Imagine two different people: one spends nearly all of their time in a small apartment. The walls are white and lacking decor, and the rest of the space is bare, except for the basic necessities of food and water. The other individual lives in a space of a similar size, but in addition to food and water has a large window, vivid paintings on the walls, a treadmill, and a television that emits a constant stream of novel images and flashing lights.
Now imagine, after spending weeks at a time in these apartments, how the individuals will respond when they walk out onto a busy city street. The person who receives constant stimuli at home will probably adapt differently, and more favorably than the person without regular environmental enrichment.
Laboratory rodents are no different. The environmental conditions of their home cage can have a profound effect on how they behave outside of the cage. As increased efforts are being made to provide laboratory rodents with enriched environments to better mimic their natural habitats and promote improved health[1], researchers must consider how these environmental conditions will affect experimental outcomes and reproducibility.
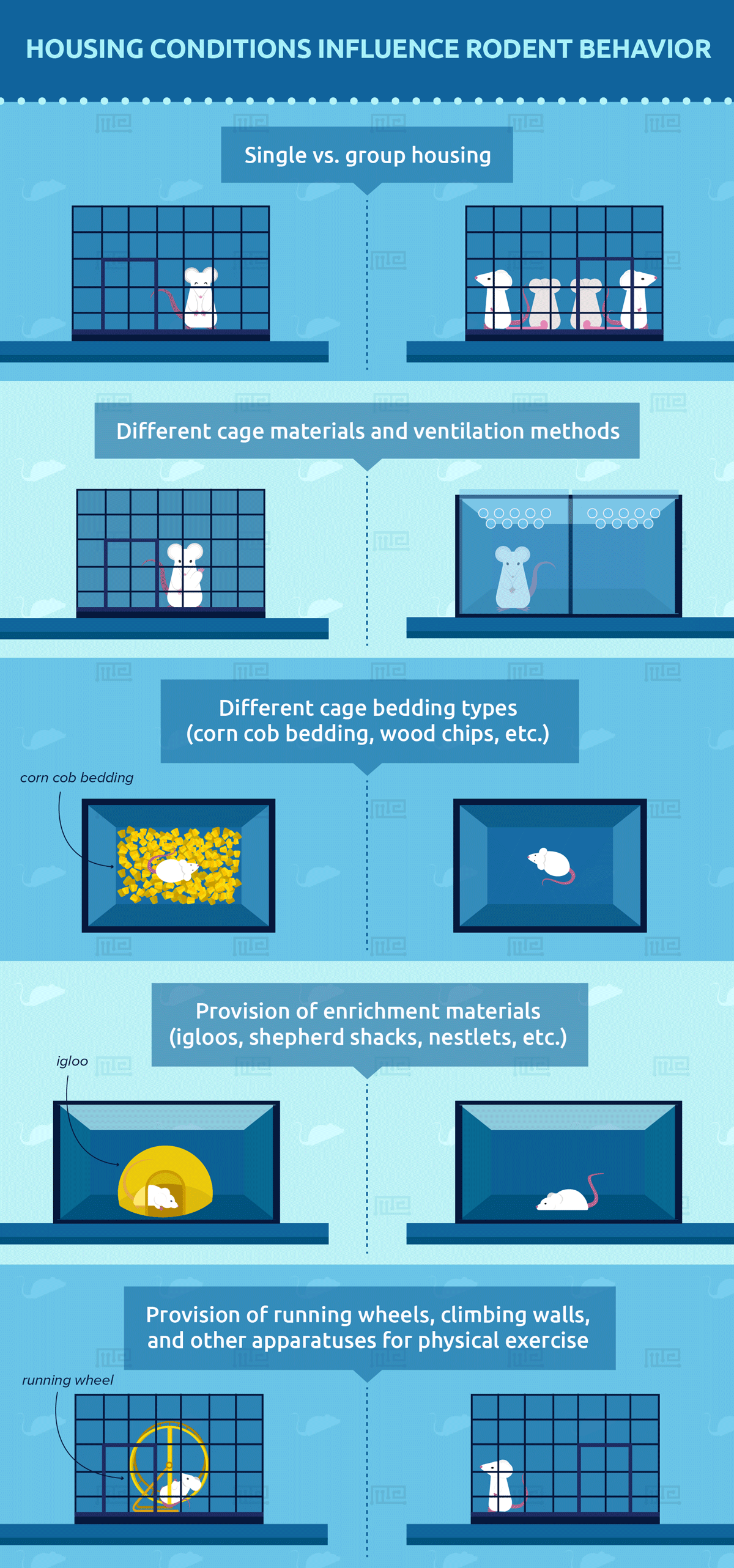
Variables in Animal Housing Conditions
The following are some of the variables in animal housing:
- Single vs. group housing
- Different cage materials and ventilation methods
- Different cage bedding types (corn cob bedding, wood chips, etc.)
