Overview
KK.Cg-Ay/J, also known as KKAy mice, is a strain of mouse model whose metabolic abnormalities make it useful for diabetes and obesity research.[1]
History
The original KK strain, from which KKAy is derived, was purchased from a Japanese dealer in 1944. The Ay mutation was then introduced by Nishimura by crossing with Ay/a mice in the late 1950s. Kondo established multiple lines of KK mice, of which KKAy is one.[2]
The KKAy mice had been inbred for 100 generations before they were acquired by Gerritsen from Takeda Chemical Industries in 1983. The precise descent of the current Jackson Laboratory stock of KKAy from these other forms of KK mice remains uncertain.[1]
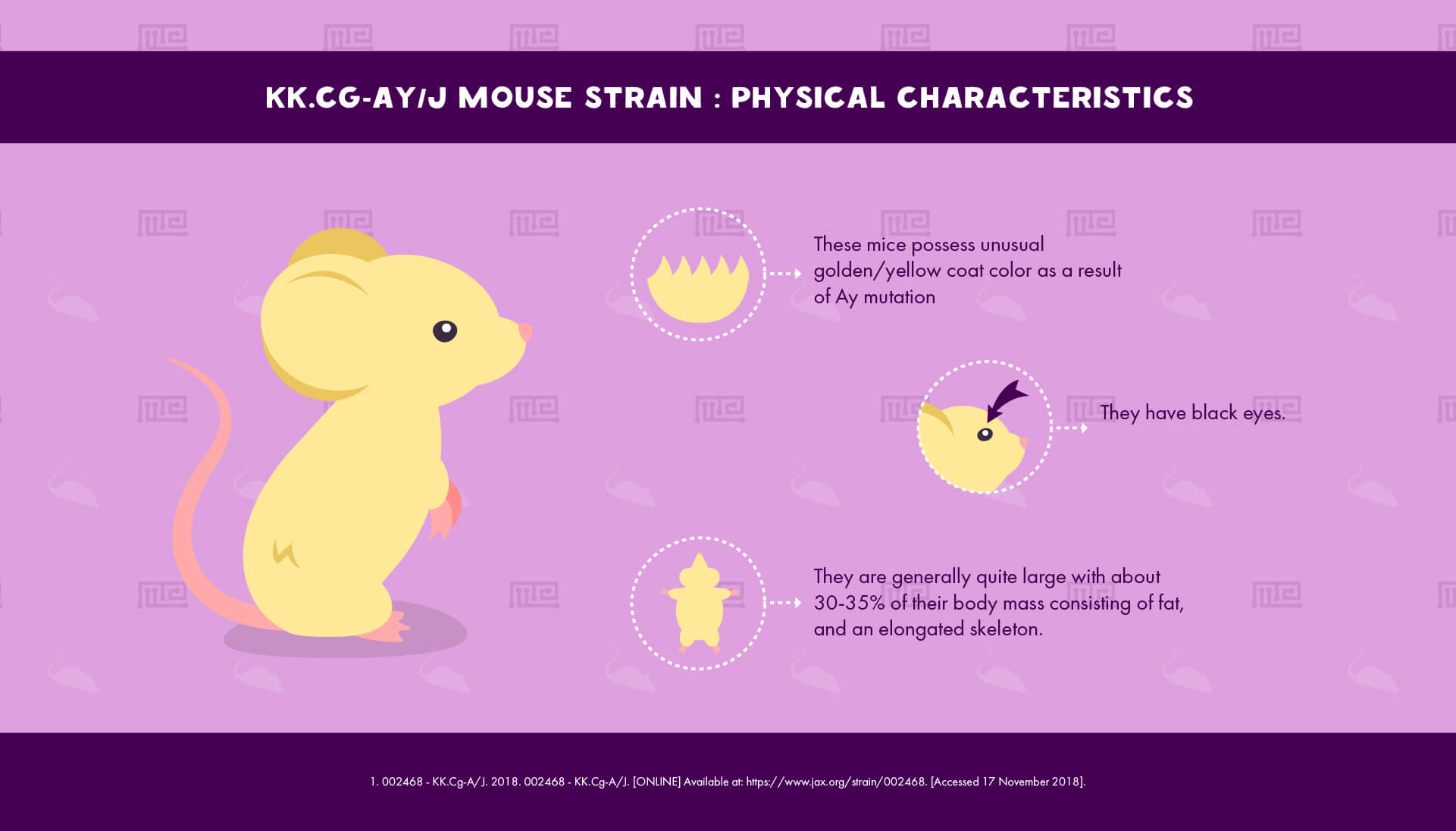
Physical Characteristics of the KKAy Mice
The Ay mutation confers on these mice an unusual golden/yellow coat color as well as black eyes. They are generally quite large, as a result of their obese phenotype (see below). Around 30-35% of their body mass consists of fat, and their skeleton is elongated.[1]
Behavioral Characteristics & Handling
KKAy mice show significantly worse performance in the Y maze test than C57BL/6J[3], and also in the Morris water maze compared to wild-type KK mice.[4] These results suggest that the KKAy strain exhibits deficits in spatial learning and memory. Further, KKAy mice show less avoidance than KK mice in the shuttle avoidance task.[4]
