the whole spectrum of the disease and the pathogenic mechanisms. Moreover, disrupting the physiological function of these proteins additionally affect other pathways and physiological mechanisms, that are not related to bipolar disorder and are normally functioning in patients. Finally, several genetic models of bipolar disorder, such as the GSK-3 overexpressing and the DAT knockout mice, are additionally used to model other neuropsychiatric diseases, e.g. schizophrenia. This is happening because it is still unclear to what extent the neuropsychiatric disorders overlap, so common mechanisms may serve several diseases, thus genetic models that have these mechanisms disrupted are used to model all of them. The arising problem is the lack of specificity of potentially discovered mechanisms or drugs with respect to bipolar disorder.
Models of Bipolar Disease Endophenotypes
A recently developed strategy to study complex human diseases is to focus on specific endophenotypes, which constitute quantifiable components of neurological function, distinct from psychiatric symptoms. The nature of endophenotypes can be neurophysiological, biochemical, anatomical or behavioral. Models of endophenotypes are not restricted to a specific disease and do not focus on specific symptoms; instead, they represent shared pathological features that can be successfully modeled in animals. For bipolar disorder, the relevant endophenotypes include but are not limited to:
- circadian rhythm instability
- alterations of brain anatomy and connectivity (including the dopamine reward pathway and the limbic-hypothalamo-pituitary-adrenocortical pathway)
- maladaptive neuroplasticity (BDNF polymorphisms are associated with bipolar disorder, and mood stabilizers such as lithium and valproate have neuroprotective action)
- mitochondrial dysfunction (a mouse model of mutant mitochondrial polymerase γ (POLG) recapitulates the periodic activity changes and is sensitive to lithium)
Environmental Models of Mania
Even though bipolar disorder has strong genetic correlates, the effect of environmental risk factors is of equal importance.[29] Thus, some mouse models of bipolar disorder focus on recapitulating the manic-like phenotype by behavioral interventions, rather than genetic manipulations.
Sleep Deprivation Paradigm
As mentioned above, circadian rhythm plays a critical role in bipolar disorder. Apart from the genetic models of circadian cycle disruption, there are additional methods to manipulate mouse sleep behavior. There are two types of long-term sleep deprivation models, the intra-cage sleep deprivation and the walking sleep deprivation types of cages that are used to induce sleep deprivation. Another, short-term approach to enforce sleep deprivation consists of placing a mouse on a small platform into a water tank, resulting in the animal staying awake to avoid falling into the water. However, a limitation of the water model is that it includes additional stressors, such as social isolation, immobility and immersion into water. All models of long term sleep deprivation (2-3 days) have great face validity, as they can capture numerous facets of mania, such as agitation, insomnia, increased sexuality and hyperactivity. They also offer good predictive validity, as the observed behaviors are sensitive to mood stabilizers, namely lithium and valproate.[30]
Resident-intruder Paradigm
Manic episodes are commonly associated with increased aggressive behavior and irritability. These aspects of human behavior can be modeled in mice, using the resident-intruder test. Typically this test has been used in the context of depression, to study the social defeat behavior displayed by the intruder. However, several studies have used the model to monitor resident aggression and found it to be reduced when the resident mice were pretreated with lithium.[31]
Another behavioral paradigm on the same track is the dominant-submissive behavior. The tube dominance test reveals the establishment of a hierarchy between mice. Studies have used this test to assess the effect of lithium on dominant behavior and found that it is decreased, following a similar temporal course to human bipolar disease patients. However, the predictive validity of this test is questionable, as anxiolytic and anxiogenic drugs that are of no use in bipolar treatment, also affect the observed behaviors.[32]
Mouse Strains for Bipolar Disease Modeling
An alternative approach to capture the complexity of neuropsychiatric diseases is to use specific mouse strains that display innate disease-like features. This approach is advantageous compared to transgenic mouse models, as single gene models of bipolar disorder have poor construct validity and enable partial understanding of the disease. On the contrary, the use of specific mouse stains is inherently characterized by face validity and also may provide insights into underlying pathogenic and pathophysiological mechanisms, displaying construct validity. Two mouse strains are mainly used as bipolar disorder mania models, the Black Swiss mice, and the Madison mice. Both strains display a manic phenotype consisting of increased locomotor activity, decreased sleeping, increased aggression and increased sexual activity. Furthermore, both models are sensitive to lithium treatment.[33][34]
Concluding remarks
Current pharmacotherapies for bipolar disorders were initially developed for schizophrenia or epilepsy, a fact that is indicative of our lack of understanding of bipolar disorder-specific mechanisms. However, during the past decade, the development of new technologies has played a fundamental role in expanding our understanding of bipolar disorder mechanisms. Genome-wide association studies and large-scale unbiased approaches, mainly genomics and proteomics, have provided numerous candidate risk factors, many of which have served as starting points for the creation and characterization of novel genetic animal models. The holy-grail of bipolar disease modeling, the spontaneous alternation of manic and depressive-like phenotypes, is gradually being achieved. In addition, new behavioral paradigms have been developed, that is capable of assessing more complex behaviors in greater detail with higher specificity, reducing the gap between human and animal behavior and increasing face and predictive validity of animal models. New hypotheses are arising and new therapeutic approaches are being tested, bringing the field of bipolar disorder research to its highest potential.[35]
References
- Merikangas, K. R., He, J. P., Burstein, M., Swendsen, J., Avenevoli, S., Case, B., … & Olfson, M. (2011). Service utilization for lifetime mental disorders in US adolescents: results of the National Comorbidity Survey–Adolescent Supplement (NCS-A). Journal of the American Academy of Child & Adolescent Psychiatry, 50(1), 32-45.