- Provision of enrichment materials (igloos, shepherd shacks, nestlets, etc.)
- Provision of running wheels, climbing walls, and other apparatuses for physical exercise
Numerous studies have demonstrated how such housing conditions can modify rodent behavior. For example, mice with environmental enrichment have been shown to eat and weigh more and to be calmer and easier to handle.[2] Specific behavioral tests have also been performed on rodents housed in different conditions.
Single vs. group housing
Depending on the aim and the needs of the experiment, researchers may choose to house their laboratory animals in individual or in group housing of various densities. If the researchers choose to house their animals in single cages, they should take into consideration the fact that isolation can produce stress, which can be expressed in variations in their physiology (stress response hormones, heart rate and blood pressure) and behavior (incidents of anxious and panicky behavior). However, there are conflicting results regarding the stress levels of isolated laboratory rodents. In rats, housing in isolation caused behavioral changes which involve a phenotype similar to that produced by social deprivation, such as reduced mobility, increased tail chasing and self-grooming.[3] In addition, when testing the animal’s reaction to a novel environment, individual housing produced some altered reactivity, which was dependent on various aversive housing conditions (such as light). The effect of isolation since weaning has been widely studied, showing that rats with this history were aggressive when introduced to other conspecifics in an environment with aversive stimuli.[4] Unlike rats, single housing of male mice did not increase the corticosterone levels compared to that of group-housed male mice.[5]
Tackling the “isolation stress”
Some researchers have suggested that rodents housed in isolation should maintain olfactory, visual and auditory contact with conspecifics in order to reduce the effect of the “isolation stress”.[6] Another way to dampen the stress response caused by isolation would be the environmental enrichment of the cage.[7] Enrichment in the form of various toys reduces the baseline levels of ACTH and corticosterone in rats housed in isolation. This is a very important observation since it goes without saying that a chronically stressed animal will perform inconsistently in behavioral tests.
Differences between rats and mice
Mice and rats are social animals; therefore, social interaction is very important for their welfare. It is also mandated by law in many countries, to house laboratory mice and rats in groups. Housing in groups can be easily considered as the ideal condition for laboratory mice and rats, however, results are again contradictory. As described above, rats and mice seem to have different preferences and needs, with different responses to isolation and group housing.
Challenges with group housing in mice – Aggression
It has been shown that social support from conspecifics improves welfare and health outcomes in mice and increases the model’s validity and integrity.[8] Nevertheless, social housing in mice gives rise to an aggressive phenotype which is threatening for the animal’s welfare. Aggressive behavior can lead to injuries and severe wounds, but also compromises the scientific processes. Aggression is a type of intense social stress which introduces into the experiments variability, invalidity and altered physiological state. An issue which is often overlooked is that many researchers use exclusively females to avoid the variable of aggression, a practice which leads to the misrepresentation of sexes in science.
