Introduction
Habituation of animals to a testing apparatus is a crucial pre-experimentation condition for the proper execution of behavioral analyses in many tests. However, in certain cases, exposure of the animal to the testing apparatus prior to experimentation may significantly interfere with subsequent results. Additionally, in cases which require or benefit from pre-experimentation environmental habituation, the procedure varies on the conditions of the number and duration of exposures as well as the specific rodent model being used. Thus, care must be taken in deciding whether or not to habituate animals to a given testing apparatus and if so, under what conditions.
Primarily, habituation to a testing apparatus transforms a potentially negative and unpredictable reaction experience into a potentially positive and more predictable response. In the case of experiments which do not rely on environmental novelty (e.g. social interaction, novel object recognition, conditional maze testing) this apparatus habituation may define the difference between an inconsistent and a consistent behavioral profile. Conversely, tasks which rely on environmental novelty (e.g. open field exploration, conditioned place preference) lose significant power from any pre-experimentation exposure and are therefore not appropriate opportunities for environmental habituation.
Here, we will discuss the impacts of and protocols for apparatus habituation in both tests which rely on environmental novelty and tests for which environmental novelty is not a parameter of interest.
Environmental novelty tests
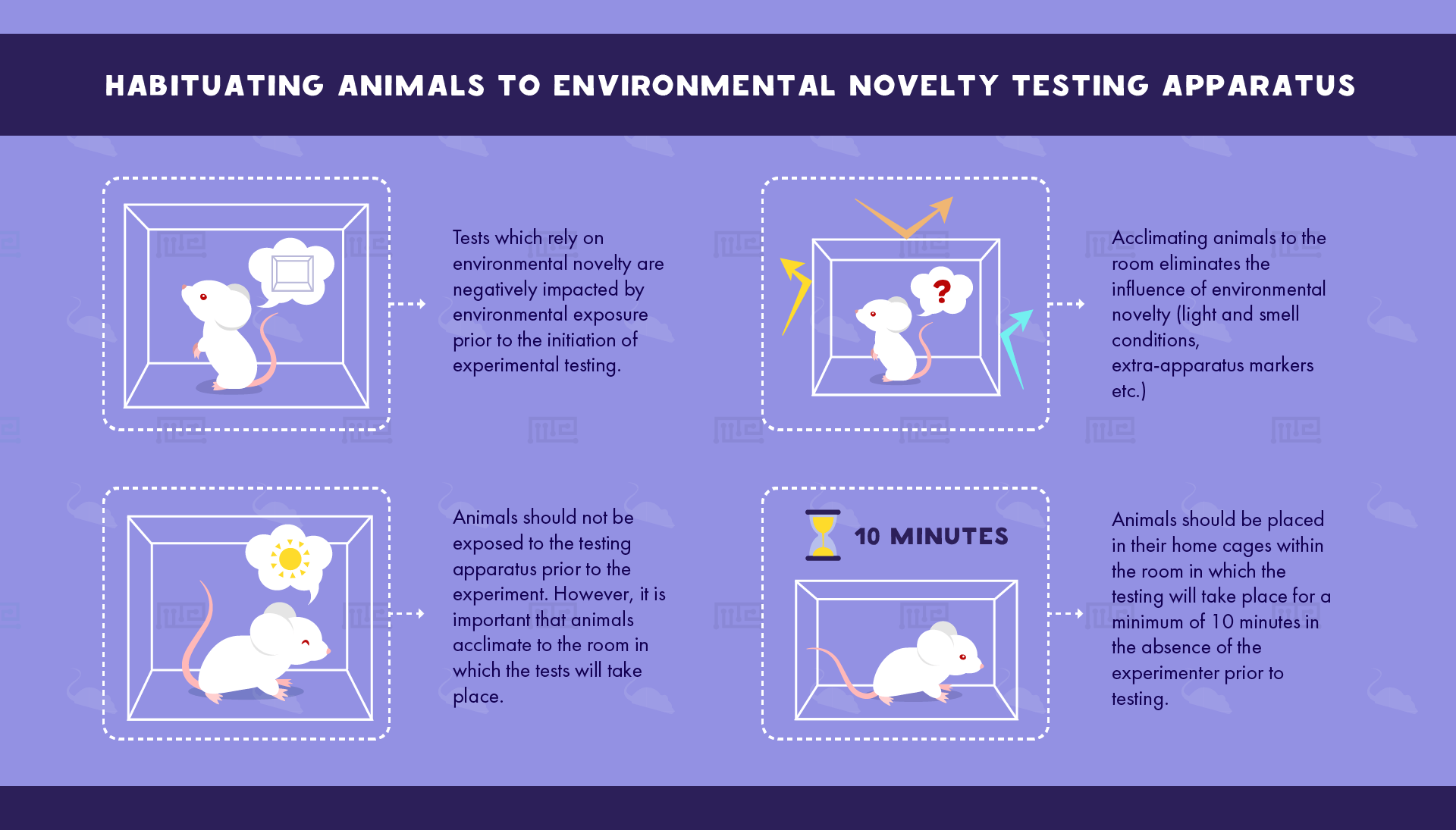
Tests which rely on environmental novelty are negatively impacted by environmental exposure prior to the initiation of experimental testing. Thus, animals should not be exposed to the testing apparatus prior to the experiment. However, it is important that animals acclimate to the room in which the tests will take place in order to eliminate the influence of environmental novelty (light and smell conditions, extra-apparatus markers etc.). Thus, animals should be placed in their home cages within the room in which the testing will take place for a minimum of 10 minutes in the absence of the experimenter prior to testing.[1]
As an example, the open field arena and elevated plus maze (EPM) are common apparati employed for a variety of behavioral measurements in rodent models of stress, anxiety, and novelty seeking as well as basic locomotor activity. End-point measurements of these tests rely on an animal’s unfamiliarity with the environment in order to assess time spent exploring perceived zones of safety (borders and corners for the open field, closed arms for the EPM) versus zones of risk or novelty (open center for the open field, open arms for the EPM). Repeated exposure to these apparati results in more time spent in the latter zones, as the animal recognizes decreased levels of risk in the now-familiar environment.[2] Thus, apparatus habituation will negatively impact behavioral performance in the open field and EPM tasks by diminishing the arena’s novelty.[3]