- American Psychiatric Association. (2000). Diagnostic and statistical manual of mental disorders, 10th revision. Washington, DC, US: American Psychiatric Association.
- Shinozaki, G., & Potash, J. B. (2014). New developments in the genetics of bipolar disorder. Current psychiatry reports, 16(11), 493.
- Ikeda, M., Saito, T., Kondo, K., & Iwata, N. (2018). Genome‐wide association studies of bipolar disorder: A systematic review of recent findings and their clinical implications. Psychiatry and clinical neurosciences, 72(2), 52-63.
- Vieta, E., Berk, M., Schulze, T. G., Carvalho, A. F., Suppes, T., Calabrese, J. R., … & Grande, I. (2018). Bipolar disorders. Nature Reviews Disease Primers, 4, 18008.
- Sagduyu, K., Dokucu, M. E., Eddy, B. A., Craigen, G., Baldassano, C. F., & Yıldız, A. (2005). Omega-3 fatty acids decreased irritability of patients with bipolar disorder in an add-on, open label study. Nutrition Journal, 4(1), 6.
- Herteleer, L., Zwarts, L., Hens, K., Forero, D., Del-Favero, J., & Callaerts, P. (2016). Mood stabilizing drugs regulate transcription of immune, neuronal and metabolic pathway genes in Drosophila. Psychopharmacology, 233(9), 1751-1762.
- Xiao, B., Cui, L. Q., Ding, C., & Wang, H. (2017). Effects of lithium and 2, 4-dichlorophenol on zebrafish: circadian rhythm disorder and molecular effects. Zebrafish, 14(3), 209-215.
- Ellis, L. D., & Soanes, K. H. (2012). A larval zebrafish model of bipolar disorder as a screening platform for neuro-therapeutics. Behavioural brain research, 233(2), 450-457.
- Logan, R. W., & McClung, C. A. (2016). Animal models of bipolar mania: the past, present and future. Neuroscience, 321, 163-188.
- Blond, B. N., Fredericks, C. A., & Blumberg, H. P. (2012). Functional neuroanatomy of bipolar disorder: structure, function, and connectivity in an amygdala–anterior paralimbic neural system. Bipolar disorders, 14(4), 340-355.
- Savitz, J. B., Price, J. L., & Drevets, W. C. (2014). Neuropathological and neuromorphometric abnormalities in bipolar disorder: view from the medial prefrontal cortical network. Neuroscience & Biobehavioral Reviews, 42, 132-147.
- Fries, G. R., Carvalho, A. F., & Quevedo, J. (2018). The miRNome of bipolar disorder. Journal of Affective Disorders, 233, 110-116.
- Ribeiro, H. C., Klassen, A., Pedrini, M., Carvalho, M. S., Rizzo, L. B., Noto, M. N., … & Hayashi, M. A. (2017). A preliminary study of bipolar disorder type I by mass spectrometry-based serum lipidomics. Psychiatry research, 258, 268-273.
- Yoshimi, N., Futamura, T., Bergen, S. E., Iwayama, Y., Ishima, T., Sellgren, C., … & Ohgi, Y. (2016). Cerebrospinal fluid metabolomics identifies a key role of isocitrate dehydrogenase in bipolar disorder: evidence in support of mitochondrial dysfunction hypothesis. Molecular psychiatry, 21(11), 1504.
- Sethi, S., & Brietzke, E. (2016). Omics-based biomarkers: application of metabolomics in neuropsychiatric disorders. International Journal of Neuropsychopharmacology, 19(3).
- Einat, H. (2014). New ways of modeling bipolar disorder. Harvard review of psychiatry, 22(6), 348-352
- Beyer, D. K., & Freund, N. (2017). Animal models for bipolar disorder: from bedside to the cage. International journal of bipolar disorders, 5(1), 35.