Researchers suggest the implementation of changes in housing conditions, handling, and experimenting; changes which will make captivity more bearable and suitable and will allow mice to control their social interactions and decrease their aggression. Since aggression is mainly affected by the group size and not the cage size, a common suggestion is to avoid housing more than 3 mice in the same cage and try to avoid housing unfamiliar mice together.[9] Likewise, conditions such as temperature and bedding material were found to influence aggression. More specifically, an increase in temperature up to 25°C, increased significantly the aggressive incidents;[10] while corncob bedding was found to elicit some endocrine effects, similarly increasing aggression.[11] Various forms of environmental enrichment act as a crucial parameter which can reduce the aggressive incidents. Enrichment resources, such as nesting material, should be abundant so that competition will be avoided. In addition, enrichment in the form of shelters or cages can also reduce aggression, given that mice can use it as a hiding place from an aggressive conspecific.[8]
The issue of crowding in group housing
While group housing permits the expression of a wide range of social behaviors and activities, researchers should protect the animals from the negative effect of crowding. To avoid crowding, the spatial and social density should be carefully considered. The term spatial density refers to the available space per animal, while social density refers to the number of animals in each group. It has been shown that male rats housed in high spatial and social density experience differences in taste, body weight gain, water intake, and stress response.
Different cage materials and ventilation methods
Factors such as cage shape, height, size and material of construction play a crucial role in the welfare of the animal and its overall psychological state. There are no concluding results on the preference of mice for cage shape, since different strains show various preferences. When choosing a cage shape, we should evaluate each strain’s thigmotactic behavior. When it comes to rats, there are only a few studies supporting the use of rectangular cages. In addition, the height of the cage is crucial for allowing the rats to express their natural habit of upright standing behavior.
Cage color
The color of the cage has been proven to strongly influence mice’s behavioral repertoire. Mice housed in white cages had the highest food consumption, lowest body weight and least anxiety, when tested in the Elevated Plus Maze, compared to mice housed in green, red and black cages. It is important to be consistent on the color of the cage in which the animals are kept from the time of their birth until their adulthood.[12]
Caging systems
In general, there is a variety of caging systems widely used in animal facilities around the globe. The distinct differences between these caging systems is an often-overlooked factor which introduces variability in the behavioral analysis of laboratory strains. There has been considerable research into the effects of various cage ventilatory systems on rodent behavior. A recent study compared the effect of housing in open-top cages (OTC), motor free ventilated cages (MFVC) and individually ventilated cages (IVC) on exploratory and anxiety-like behavior in mice.[13] Mice were tested in the Open Field Test and Elevated Plus Maze, respectively. Mice housed in the MFVCs had increased exploratory and reduced anxiety-like behavior.
Other studies have shown that IVC housing increases social activity and anxiety, but does not affect locomotion or cognition.[14][15] IVC housing may cause these behavioral changes by altering the levels of the neurotransmitters serotonin and dopamine.[16]
Position of the cage
Apart from the effect of the cage materials and ventilation methods, even simpler factors such as the position of the cage on the racks of the facility have been proven to affect physiology and behavior of laboratory rats.[17] Two strains of rats with distinct emotionality-related behaviors and genotype, Lewis (LEW) and spontaneously hypertensive rats (SHR), were tested using the Open field, Elevated plus maze and the Black/White Box to estimate the effect of the position of the home cage (top or bottom shelf of the tank). The two strains when housed in top cages showed less anxiety in the Open Field and Black/White Box tests compared to the rats housed in the bottom cages. It is evident that common and often disregarded variations in the laboratory setting can interact with genotype in behavioral tests of anxiety.
Different cage bedding types
Another very important factor to consider when organizing your behavioral experiments are the materials chosen for bedding and nesting. Without a doubt, bedding and nesting materials help animals express more of their species-specific behavior and attempt some degree of control over their environment. Several studies have shown that there are preferences for the various types of bedding in rats and mice, as every bedding material has various potential effects and experimental impact. Older studies showed no preferences over six different types of nesting materials in males and females of two strains of mice (BALB/c & C57BL/6J).[18] However paper-derived materials such as tissues, towels and paper strips were preferred over wood-derived materials. Differences were also observed when various densities of bedding were used in groups of male BALB/c and C57BL/6 mice.[19] Although no behavioral differences were observed, mice showed a strong preference for large bedding volumes. A significant difference was that mice housed in shallow bedding engaged more in nest-building behavior compared to groups housed in larger volumes.
