In regular conversation, guinea pigs are near synonymous with scientific experimentation. Yet despite this ubiquitous metaphor, guinea pigs are not often the rodent of choice in modern scientific research. Some other rodent models are far more common, namely the mouse or the rat. The utility of these widespread rodent models is clear. They satisfy the normal standards for good animal models (e.g. easy and quick to breed, relatively low-cost). They have been well-characterized in a large variety of experimental techniques and lend themselves to some methods that are not currently possible in many other species, such as transgenics or optogenetics. Still, there are unique ways in which the guinea pig contributes to research that serves to retain its value as an animal model. As we continue to characterize its physiology and behavior, the possible insights we may glean from the guinea pig can only grow.
How the Guinea Pig Enabled Major Biomedical Breakthroughs
The scientist Dr. Robert Koch was awarded the 1905 Nobel Prize in Physiology or Medicine for his discovery of the bacterium that causes tuberculosis. Much of this famous work, widely regarded as one of the greatest scientific breakthroughs in medicine, was carried out in guinea pigs. These rodents have been a fixture in biomedical research and guinea pig models of disease, including the guinea pig model of tuberculosis, have endured in the modern era.
Guinea pigs have an immune response that more closely resembles humans compared to other rodent models. The guinea pig is an especially good choice for a rodent model in the field of immunology. Guinea pigs are particularly susceptible to infection, which means that lower doses of infectious agents may be required to trigger a response from the animal. Studies of tuberculosis, for example, necessitate only a small aerosol dose of M. tuberculosis bacteria to infect a guinea pig.[1] Such susceptibility is an advantageous feature of an animal model of disease, as it reduces potential exposures to the experimenter.
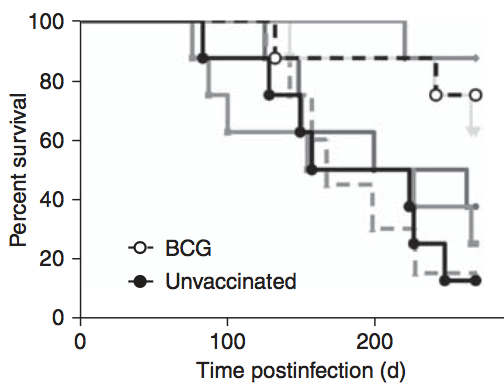
Figure 1. Survival plot of guinea pigs treated with test vaccines for tuberculosis (solid gray lines) compared to unvaccinated animals (solid black line) and animals treated with the Bacillus Calmette-Guérin (BCG) vaccine (dashed line).[1]
While a guinea pig model of tuberculosis may not exhibit many outward signs of infection until much later in the progression of the disease, the pathologies that do present are both more varied and more similar to human pathologies than other rodents.[1] Furthermore, their immune responses have contributed greatly to our understanding of innate and adaptive immunity. As such guinea pigs are an especially common choice for research on vaccines. Their immune similarity to humans renders them capable of recognizing certain antigens while other rodent models cannot.[1] By virtue of this, vaccines that would not function in mice can still be tested in guinea pigs. While the guinea pig model of tuberculosis is among the most famous applications of the guinea pig to disease research, they are still used to study the effects of more current disease outbreaks and to screen possible interventions, including the recent outbreak of the Ebola virus.[2]
Guinea pigs have a variety of unique physiological traits that set them apart from their popular mouse and rat counterparts. In addition to their high susceptibility to infection, they also demonstrate responses to allergens, including sensitive skin, respiratory, and anaphylactic responses.[1] This led to the early adoption of guinea pigs for research on allergic reactions. They also have different dietary necessities than other laboratory rodents. This particular feature of the guinea pig led to the discovery of vitamin C in 1907, because unlike other small rodents they do not naturally produce vitamin C.[3] Instead guinea pigs, like humans, must rely on an external source of this essential nutrient.

Figure 2: New hair cells grow in the cochlea of young adult guinea pigs following Math1 gene therapy.[2]
The guinea pig auditory system is relatively similar to that of humans. Auditory research has also seen significant advances from work with guinea pig models. Guinea pigs have a similar ear structure and hearing range to humans, and their ears are easily accessible for experimentation.[1] These little rodents will even conveniently let you know they can hear you; the outer ear of the guinea pig will move in response to auditory stimuli, a behavior known as the Preyer reflex. Researchers leveraged these unique features to perform the first in vivo generation of new cochlear hair cells in a mature mammal in 2003.[4] This study employed viral-mediated overexpression of Math1, a developmental gene that encodes hair cell differentiation, in young adult guinea pigs. Following gene therapy with the adenovirus encoding Math1 overexpression into existing nonsensory cells of the cochlea, these cells converted into new hair cells and
recruited axons from neurons in the organ of Corti. Not only were these experiments the first to grow new hair cells in a mammal, but also the first to demonstrate cellular regeneration in a tissue that does not naturally regenerate.