However, animals should still be acclimated to the room in which the testing apparatus will be used. Pre-experimentation exposure to the environmental area using the home cage procedure noted at the beginning of this section (using a duration of 30 minutes[4]) will habituate the animal to the room and therefore eliminate confounding novelty variables. Conversely, exposure to the EPM itself, for example, will have a deleterious effect on behavioral performance. [5]
Morris water maze
The morris water maze (MWM) is one of the most common tasks for the assessment of spatial memory. Animals use spatial cues (most often visual cues) in order to help locate a platform hidden in opaque water in which they must otherwise continuously swim. Several variables determine whether or not habituation to the MWM apparatus is appropriate prior to experimentation. First, depending on the duration of the test, many animals (especially rats) will not actively seek escape but will rather explore the swimming arena continuously. This is more common in short-duration experiments, for example, those lasting less than one minute. In this case, pre-experiment exposure to the arena with the platform exposed (i.e. un-dyed water in order to reveal the platform location) will indicate the presence of the platform, thereby ensuring that the animal understands the breadth of the task.
Additionally, a pre-experimentation acquisition exposure to the MWM arena performed in the absence of spatial cues (such as objects or wall-markings within the room which are visible from within the MWM) eliminates the confounding variable of environmental novelty in measurements such as distance moved and time spent per quadrant in the acquisition phase of the test.
Conversely, the absence of a platform may induce significant stress in animals seeking an escape and thus confounds any measures of learning acquisition with increased levels of stress and anxiety as a result of the no-platform habituation procedure. Similarly, exposure to the arena in a no-platform condition followed by a test in which the platform is present has been argued to be a form of reversal learning, rather than spatial acquisition, and thus significantly changes the interpretation of outcome results.[6]
Thus, while apparatus habituation is not commonly used for tests involving environmental novelty, exposure to the MWM in controlled conditions may be useful for achieving uniform behavioral results in certain circumstances. Researchers using short protocols (i.e. less than one minute) may pre-expose their animals to the MWM in un-dyed water in order to indicate the presence of an escape platform. Importantly, this habituation test should be done in the same environment (i.e. testing room) in which the experiment will take place, however, any spatial cues such as wall markings which are visible from within the MWM should be removed for the duration of the test. This habituation session should be performed on the day prior to the experiment.
