
A transgenic zebrafish embryo in which GFP is conditionally expressed in radial glial cells. [Brion et al., 2012].
The zebrafish (Danio rerio) is a very commonly used model organism in biological sciences. As with some other models, their genome is fully sequenced and transgenic lines of zebrafish can be made. Additionally, they offer several unique advantages that other models simply can’t offer. They are especially useful to developmental biologists, for example, because in addition to a relatively quick rate of development, zebrafish embryos are transparent. This makes it substantially easier to monitor the development of this species, especially when fluorescent proteins like GFP (green fluorescent protein) are employed.
While the zebrafish has been incredibly valuable to developmental and cancer biologists, it hasn’t made the same inroads into behavioral research that other models have made. Behavior studies often make extensive use of rodent models. There are many assays that can be run with rodent subjects and lots of effort has been put into determining which natural rodent behaviors make the best analogs for the human condition. It has been somewhat more challenging for researchers to fully characterize the behavior of zebrafish in a way they could translate to human behavior. These animals require an aquatic environment, which inherently requires some extra translation when the animals one ultimately seeks to understand are terrestrial. Likewise, developing the behavioral assays themselves is not always as straightforward in this distinct environment. Whenever designing a behavioral experiment with any kind of animal model, it is wise to leverage the natural behaviors of that species.
These kinds of innate behaviors should reflect inner behavioral states that would occur in nature, and then can be translated to relevant human behavioral states. To the casual observer, the natural behavior of a fish may seem wholly unrelated to that of a human. However, zebrafish and humans share a common ancestry, and as such there are some behaviors that have been conserved over the course of evolution. They demonstrate patterned social behaviors, can learn through Pavlovian or operant conditioning, and have navigational abilities that can all be translated to human behaviors. While we have not yet investigated natural zebrafish behaviors in as much detail as other model organisms, comprehensive catalogs of zebrafish behavior have been assembled to aid in the translation of their behavior to different species[1].
Here we will review commonly used behavioral assays that utilize these detailed zebrafish behaviors to shed light on neuroscientific questions. Some zebrafish assays are rather analogous to rodent behavioral assays. These tests employ behaviors that have been relatively conserved between species and translated into an aquatic environment for use in zebrafish. Other assays measure behaviors that are more unique to zebrafish, but they are still useful to us because the underlying mechanisms of those behaviors may have cross-species relevance.
Considerations for Testing Zebrafish Behavior
As with behavioral research in any species, it is important to consider the specific needs of your experiment before you begin. Although several of the following assays are remarkably similar to some used with rodent models, zebrafish will inherently require special consideration due to their need for an aquatic environment. It may seem obvious, but it can be easy to overlook simple things like water temperature or pH if working with fish is a new endeavor. Zebrafish are generally kept in tanks with a temperature between 26 and 28.5 ֯C with a pH between 6.8 and 7.5[2]. Behavioral testing tanks should not stray from these guidelines, as it can be a shock to any fish to suddenly change temperatures or pHs. Just like rodents, certain behavioral testing environments will be more or less stressful to a zebrafish, so it is important to know whether or not your setup induces stress (intentionally or unintentionally). A zebrafish in a novel environment will generally dive to the bottom of the tank and look for dark or concealed places, so be sure to take note of the size and depth of the tank as well as whether or not the setup should include some shelter (as in the rotating drum concealment assay described below).
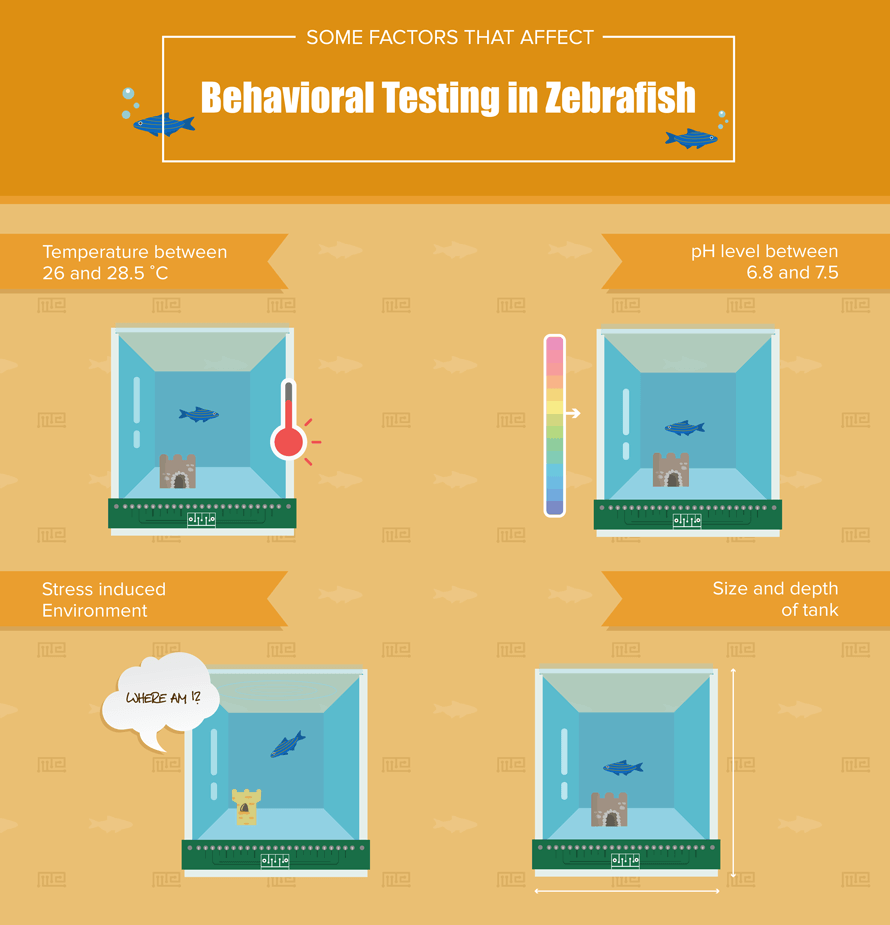
Finally, the tests outlined below are primarily designed to assess the behavior of a single subject. However, there has been an increase in the use of zebrafish to study social behavior due to their tendency towards shoaling behavior. Shoaling refers to a group of fish swimming together; it differs somewhat from schooling based upon the group’s pattern of movement. In a test that will involve more than one zebrafish subject, it is important to understand that shoaling behaviors of zebrafish can vary based upon different factors, including the number of fish present, the sexes of the fish present, and the novelty of the environment[3].