Uniquely “Guinea Pig” Behaviors
The physiological traits that distinguish guinea pigs from other rodents represented opportunities to develop better models for specific experimental goals, and subsequently led to great scientific breakthroughs. Likewise, the guinea pig possesses unique behaviors that may also be better suited to behavioral research than its murine counterparts. The early life of young guinea pigs presents perhaps the most distinct contrast from the early lives of either mice or rats. While murine models are born in an altricial state and must rely heavily on their mothers, guinea pig pups are precocious and are fairly well-developed at birth.[5] Mouse pups need three weeks with their mother before they can be weaned if they are to survive, but a guinea pig pup can be up and about and feeding itself in just one day.
The heavy dependence on the mother in mouse and rat models has contributed to a wide body of research on the physiological effects of early life care. Studies from these rodents have demonstrated that pups who were groomed more frequently by their mothers are likely to show increased growth of new synapses in the hippocampus and superior performance on learning and memory tasks.[6] That mouse and rat pups are born in such a relatively undeveloped state requires that mouse and rat mothers are highly attentive to ensure the continued positive development of their offspring.
Guinea pigs, on the other hand, do not exhibit very much maternal behavior. Perhaps this is due to their pups’ rather advanced developmental stage at birth. However, this is not to say that guinea pig pups are not attached to their mothers. On the contrary, guinea pigs are very social and demonstrate prominent attachment to their mothers. Young guinea pigs that are separated from their mothers exhibit a two-phased stress response.[4] First, the pup moves about restlessly and vocalizes. This “active” phase of distress is accompanied by activation of the hypothalamic-pituitary-adrenal (HPA) axis, a cornerstone of the physiological stress response. This short-lived active phase is followed by a prolonged passive response in which the infant guinea pig assumes a crouched stance with closed eyes and its hair will stand on end.
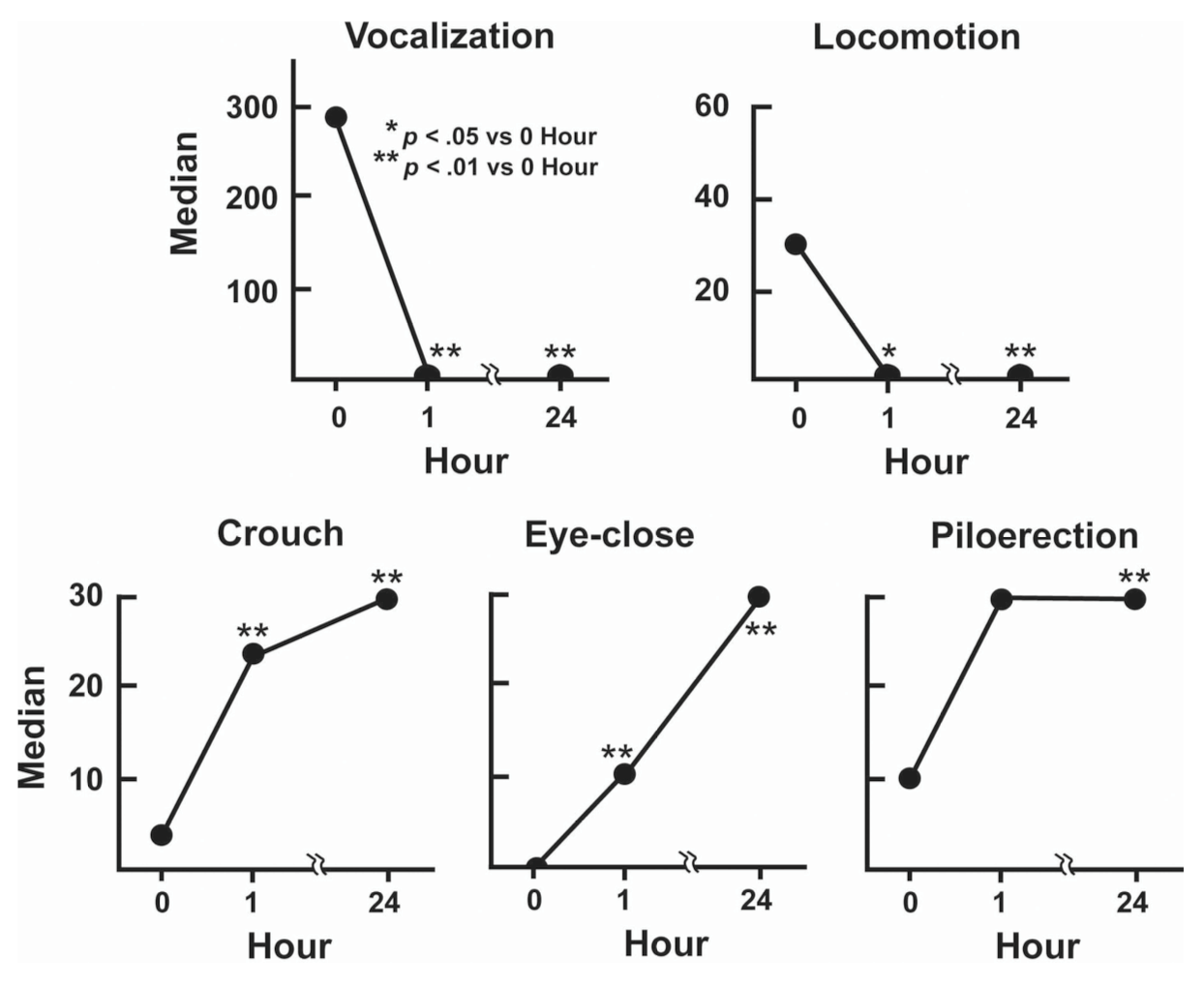
Figure 3: Behaviors seen during the two-phase active/passive separation response following isolation of infant guinea pigs in a novel environment.[3]
This characteristic separation response is not simply an infant guinea pig’s reaction to being left alone. If the pup were to be placed inside the novel environment with a littermate, indicators of stress such as cortisol levels would remain as high as if the pup were in the arena alone.[5] Instead, it is believed that this stress response is indicative of the pup’s strong emotional attachment to its mother. Consequently, guinea pigs may provide more relevant insights into the psychobiological effects of maternal separation than the conventional altricial rodent models. In fact, the two-phase separation response and prolonged depressive demeanor have already been observed in macaques. Thus, guinea pigs represent a pertinent model of maternal separation and may be preferable to nonhuman primates for many studies.