Non-environmental novelty testing
Sociability, social novelty, and social defeat
Tests of sociability and social novelty which assess an animal’s social approach/avoidance behaviors as well as cognition and memory in the recognition of novel vs. non-novel intruders rely on non-environmental subjects and therefore benefit from apparatus habituation. Rats or mice which are not habituated to the testing apparatus will exhibit exploratory and investigative behaviors directed towards both the environment and the partner (in the case of a social novelty test) or intruder animals (in the case of a resident intruder test) and therefore conflate the result interpretation for researchers.[7] Animals should be habituated to the social testing apparatus for 5-15 minutes in the absence of the experimenter and without partner animals on the day prior to the commencement of testing. Additionally, on the day of testing the animals should be placed in the testing room, within their home cages, for a minimum of one hour prior to testing. This protocol should be similarly followed for testing of social defeat stress, as non-habituated animals will exhibit anxiety-like responses to the novel environment which will interfere with their behavioral responses to the subsequent social defeat.[8]
Barnes Maze
The barnes maze is a common test of visuospatial learning comprised of an elevated platform illuminated by aversive bright lights with escape holes located on the edge allowing escape to a dark compartment. Prior to experimentation in the Barnes maze, habituation training allows rats or mice to habituate to the light intensity such that they can properly navigate the maze during the following training periods (acquisition and, if applicable, reversal training). In order to accomplish proper habituation to the Barnes Maze, experimenters should place the rodent in the arena, situated under a glass beaker or other appropriately-sized transparent holding container for a duration of 5 minutes. All intra-maze cues should be removed and if present, extra-maze cues should be blocked from view in order to prevent the animal from learning intra/extra-maze location indicators.[9]
Pain assays
Experiments used to assess pain responses in rodents are designed to specifically measure responses directly to painful stimuli. Thus, any behaviors associated with environmental novelty will interfere with the behavioral analysis of stimulus-induced responses. Therefore, animals should be habituated to the testing apparatus prior to experimentation. For example, in order to properly habituate animals prior to the Von Frey test which uses an instrument pushed with increasing pressure into a rat or mouse’s paw until eliciting a paw retraction reflex, rats or mice should be left to freely explore the testing arena until exploratory behavior ceases. This process normally requires 15 minutes or less in rats.[10] Importantly, mice require an extended period of one hour of environmental habituation prior to testing.[11] Similarly, the Hargreaves pain test requires as little as 5 minutes for rat acclimation[12] but 30 or more minutes for the proper environmental habituation of mice.[13] Finally, since the thermal gradient pain threshold test requires environmental restraint, animals must be acclimated to both the testing arena and the restraint apparatus for 5-10 minutes prior to testing.[14]
Novel object (recognition and place tasks)
Performance in learning and memory tasks such as novel object recognition and object placement memory is most frequently measured as an index of the duration of time which an animal spends with one object as compared to another, or as compared to the same object in a different location.[15] Thus, any pre-existing tendency for an animal to spend more time in one area of the arena over another will inherently skew the interpretation of these measures. Rats or mice must, therefore, be habituated to the testing area prior to commencing the acquisition training period of the experiment. In order to achieve environmental habituation, researchers should place the rat or mouse in the testing arena in the absence of any object. This procedure may be done once or twice on the day prior to the first phase of testing and may last between 5 and 20 minutes.[15] As a measure of proper acclimation to the environment, animals should display an approximately uniform duration of time in all areas of the arena.
Operant chambers (choice tasks, self-administration)
Experiments which employ operant chambers, such as discrimination or choice tasks (such as the 5 choice serial reaction time task) and self-administration protocols, require habituation to the apparatus prior to experimentation. Since the desired behavior does not rely on environmental novelty, any attention given by the animal to the apparatus environment will detract from behavioral performance and therefore from the experimental results. The duration of exposure to the operant chamber varies based on the murine model and the particular protocol, but the procedure is similar to other environmental habituation protocols. The animal should be left alone in the operant chamber in the absence of the experimenter and without any cues (such as conditional lights, reward levers etc.) for an equivalent duration of the experiment which follows.