Of course, nest building (using bedding provided) can act as an indicator of the health condition and the emotional state of laboratory mice. A recent study showed that changes in nest complexity can help scientists identify any irregularities in the welfare and handling of mice. Problems which when left unattended could seriously affect the results of a behavioral analysis.[20]
Contrary to mice, adult laboratory rats do not spontaneously engage in nest-building. Van Loo and Baumans (2004)[21] have shown that nest building in rats is an acquired behavior, as rats learn to utilize the nesting material only if it was provided from birth. Similarly, rats have been shown to prefer the type of bedding on which they were raised.[22]
In a very interesting experiment, researchers tested the effect of bedding on the 50-kHz ultrasonic vocalization in laboratory rats.[23] Ultrasonic vocalizations have been thoroughly studied as an indication of a rat’s emotional state, and more specifically the 50-kHz is emitted when rats are petted and rewarded, treated with drugs of abuse or exposed to a clean housing cage. Interestingly, the specific study showed that rats emit these calls significantly more when put in cages with bedding material. This can help us conclude that the general cage conditions such as the bedding can create a more pleasant environment for the rats and can alleviate the aversiveness of an experimental context.
Provision of enrichment materials
Environmental enrichment has been widely used as a term referring to all the items placed in an animal’s cage as an effort to improve the animal’s physiological and psychological state. The basis of it is that the researcher needs to provide all the necessary environmental stimuli which can help the animal express the species-specific activities (such as nest building) and can diminish all the causes of abnormal behavior (such as self-injurious behavior).[24] Official guidelines suggest that environmental enrichment should include social interaction, chewing, locomotion, nest building and hiding, and manipulating, carrying and hoarding food and objects.
A significant amount of studies support the benefits of enrichment materials on the psychological and physiological state of laboratory animals. Laboratory mice housed in an enriched environment exhibited an increased behavioral repertoire and a reduction in offensive behavior and aggression.[2]
However, studies of environmental enrichment on anxiety have produced varying results. The results may be strain-dependent, as van de Weerd et al. found that BALB/c mice provided with nesting material, a nestbox, and a tube in their home cage had reduced locomotion in the open-field test, whereas this outcome was not seen in C57BL/6 mice provided this same enrichment.[25]
Conversely, another study showed that the effect on elevated plus maze activity was sex-dependent, where enriched males had increased locomotion in the elevated plus maze, and enriched females had reduced locomotion.[16] Other studies have demonstrated that enrichment reduces anxiety-related behavior in mice.[26][27]
Female mice housed in standard cages were more anxious and fearful compared to mice housed in an enriched environment, a phenotype expressed by an increase in the self-administration of anxiolytics.[28] In addition, mice in enriched environments spent significantly more time active and exploring the cage.
When considering the provision of enrichment materials, the researcher should consider that different genetic strains respond differently to environmental enrichment protocols. There is an extensive body of evidence showing that the introduction of novel enrichment objects and frequent changes in the enrichment material can induce different emotionality and sensory responsiveness in various strains.[29] ICR(CD-1) mice, which is a strain less stressed and more explorative, interacted in a greater extent to various novel enrichment objects compared to C57BL/6 mice.[30] These findings indicate that behavioral studies should be conducted under systematically recorded variations in environmental enrichment, or else data can easily be misinterpreted.
Provision of running wheels, climbing walls, and other apparatuses for physical exercise
Enrichment in the form of various apparatuses such as running wheels and climbing walls is considered a form of voluntary exercise in laboratory animals. Numerous studies have highlighted the beneficial effects of voluntary exercise on the behavior and physiology of laboratory animals. More specifically, experiments with voluntary wheel running improved the spatial memory in mice tested in Water Maze.[31] In addition, environmental enrichment combined with voluntary exercise, improved the object recognition memory and contextual fear conditioning in mice with diminished hippocampal neurogenesis.[32] Finally, female BALB/c and 129/Sv mice housed in an enriched environment with voluntary exercise, showed an increased social preference when tested in the 3-chamber sociability test.[33]
In one study, mice from standard and enriched housing conditions were studied in the open-field test, elevated plus maze, and barrier test.[34] The enriched conditions included a wooden climbing frame and a plastic inset for use as a hiding place. In the barrier test, the latency to climb over the barrier was shorter in the mice with enriched housing. Other studies have confirmed that rodents with physical enrichment perform better in behavioral tests with a climbing component.[26]
These are just some examples of how variations in cage conditions can affect rodent behavior. Standardization of housing conditions is an important aspect of preclinical research, and careful consideration should be given to the habitat (and any changes thereto) of animals to be used in behavioral testing.
