Introduction
Mice have proven central to scientific research over the past century, serving as one of the most trusted and ubiquitous animal models for the study of diseases, behaviour, and pharmacology. To ensure that data obtained in mouse experiments transfers well to a human context, it is necessary to develop and hone techniques for the proper care of laboratory mice and to give due consideration to possible effects of husbandry practices on experimental observations.
The accurate measurement of a range of behavioural phenomena is crucial for the proper assessment of mouse responses to drugs, disease states and other stimuli of interest. Such phenomena include depression, aggression, locomotion, and anxiety. Mood-related variables such as anxiety are of special concern in the endeavour to optimise mouse husbandry, as they can be significantly affected by differences in handling technique.
In this article, we will discuss how improper handling techniques can raise the anxiety level of model mice in a way that potentially confounds experimental results. We will then examine the evidence for how more carefully crafted handling techniques, such as “tunneling” and “cupping”, can help mitigate this issue.
Anxiety & Experimental Performance
It could be argued that as long as the handling technique is consistent, the anxiety induced by improper handling will constitute a controlled variable, and hence will not skew experiment results. However, this counterpoint misses several key issues, such as the fact that researchers who have not seriously considered the deleterious effects of their handling technique on mouse behaviour are unlikely to have taken steps to develop a consistent handling protocol.
Other issues include the need to be able to compare results between different batches of mice, different experiments, and different research groups and institutions. Scientific research is not considered good and reliable if it cannot be replicated by independent researchers; lack of consideration for handling technique could prevent successful replication and cast doubt on the validity of a large body of otherwise valuable scientific work.[1]
Scientists have noted a few ways that anxiety could affect mouse performance in particular kinds of experiment. Heightened anxiety reduces focus, and will make it harder for mice to react appropriately to a stimulus of interest, like the urine of another mouse. Tasks that require learning, memory, and problem solving (such as the frequently used T maze) will become more difficult. Motivation to explore and move around could be reduced, affecting performance in paradigms such as the elevated plus maze, open field, light-dark box, and forced swim test.[1]
With the open field test for example, while improper handling might not affect the test’s ability to assess overall anxiety level, it will prevent researchers from assessing the reaction of the mice to a particular stimulus, on account of the reduction in focus in highly anxious mice.[1]
It is clear then that confounding effects of improper handling cannot be ignored, and that a detailed consideration of handling optimisation is necessary.
The Downsides of Tail Handling
In the methods sections of their publications, researchers have frequently described handling mice by the tail. For instance, in Wahlsten et al (2003)[2] we are told that mice were grasped by the tail during the elevated plus maze, a test specifically designed to measure anxiety. The researchers, clearly recognising the risks involved in this handling technique, make an effort to assure the reader that the handling was done “quickly but smoothly”.
Similar practices are described in Deacon (2006)[3] and Leach & Main (2008).[4] A charge might be made that cherry-picking and criticising
the protocols of particular studies does not give an overall view of how researchers, in general, are handling mice. But this criticism would miss the most important point, which is that any improper handling exhibited in high impact literature will prevent the successful replication of data that many researchers rely upon.
It is well known both within and outside the sphere of scientific research that mice do not like being handled by their tails, and that such practice causes them great distress, elevating their anxiety levels. Such is the basis for the popular tail suspension test, in which depression-like behaviour is measured according to how much a mouse struggles to escape when lifted up by its tail.
It may be that the tail handling technique rose to prominence in an effort to minimise the risk of being bitten by the mice. Alternative techniques have now been developed to supplant this popular but arguably ill-advised method, without raising any risk of biting. These techniques demonstrate great promise in allowing researchers to deal with their mouse models in a way that does not risk the confounding issues detailed above.
Alternative Handling Methods
In light of the shortcomings of tail handling, a research group at the University of Liverpool’s Institute of Integrative Biology has done extensive research[5][6] into the effects of alternative handling techniques on murine anxiety levels. The group compared the tail handling method to two alternative methods referred to as “tunnelling” and “cupping”, designed to minimise unnecessary, stress-inducing physical contact with the model mice.
