~How many different behavioral tests can be performed on a single animal in their lifetime?~
~How many different behavioral tests can be performed on a single animal in a day/in a battery of experiments?~
If you are a scientist who uses laboratory animals for your experiments then most probably you will be faced with several dilemmas of ethical nature related to your procedures and protocols. Is it appropriate to perform many different behavioral tests on one animal and how long should you wait before using an animal again?
Traditionally, behavioral experiments performed in rodents and other animal species require the use of naïve animals, meaning animals which have not been used in other tests and have not been exposed to these experimental apparatus and conditions. However, there has been a shift and this practice appears to be left behind, as repeated testing and reuse of laboratory animals in related or unrelated experiments have gained ground. Ethical, practical and economic considerations demand the collection of the maximum possible relevant information while minimizing the number of animals.
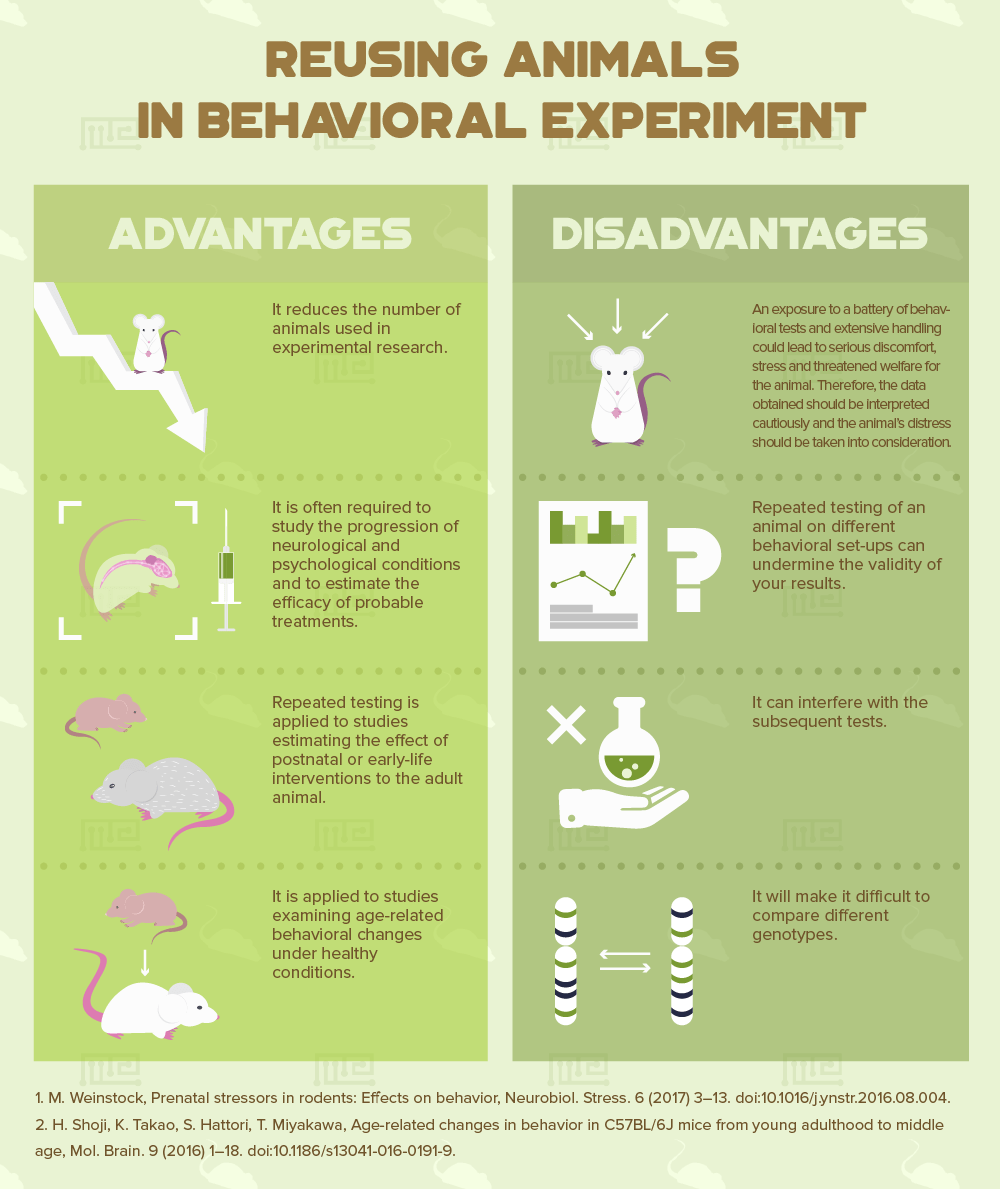
Repeated testing, apart from helping reduce the number of animals used in experimental research, is often required to study the progression of neurological and psychological conditions and to estimate the efficacy of probable treatments. Furthermore, repeated testing has been applied to studies estimating the effect of postnatal or early-life interventions to the adult animal [1] and to studies examining age-related behavioral changes under healthy conditions [2]. Reusing animals can be divided into two broad categories, either using animals in one specific study employing a battery of behavioral tests or in successive studies that may address scientific questions unrelated to those of the first study.