No advice on the handling of this strain could be found in the literature. Researchers are thus advised to exercise caution and may want to choose another strain if a high degree of docility is needed.
Health Characteristics
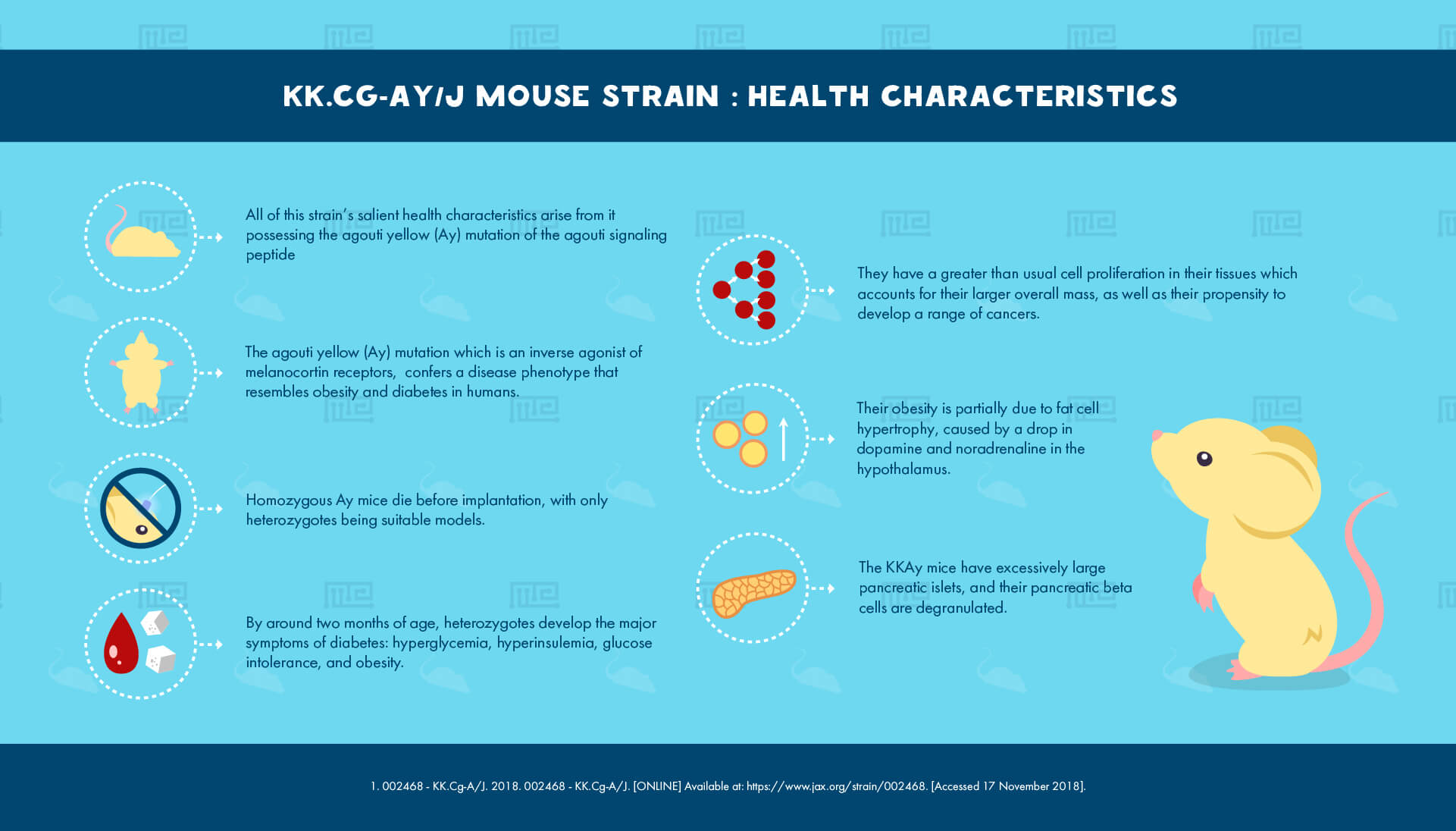
All of this strain’s salient health characteristics arise from it possessing the agouti yellow (Ay) mutation of the agouti signaling peptide, an inverse agonist of melanocortin receptors. The mutation confers a disease phenotype that resembles obesity and diabetes in humans.
Homozygous Ay mice die before implantation, with only heterozygotes being suitable models. By around two months of age, these heterozygotes develop the major symptoms of diabetes: hyperglycemia, hyperinsulemia, and glucose intolerance, as well as becoming obese.
The major cause of this phenotype is resistance to insulin. The KKAy mice have excessively large pancreatic islets, and their pancreatic beta cells are degranulated. These abnormalities are believed to result from the agouti protein being expressed in incorrect locations.
Their obesity is partially due to fat cell hypertrophy, caused by a drop in dopamine and noradrenaline in the hypothalamus. Greater than usual cell proliferation is seen in the tissues of this strain, which helps to account for their larger overall mass, as well as their propensity to develop a range of cancers.[1]
Major Experimental Uses