Anxiety levels in the mice were measured according to three criteria: (1) how much the mice voluntarily approached and interacted with experimenters, (2) how much the mice urinated and defecated during handling (with increased waste excretion interpreted as a sign of higher anxiety), and (3) how much the mice performed the “stretch-attend” posture in the elevated plus maze, which is associated with a cautious style of investigation.
The publications of this group are currently the only high profile research that focuses specifically on this issue. Hence, care must be taken to ensure that their results are confirmed by other groups before definitive conclusions can be drawn.
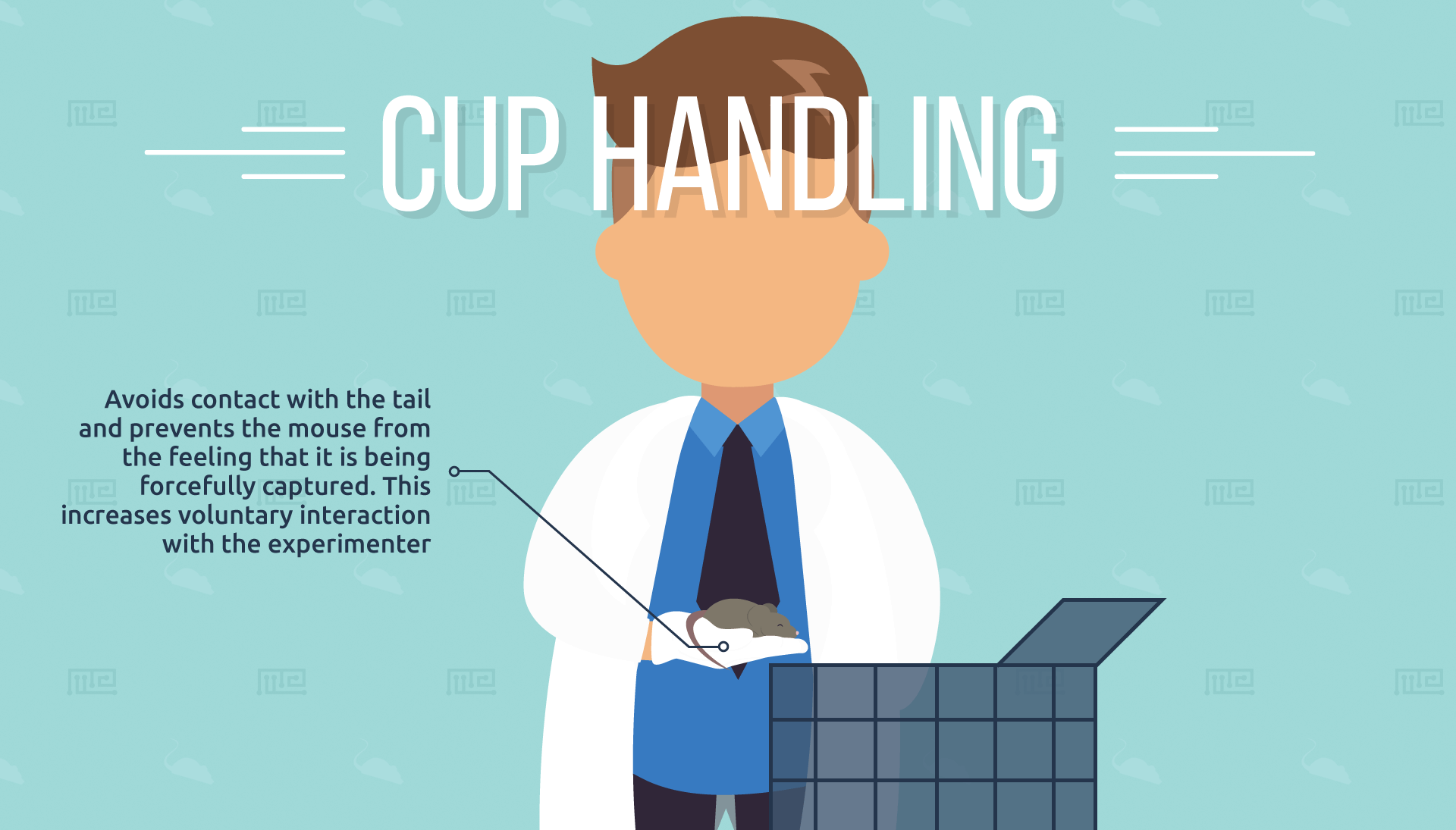
Cup Handling
For a mouse to be “cupped”, it must be trained to walk onto and remain on the open gloved hand of the experimenter. (A slight amount of hand closure is required initially to teach the mouse that it must not jump off when lifted).[5] The researcher can then move their hand to the desired destination, and unfurl their fingers to allow the mouse to voluntarily step off. This method avoids contact with the tail and should prevent the mouse from the feeling that it is being forcefully captured.