References
- Würbel H. Ideal homes? Housing effects on rodent brain and behaviour. Trends Neurosci. 2001 24(4):207-11.
- Van de Weerd HA, Aarsen EL, Mulder A, Kruitwagen CL, Hendriksen CF, Baumans V. Effects of environmental enrichment for mice: variation in experimental results. J Appl Anim Welf Sci. 2002 5(2):87-109.
- L.J. Hurst, C. Barnard, C. Nevison, C. West, Housing and welfare in laboratory rats: welfare implications of isolation and social contact among caged males., Anim. Welf. 6 (1997).
- N. Wongwitdecha, C.A. Marsden, Social isolation increases aggressive behaviour and alters the effects of diazepam in the rat social interaction test., Behav. Brain Res. 75 (1996).
- C. Hunt, C. Hambly, Faecal corticosterone concentrations indicate that separately housed male mice are not more stressed than group housed males., Physiol. Behav. 87 (2006) 519–526.
- P. Brain, D. Benton, The interpretation of physiological correlates of differential housing in laboratory rats., Life Sci. 24 (1979) 99–116.
- E.E. Belz, J. Kennell, R. Czambel, R. Rubin, M. Rhodes, Environmental enrichment lowers stress responsive hormones in singly housed male and female rats., Pharmacol. Biochem. Behav. 76 (2003) 481–486.
- E.M. Weber, J.A. Dallaire, B.N. Gaskill, K.R. Pritchett-Corning, J.P. Garner, Aggression in group-housed laboratory mice: Why can’t we solve the problem?, Lab Anim. (NY). 46 (2017) 157–161. doi:10.1038/laban.1219.
- Van Loo, P.L., Mol, J.A., Koolhaas, J.M., V Van Zutphen, B.F. & Baumans,V V. Modulation of aggression in male mice: influence of group size and cage size. Physiol. Behav. 72, 675–683 (2001).
- Greenberg, G. T The effects of ambient temperature and population density on aggression in two inbred strains of mice, Mus musculus. Behavior 42, 119–130 (1972).
- Villalon Landeros, R R. et al. C Corncob bedding alters the effects of estrogens on aggressive behavior and reduces estrogen receptor-alpha expression in the brain. Endocrinology 153, 949–953 (2012).
- C. Sherwin, E. Glen, Cage colour preferences and effects of home cage colour on anxiety in laboratory mice., Anim. Behav. 66 (2003) 1085–1092.
- A. Polissidis, S. Zelelak, M. Nikita, P. Alexakos, M. Stasinopoulou, Z.I. Kakazanis, et al., Assessing the exploratory and anxiety-related behaviors of mice. Do different caging systems affect the outcome of behavioral tests?, Physiol. Behav. 177 (2017) 68–73. doi:10.1016/j.physbeh.2017.04.009.
- Logge W, Kingham J, Karl T. Behavioural consequences of IVC cages on male and female C57BL/6J mice. Neuroscience. 2013 237:285-93.
- Burman O, Buccarello L, Redaelli V, Cervo L. The effect of two different Individually Ventilated Cage systems on anxiety-related behaviour and welfare in two strains of laboratory mouse. Physiol Behav. 2014 124:92-9.
- Pasquarelli N, Voehringer P, Henke J, Ferger B. Effect of a change in housing conditions on body weight, behavior and brain neurotransmitters in male C57BL/6J mice. Behav Brain Res. 2017 Aug 30;333:35-42.
- G.S. Izídio, D.M. Lopes, L. Spricigo, A. Ramos, Common variations in the pretest environment influence genotypic comparisons in models of anxiety, Genes, Brain Behav. 4 (2005) 412–419. doi:10.1111/j.1601-183X.2005.00121.x.