This strain is especially useful for research into type 2 diabetes and obesity. Its disease phenotype is severe, making it a good model for extreme varieties of hyperglycemia, hyperinsulemia, insulin resistance, and obesity. KKAy mice also find an outlet in the study of dermatological abnormalities, endocrine defects, and immunology.
Given this strain’s severe obesity, it would likely not be the best choice for paradigms involving a high degree of strenuous movements, such as the rotarod test or forced swimming test. Researchers would be advised to opt for a more lean mouse if they wish to employ such techniques.[1]
References
- 002468 – KK.Cg-A/J. 2018. 002468 – KK.Cg-A/J. [ONLINE] Available at: https://www.jax.org/strain/002468. [Accessed 17 November 2018].
- MGI – Inbred Strains: KK. 2018. MGI – Inbred Strains: KK. [ONLINE] Available at: http://www.informatics.jax.org/inbred_strains/mouse/docs/KK.shtml. [Accessed 17 November 2018].
- Nakaoka, Hirotomo & Mogi, Masaki & Kan-No, Harumi & Tsukuda, Kana & Ohshima, Kousei & Wang, Xiao-Li & Chisaka, Toshiyuki & Bai, Hui-Yu & Shan, Bao-Shuai & Kukida, Masayoshi & Jun, Iwanami & Horiuchi, Masatsugu. 2015. Angiotensin II type 2 receptor signaling affects dopamine levels in the brain and prevents binge eating disorder. Journal of the renin-angiotensin-aldosterone system. 16.10.
- Akiko Sakata, Masaki Mogi, Jun Iwanami, Kana Tsukuda, Li-Juan Min, Fei Jing, Kousei Ohshima, Masaharu Ito and Masatsugu Horiuchi. 2011. Improvement of cognitive impairment in female type 2 diabetes mellitus mice by spironolactone. Journal of the Renin-Angiotensin- Aldosterone System. 13(1). 84–90.