Characterizing the Guinea Pig’s Behavioral Repertoire
While researchers have found robust analogs for human behavioral phenotypes in mouse and rat models, the rather profound similarity between guinea pigs and macaques following maternal separation suggests that there could be other nuances within the guinea pig behavioral repertoire that offer a closer approximation to human behaviors than other conventional rodent models. As such there have been efforts to more fully characterize the guinea pig as a behavioral model. One such study published in 2014 by Kiera-Nicole Lee and colleagues [7] utilized PhenoTyper cages with video tracking software to monitor the behavior of Hartley guinea pigs (an outbred guinea pig strain commonly used in research). Their goal was to provide a detailed description of the baseline behavior of the guinea pig. This kind of characterization is absolutely essential if the guinea pig is ever to be adopted as a common behavioral model in the lab. A brief summation of their findings, as well as how those findings relate to popular mouse models, follows.
- Guinea pigs prefer shelter. The subjects of this study had three zones within their PhenoTyper cage: a hidden sheltered zone, an open zone, and a feeding zone. The guinea pigs spent most of their time in the hidden zone by a wide margin – approximately 65% of their time was spent in the shelter. When they were out in the open, most of that time was spent in the feeding zone. Furthermore, their time spent in the shelter was significantly greater during the light phase compared to the dark phase. Thus, the guinea pigs appear to view the sheltered area as a refuge. Preference for closed areas rather than open ones is a feature shared by mice, and the strength of this preference is generally accepted as a method of quantification for anxiety-like behavior. More anxious subjects tend to demonstrate a stronger preference for closed areas to open ones, and this preference is exaggerated under anxiety-inducing conditions.
- Guinea pigs will freeze when startled. Freezing behavior is a common metric in mouse and rat behavioral studies. The authors of this study also noted freezing behavior present in guinea pigs, particularly when they first entered the experimental room or loud noises occurred outside the room. This could indicate that the guinea pig would be well-suited to assays that rely on freezing behavior as a primary measurement, such as fear conditioning paradigms.