- V. de H. Weerd, V.P. Loo, V.L. Zutphen, J. Koolhaas, V. Baumans, Nesting material as environmental enrichment has no adverse effects on behavior and physiology of laboratory mice, Physiol. Behav. 62 (1997) 1019–1028.
- J. Freymann, P.P. Tsai, H. Stelzer, H. Hackbarth, The impact of bedding volumes on laboratory mice, Appl. Anim. Behav. Sci. 186 (2017) 72–79. doi:10.1016/j.applanim.2016.11.004.
- B.N. Gaskill, K.R. Pritchett-Corning, Nest building as an indicator of illness in laboratory mice, Appl. Anim. Behav. Sci. 180 (2016) 140–146. doi:10.1016/j.applanim.2016.04.008.
- P.L.P. Van Loo, V. Baumans, The importance of learning young: the use of nesting material in laboratory rats., Lab. Anim. 38 (2004) 17–24.
- T. Ras, M. Van de Ven, E.G. Patterson-Kane, K. Nelson, Rats’ preferences for corn versus wood-based bedding and nesting materials, Lab. Anim. 36 (2002) 420–425. doi:10.1258/002367702320389080.
- C. Natusch, R.K.W. Schwarting, Using bedding in a test environment critically affects 50-kHz ultrasonic vocalizations in laboratory rats., Pharmacol. Biochem. Behav. 96 (2010) 251–259. doi:10.1016/j.pbb.2010.05.013.
- K. Bayne, H. Würbel, The impact of environmental enrichment on the outcome variability and scientific validity of laboratory animal studies., Rev. Sci. Tech. 33 (2014) 273–80. http://www.ncbi.nlm.nih.gov/pubmed/25000800.
- van de Weerd HA, Baumans V, Koolhaas JM, van Zutphen LF. Strain specific behavioural response to environmental enrichment in the mouse. J Exp Anim Sci. 1994 Aug;36(4-5):117-27
- Chamove AS. Cage design reduces emotionality in mice. Lab Anim. 1989 23(3):215-9.
- Chapillon P, Manneché C, Belzung C, Caston J. Rearing environmental enrichment in two inbred strains of mice: 1. Effects on emotional reactivity. Behav Genet. 1999 29(1):41-6.
- I. Olsson, C. Sherwin, Behaviour of laboratory mice in different housing conditions when allowed to self-administer an anxiolytic, Lab. Anim. 40 (2006) 392–399.
- V. Tucci, H.V. Lad, A. Parker, S. Polley, S.D.M. Brown, P.M. Nolan, Gene-environment interactions differentially affect mouse strain behavioral parameters, Mamm. Genome. 17 (2006) 1113–1120.
- C.J. Nicol, S. Brocklebank, M. Mendl, C.M. Sherwin, A targeted approach to developing environmental enrichment for two strains of laboratory mice, Appl. Anim. Behav. Sci. 110 (2008) 341–353. doi:10.1016/j.applanim.2007.05.006.
- A. Garthe, I. Roeder, G. Kempermann, Mice in an enriched environment learn more flexibly because of adult hippocampal neurogenesis, Hippocampus. 26 (2016) 261–272.
- M.E. Sakalem, T. Seidenbecher, M. Zhang, R. Saffari, M. Kravchenko, S. Wördemann, et al., Environmental enrichment and physical exercise revert behavioral and electrophysiological impairments caused by reduced adult neurogenesis., Hippocampus. 27 (2017) 36–51.
- J.W. Whitaker, S.S. Moy, K.R. Pritchett-Corning, C.A. Fletcher, Effects of enrichment and litter parity on reproductive performance and behavior in BALB/c and 129/Sv mice., JAALAS. 55 (2016) 387–399.
- Lewejohann L, Reinhard C, Schrewe A, Brandewiede J, Haemisch A, Görtz N, Schachner M, Sachser N. Environmental bias? Effects of housing conditions, laboratory environment and experimenter on behavioral tests. Genes Brain Behav. 2006 5(1):64-72.