The researchers found that cupping significantly increased voluntary
interaction with experimenters compared to tail handling. These results were seen with all three strains that were tested (BALB/c, C57BL/6, and CD1). Interestingly, cupping was more successful at increasing voluntary interaction with female mice, especially for the BALB/c strain. The incidence of stretch-attend posture in the elevated plus maze was also significantly reduced with this method. Results on the frequency of waste excretion are not reported.[5]
It is also interesting to note that acclimatization to this new, gentler handling method was associated with a lessened anxiety response if the mice are then later lifted by the tail or the scruff of the neck.[5] It appears that switching to the cupping method put the mice at ease with handling in general, allowing the researchers to safely perform many different kinds of interaction, even those which would otherwise induce high anxiety.
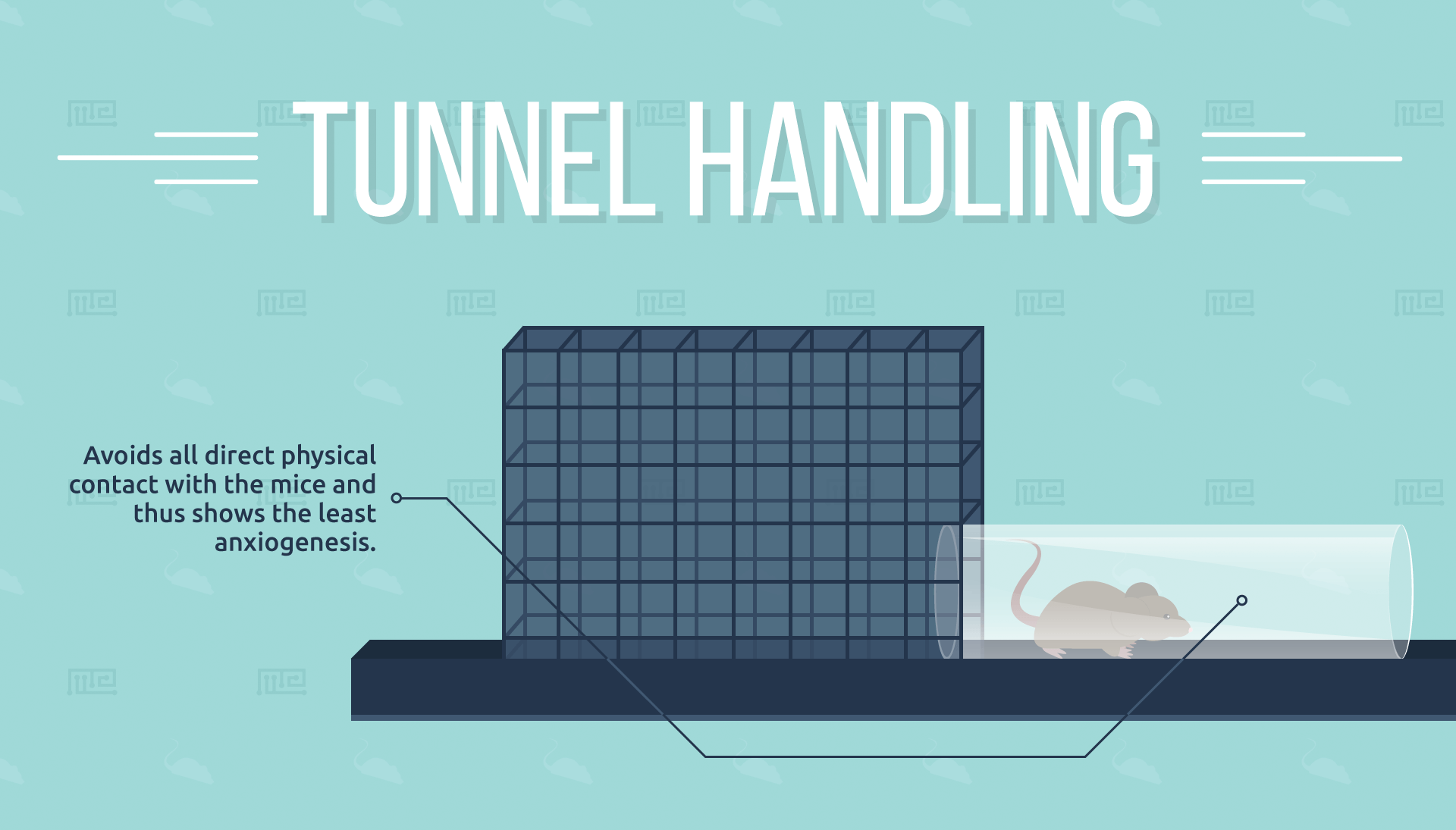
Tunnel Handling
With the tunnelling method, mice are permitted to walk into a cylindrical, acrylic tunnel, which is then carried to a new location.[5] This method avoids all direct physical contact with the mice and thus would be expected to show the least anxiogenesis. However, it requires more equipment and a greater degree of training than the cupping method.
The major advantages of the tunnelling method are seen with the more naturally anxious strains such as C57BL/6: while experimental data suggest that tunnelling and cupping are about equally effective for the less anxious BALB/c and CD1 mice, tunnelling results in double the
frequency of voluntary interaction compared to cupping with the C57BL/6J strain.
A second study examined the tunnelling method in greater detail, comparing tunnels that are shared between multiple mice to a personal home tunnel which is only used for one mouse and is permanently kept in that mouse’s enclosure.[6] The experimenters report that the home tunnel provides significantly better results than the shared tunnel for C57 mice, although not for ICR mice. They also add that a home tunnel avoids possible confounding effects on behaviour from mice of opposite sex sharing the same apparatus.