- Guinea pigs exhibit thigmotaxis. Mice placed in an open field arena will often demonstrate thigmotaxis – that is, when they move about they tend to stick by the walls of the arena. It is generally thought that this proximity to the wall provides a sense of safety. When used as an anxiety assay, time spent in the periphery of the open field compared to the center is viewed as a sign of greater anxiety. Guinea pigs also displayed thigmotaxis during this behavioral monitoring study. They explored their entire cage but tended to spend most of their time in the periphery and also traveled greater distances during the dark phase.
- Guinea pig open field behavior is responsive to drug administration. Following baseline characterization of some guinea pig behaviors, the authors characterized the effects of a variety of drugs via intraperitoneal (IP) injection to see if any subsequent behavioral changes would be observed. This is a common framework for experiments in murine models, in which mice or rats are given a drug and then allowed to freely explore while an experimenter (or video tracking software) monitors behaviors generally related to locomotor activity. While robust conclusions regarding these data and the mechanisms that underlie them cannot yet be determined, these experiments contribute valuable information to the overall characterization of drug-induced guinea pig behavior. The guinea pigs responded to these drugs in the following ways:

Figure 4: A Hartley guinea pig. (photo: Charles River Laboratories).
SCH23390: This D1 receptor antagonist reduced the amount of time guinea pigs spent in the hidden zone of their cage, which may indicate a decrease in anxiety-like behavior. Sulpiride (D2R antagonist) and SKF38393 (D1R agonist) did not produce effects on occupancy time in different zones.
Methamphetamine: Guinea pigs that received either high or low dose amphetamine (10 mg/kg and 1 mg/kg, respectively) both exhibited increased locomotion compared to saline-injected controls. Only the high dose treatment resulted in a significantly greater distance traveled over the entire observation period. Furthermore, chronic administration of methamphetamine over the course of seven days revealed that only guinea pigs that received the high dose exhibited the behavioral phenomenon known as behavioral sensitization, in which the locomotor effects of a drug increase following repeated administration (i.e. the opposite of tolerance).
Guinea Pigs as a Modern Animal Model
In practice, use of the guinea pig in behavioral research would require many of the same considerations as the use of other murine models. Their housing needs are relatively similar and common behavioral setups may be easily adapted to work with a guinea pig. Many behaviors overlap between mice, rats, and guinea pigs, which can facilitate the scoring of guinea pig behavior during standard mouse or rat assays. Of course, some tools cannot yet be readily translated to a guinea pig model. For example, transgenic tools are most accessible for experimenters that use mice in their research. However, there are services available to aid in the creation knockin and knockout lines for guinea pigs.[8] If the guinea pig becomes a popular choice for behavioral experiments, then readily available transgenic lines may not be too far behind.
Overall, the guinea pig has been a valuable animal model in biomedical research for well over a century. Although they are not currently a common choice for behavioral studies, there is considerable evidence that the guinea pig has distinct features among rodent models that may be advantageous in the study of brain-behavior interactions, particularly with regard to questions of development, emotional attachment, audition, and immune function. With further characterization, the guinea pig will continue to serve the scientific community for years to come.
Reference
- Clark, S., Hall, Y., and Williams, A. (2015). Animal Models of Tuberculosis: Guinea Pigs. Cold Spring Harbor Perspect Med 2015;5ca018572.
- Cross, R.W., Fenton, K.A., Geisbert, J.B., Mire, C.E., and Geisbert, T.W. (2015). Modeling the disease course of the Zaire ebolavirus infection in the outbred guinea pig. JID 212: S305-S315.
- Holst, A. (1907). Experimental studies relating to “ship-beri-beri” and scurvy. Journ of Hyg 7(5): 619-633.
- Kawamoto, K., Ishimoto, S., Minoda, R., Brough, D.E., and Raphael, Y. (2003). Math1 gene transfer generates new cochlear hair cells in mature guinea pigs in vivo. J Neurosci 23(11): 4395-4400.
- Hennessy, M.B. (2014). Filial attachment and its disruption: Insights from the guinea pig. Dev Psychobiol 56(8): 1747-1754.
- Liu, D., Diorio, J., Day, J.C., Francis, D.D., and Meaney, M.J. (2000). Maternal care, hippocampal synaptogenesis and cognitive development in rats. Nat Neurosci 3(8): 799-806.
- Lee, K., Pellom, S.T., Oliver, E., and Chirwa, S. (2014). Characterization of the guinea pig animal model and subsequent comparison of the behavioral effects of selective dopaminergic drugs and methamphetamine. Synapse 68(5): 221-233.
- http://www.renovalife.com/gene%20editing.php