For instance, in an odor discrimination task which normally lasts 45 minutes, animals are to be left in the chamber in the absence of odors for a duration of 45 minutes on the day prior to testing.[16] For self-administration chambers, the acclimatization procedure includes both environmental and operant habituation over the course of several days. First, the animal should be exposed to the chamber for an equivalent duration to the desired experimental session which will follow. Then, the animal can be trained to understand the operant mechanism (e.g. nose-poke or lever) using either the experimental stimulus or another rewarding stimulus (such as sucrose delivery). Again, if the eventual self-administration session is intended to last for one hour, the habituation and training sessions should also be of a duration of one hour.[17]
Conclusions
Clearly, habituating animals to a testing apparatus or environment has significant benefits for many behavioral testing protocols. However, in certain conditions, it may be counterproductive or even harmful to the results to perform a habituation or acclimatization procedure on subject rodents prior to testing. In short, consider whether or not the test you are performing requires environmental novelty for proper behavioral performance before deciding whether or not to habituate your animals to the testing apparatus. Then, if environmental habituation is appropriate, consider the duration of the intended experiment when deciding how to perform the habituation session. In all cases, ensure that animals in all conditions to be used in a test (e.g. drug-treated vs. naive, male vs. female etc.) undergo identical habituation procedures to ensure uniform behavioral outcomes.
References
- Tatem, K. S., Quinn, J. L., Phadke, A., Yu, Q., Gordish-Dressman, H., & Nagaraju, K. (2014). Behavioral and locomotor measurements using an open field activity monitoring system for skeletal muscle diseases. Journal of visualized experiments : JoVE, (91), 51785.
- Stanford, S. C. (2007). The Open Field Test: reinventing the wheel. Journal of Psychopharmacology, 21(2), 134–135.
- Tatem, K. S., Quinn, J. L., Phadke, A., Yu, Q., Gordish-Dressman, H., & Nagaraju, K. (2014). Behavioral and locomotor measurements using an open field activity monitoring system for skeletal muscle diseases. Journal of visualized experiments : JoVE, (91), 51785.
- Komada, M., Takao, K., & Miyakawa, T. (2008). Elevated plus maze for mice. Journal of visualized experiments : JoVE, (22), 1088.
- Walf, A. A., & Frye, C. A. (2007). The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nature protocols, 2(2), 322-8.
- Gulinello, M., Gertner, M., Mendoza, G., Schoenfeld, B. P., Oddo, S., LaFerla, F., Choi, C. H., McBride, S. M., … Faber, D. S. (2008). Validation of a 2-day water maze protocol in mice. Behavioural brain research, 196(2), 220-7.
- Kaidanovich-Beilin, O., Lipina, T., Vukobradovic, I., Roder, J., & Woodgett, J. R. (2011). Assessment of social interaction behaviors. Journal of visualized experiments : JoVE, (48), 2473.
- Golden, S. A., Covington, H. E., Berton, O., & Russo, S. J. (2011). A standardized protocol for repeated social defeat stress in mice. Nature protocols, 6(8), 1183-91.
- O’Leary, T. P., & Brown, R. E. (2013). Optimization of apparatus design and behavioral measures for the assessment of visuo-spatial learning and memory of mice on the Barnes maze. Learning & Memory (Cold Spring Harbor, N.Y.), 20(2), 85–96.
- Quantitative assessment of tactile allodynia in the rat paw. Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL, J Neurosci Methods. 1994 Jul; 53(1):55-63.
- Deuis, J. R., Dvorakova, L. S., & Vetter, I. (2017). Methods Used to Evaluate Pain Behaviors in Rodents. Frontiers in molecular neuroscience, 10, 284
- Hargreaves K, Dubner R, Brown F, Flores C, Joris J. (1988). A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain, 32(1):77-88.
- O’Brien, D. E., Brenner, D. S., Gutmann, D. H., & Gereau, R. W. (2013). Assessment of pain and itch behavior in a mouse model of neurofibromatosis type 1. The journal of pain : official journal of the American Pain Society, 14(6), 628-37.
- Deuis, J. R., & Vetter, I. (2016). The thermal probe test: A novel behavioral assay to quantify thermal paw withdrawal thresholds in mice. Temperature (Austin, Tex.), 3(2), 199-207.
- Antunes, M., & Biala, G. (2011). The novel object recognition memory: neurobiology, test procedure, and its modifications. Cognitive processing, 13(2), 93-110.
- Arbuckle, E. P., Smith, G. D., Gomez, M. C., & Lugo, J. N. (2015). Testing for odor discrimination and habituation in mice. Journal of visualized experiments : JoVE, (99), e52615.
- Kmiotek, E. K., Baimel, C., & Gill, K. J. (2012). Methods for intravenous self administration in a mouse model. Journal of visualized experiments : JoVE, (70), e3739.