Tunnelling, specifically tunnelling with a non-shared tunnel, is advantageous for handling naturally anxious mouse strains, as well as helping to mitigate confounding variables. It is further speculated that the presence of tunnels in the cages could reduce stress and depression by providing the mice with a more enriched exploratory environment.[6] However, cupping also appears to significantly reduce anxiety and is preferable when it is necessary to avoid additional expenses or training.
Conclusion
The traditional method of handling model mice by their tails has been shown to significantly elevate their anxiety level, and interfere with their ability to properly perform experimental tasks. However, newly developed handling methods, causing less distress to mice, have been demonstrated to lead to a much more fruitful relationship between researchers and laboratory animals. It is hoped that these new methods will strengthen the validity and reliability of future data.
References
- Gouveia, K. and Hurst, J. L. Optimising reliability of mouse performance in behavioural testing: the major role of non-aversive handling. Sci. Rep. 7, 44999.
- Wahlsten D, Metten P, Phillips TJ, Boehm SL 2nd, Burkhart-Kasch S, Dorow J, Doerksen S, Downing C… Different data from different labs: lessons from studies of gene-environment interaction. J Neurobiol. 2003 Jan;54(1):283-311.
- Deacon RM. Housing, husbandry and handling of rodents for behavioral experiments. Nat Protoc. 2006;1(2):936-46.
- Leach, MC & Main, DCJ. An assessment of laboratory mouse welfare in UK animal units. Animal Welfare, Volume 17, Number 2, May 2008, pp. 171-187(17).
- Hurst, JL & West, RS. Taming anxiety in laboratory mice. Nature Methods volume 7, pages 825–826 (2010).
- Gouveia K, Hurst JL (2013) Reducing Mouse Anxiety during Handling: Effect of Experience with Handling Tunnels. PLoS ONE 8(6).