However, there are several disadvantages that need to be addressed when planning to reuse animals in your experimental procedures. An exposure to a battery of behavioral tests and extensive handling could lead to serious discomfort, stress and threatened welfare for the animal. Therefore, the data obtained should be interpreted cautiously and the animal’s distress should be taken into consideration. Moreover, repeated testing of an animal on different behavioral set-ups could undermine the validity of your results, interfere with the subsequent tests and make it difficult to compare different genotypes.
Prior experience is undoubtedly a substantial influence on behavior and personality aspects, but there is still more research to be conducted on the mechanisms and extent of this effect. Võikar et al [3] tested the effect of training history in two of the most commonly used inbred strains of mice (C57BL/6JOlaHsd and 129S2/SvHsd) by using an extensive battery of behavioral tests (12 tests in total). The study showed that earlier experience and training can strongly alter some behavioral aspects. More specifically, mice that underwent the extensive testing showed reduced exploratory activity, emotionality and contextual fear and increased coordination ability and nociceptive sensibility compared to naive mice.
According to the available bibliography, a battery of behavioral challenges includes maximum 8 to 12 tests [2–5]. These tests are normally performed in the course of several weeks, for protocols lasting from 15 to 50 days. Depending on the test and the purpose of each experiment, several trials are required for every test, hence animals are subjected to each test multiple times. Nevertheless, before proceeding to the subsequent test, animals need to be left unchallenged and untrained to recover for at least one day. This recovery time is required not only due to the stressful conditions of the sequence of trials and handling, but also because training and experience can strongly interfere and alter the performance of the animal to the next test. For instance, prior testing has been proved to cause an anxiogenic phenotype by increasing the open arm avoidance behavior (in the elevated plus maze) [6]. To restore open-arm exploration, a 3 week interval between test sessions has been suggested [7].
When it comes to the number of tests that an animal can withstand in its lifetime, there are many factors to consider. As mentioned above, there have been studies proving that certain behaviors change in an age-dependent manner. This is also supported by the fact that some behavioral tests are age-sensitive (such as Morris water maze, Barnes maze, rotarod and fear conditioning) and are constantly used in experiments studying ageing [8]. In addition, another important point is that with each additional experiment, animals get physically and mentally older, since knowledge and experience increase and pile up. This condition could restrict the animal’s performance to a plateau.
Inevitably it would be quite arbitrary to simply set a number as a maximum for the behavioral tests performed on an animal in their lifetime. This number strongly depends on numerous factors ranging from the housing conditions, handling, stress exposure and animal’s genetic line to the nature and intensity of the behavioral tests already performed.
References
- M. Weinstock, Prenatal stressors in rodents: Effects on behavior, Neurobiol. Stress. 6 (2017) 3–13. doi:10.1016/j.ynstr.2016.08.004.
- H. Shoji, K. Takao, S. Hattori, T. Miyakawa, Age-related changes in behavior in C57BL/6J mice from young adulthood to middle age, Mol. Brain. 9 (2016) 1–18. doi:10.1186/s13041-016-0191-9.
- V. Võikar, E. Vasar, H. Rauvala, Behavioral alterations induced by repeated testing in C57BL/6J and 129S2/ Sv mice: Implications for phenotyping screens, Genes, Brain Behav. 3 (2004) 27–38. doi:10.1046/j.1601-183X.2003.0044.x.
- H.V. Lad, L. Liu, J.L. Paya-cano, M.J. Parsons, R. Kember, C. Fernandes, et al., Behavioural battery testing: Evaluation and behavioural outcomes in 8 inbred mouse strains, Physiol. Behav. 99 (2010) 301–316. doi:10.1016/j.physbeh.2009.11.007.
- K.L. Mcilwain, M.Y. Merriweather, L.A. Yuva-paylor, R. Paylor, The use of behavioral test batteries : Effects of training history, Physiol. Behav. 73 (2001) 705–717.
- L.E. Gonzalez, S.E. File, A Five Minute Experience in the Elevated Plus-Maze Alters the State of the Benzodiazepine Receptor in the Dorsal Raphe Nucleus, J. Neurosci. 17 (1997) 1505–1511. http://www.jneurosci.org/content/17/4/1505
- R. Adamec, T. Shallow, Effects of baseline anxiety on response to kindling of the right medial amygdala, Physiol. Behav. 70 (2000) 67–80. doi:10.1016/S0031-9384(00)00247-X.
- J.A. Kennard, D.S. Woodruff-Pak, Age sensitivity of behavioral tests and brain substrates of normal aging in mice, Front. Aging Neurosci. 3 (2011) 1–22. doi:10.3389/fnagi.2011.00009.