- Machado-Vieira, R., Soeiro-De-Souza, M. G., Richards, E. M., Teixeira, A. L., & Zarate Jr, C. A. (2014). Multiple levels of impaired neural plasticity and cellular resilience in bipolar disorder: developing treatments using an integrated translational approach. The World Journal of Biological Psychiatry, 15(2), 84-95
- Jornada, L. K., Moretti, M., Valvassori, S. S., Ferreira, C. L., Padilha, P. T., Arent, C. O., … & Quevedo, J. (2010). Effects of mood stabilizers on hippocampus and amygdala BDNF levels in an animal model of mania induced by ouabain. Journal of psychiatric research, 44(8), 506-510.
- Machado-Vieira, R., Andreazza, A. C., Viale, C. I., Zanatto, V., Cereser Jr, V., da Silva Vargas, R., … & Gentil, V. (2007). Oxidative stress parameters in unmedicated and treated bipolar subjects during initial manic episode: a possible role for lithium antioxidant effects. Neuroscience letters, 421(1), 33-36.
- Gould, T. D., & Einat, H. (2007). Animal models of bipolar disorder and mood stabilizer efficacy: a critical need for improvement. Neuroscience & Biobehavioral Reviews, 31(6), 825-831.
- (Yang et al 2017 Mol Psychiatry)
- Prickaerts, J., Moechars, D., Cryns, K., Lenaerts, I., van Craenendonck, H., Goris, I., … & Steckler, T. (2006). Transgenic mice overexpressing glycogen synthase kinase 3β: a putative model of hyperactivity and mania. Journal of Neuroscience, 26(35), 9022-9029.
- Leussis, M. P., Berry-Scott, E. M., Saito, M., Jhuang, H., de Haan, G., Alkan, O., … & Root, D. E. (2013). The ANK3 bipolar disorder gene regulates psychiatric-related behaviors that are modulated by lithium and stress. Biological psychiatry, 73(7), 683-690.
- Yang, Y. R., Jung, J. H., Kim, S. J., Hamada, K., Suzuki, A., Kim, H. J., … & Kim, E. K. (2017). Forebrain-specific ablation of phospholipase Cγ1 causes manic-like behavior. Molecular psychiatry, 22(10), 1473.
- Ralph-Williams, R. J., Paulus, M. P., Zhuang, X., Hen, R., & Geyer, M. A. (2003). Valproate attenuates hyperactive and perseverative behaviors in mutant mice with a dysregulated dopamine system. Biological psychiatry, 53(4), 352-359.
- Young, J. W., Cope, Z. A., Romoli, B., Schrurs, E., Joosen, A., Enkhuizen, J., … & Dulcis, D. (2018). Mice with reduced DAT levels recreate seasonal-induced switching between states in bipolar disorder. Neuropsychopharmacology, 1.
- Bortolato, B., Köhler, C. A., Evangelou, E., León‐Caballero, J., Solmi, M., Stubbs, B., … & Vieta, E. (2017). Systematic assessment of environmental risk factors for bipolar disorder: an umbrella review of systematic reviews and meta‐analyses. Bipolar disorders, 19(2), 84-96.
- Gessa, G. L., Pani L., Serra G. & Fratta W.(1995). Animal models of mania. Adv Biochem Psychopharmacol. 49:43-66.
- O’donnell, K. C., & Gould, T. D. (2007). The behavioral actions of lithium in rodent models: leads to develop novel therapeutics. Neuroscience & Biobehavioral Reviews, 31(6), 932-962.
- Malatynska, E., Pinhasov, A., Creighton, C. J., Crooke, J. J., Reitz, A. B., Brenneman, D. E., & Lubomirski, M. S. (2007). Assessing activity onset time and efficacy for clinically effective antidepressant and antimanic drugs in animal models based on dominant–submissive relationships. Neuroscience & Biobehavioral Reviews, 31(6), 904-919.
- Scotti, M. A. L., Lee, G., Stevenson, S. A., Ostromecki, A. M., Wied, T. J., Kula, D. J., … & Gammie, S. C. (2011). Behavioral and pharmacological assessment of a potential new mouse model for mania. Physiology & behavior, 103(3-4), 376-383.
- Flaisher-Grinberg, S., & Einat, H. (2010). Strain-specific battery of tests for domains of mania: effects of valproate, lithium and imipramine. Frontiers in psychiatry, 1, 10.
- Sigitova, E., Fišar, Z., Hroudová, J., Cikánková, T., & Raboch, J. (2017). Biological hypotheses and biomarkers of bipolar disorder. Psychiatry and clinical neurosciences, 71(2), 77-103.