Common Zebrafish Assays
The following are some Zebrafish assays that we will consider.
Conditioned Place Preference
The conditioned place preference test is a great example of a zebrafish behavioral assay that has a clear analog in the world of rodent behavioral tests. Just as in the rodent assay, zebrafish are placed into an environment with three chambers. In one of the side chambers they will be presented with an unconditioned stimulus, which can either be positive (e.g. cocaine) or aversive (e.g. shock). The testing phase consists of placing the fish back into the middle chamber and assessing how much time is spent in either of the side chambers. Like rodents, zebrafish will demonstrate a preference for whichever chamber is associated with reward (or not associated with punishment).
Diving Response Test
The diving response test is inherently unique to zebrafish, as rodents don’t typically dive into their cages. However, it compares very nicely to the open field test when used as an anxiety assay. Zebrafish will naturally dive to the bottom of a tank when placed in a novel environment. Only with time will the fish begin to explore the upper regions of the tank. The longer this latency, the more anxiety-like behavior the fish is displaying. This is very similar to thigmotaxis in rodents. When a mouse is first placed into an open field arena, it tends to stay towards the edges of the arena where it feels safe against the walls. With time, the mouse ventures into the center. This is a prime example of how species-specific behaviors can be translated between environments.
Exploratory Biting (Bite Test)
The bite test is another example of a unique zebrafish behavior that can be smoothly translated to terrestrial animals. In this test, the fish is trained to enter a chamber that contains a novel object. Zebrafish will explore new stimuli by first approaching the object with their right eye and then often by biting at the object. Once the object is no longer novel, exploration with the right eye and biting behavior decrease. This is analogous to the novel object recognition test in rodents. In that test, the rodent is presented with two objects. In the next round of testing, one of those items is swapped with a new object. The rodent is likely to spend more time investigating the novel object, provided that it recognizes that the other object is not novel. Both of these tests allow us to observe recognition and habituation behaviors across species.
Rotating Drum Concealment Assay
In the rotating drum concealment assay, zebrafish are placed inside a tank with a rotating drum. On the drum is a vertical black band, which moves around the drum. In the center, there is a cylinder behind which the fish can hide. This behavior relies on the zebrafish’s reflex to conceal itself behind a known barrier when threatened and is directly analogous to targeted response concealment behavior in mice, in which a mouse will flee directly to a known place of refuge when threatened. This behavior has been especially useful when adapted for vision testing. Zebrafish have excellent color vision, and differentially contrasting bands and columns can be used to assess the fish’s ability to see whether or not there is somewhere to hide during this test.
T-Maze
The T-Maze, so called because it resembles the letter T, is a maze that is commonly used in rodent behavioral tasks. In this task, the rodent is placed at the end of the long arm of the maze, and when it reaches the T-intersection it has to choose whether to go left or right. The rodents are expected to learn that turning one direction will result in a reward (e.g. food) and the other direction will not be rewarded. The concept is the same in zebrafish – turning in one direction results in feed in that arm, while the other arm goes unrewarded. The task has been modified to take advantage of zebrafish’s exceptional color vision by coloring the arms and conditioning the zebrafish to learn that certain colors are associated with reward while others are not.
Tap-Elicited Startle Reflex
The tap-elicited startle reflex is an assay that takes advantage of a fish behavior we’ve all seen when a child gets too excited in a pet store. It is the scientific equivalent of a kid tapping on the glass and startling the fish, causing it to reflexively swim away. In the laboratory, the overexcited child is replaced by a push-solenoid that delivers taps at a specific frequency. With repeated exposure, the zebrafish will begin to habituate to the taps and become less startled. However, if the frequency of the taps is changed following this habituation, the zebrafish will once again demonstrate a startle reflex, if only for a short time. This can be used as a test of short-term memory in zebrafish, and is related to acoustic startle tests in rodents.
Three-Chamber Maze
The three-chamber maze utilizes a setup up similar to conditioned place preference and a procedure similar to that of the T-maze. The fish is placed in the middle chamber and then given a choice to enter one of the side chambers. Like the T-maze, the chambers may be different colors to leverage the zebrafish’s capable color discrimination skills. In the T-maze a correct choice is rewarded; in this maze the incorrect choice is punished. The walls of this maze can slide, so if the fish enters the wrong chamber, the doors close and the wall can slide towards the tank edge, restricting the area of the chamber. This tight space is aversive to the fish, so it should learn to prefer the more spacious chamber.
As we learn more about innate zebrafish behavior, we can make more detailed translations of that behavior across species. This will help to contextualize zebrafish behavioral experiments among the vast existing literature of rodent behavioral experiments, and even more broadly to human behavior. This model organism has been a fixture of developmental and genetic research thus far, and is continuously making inroads with behavioral studies.
References
- Kalueff, AV et al.. Zebrafish Neuroscience Research Consortium. (2013). Towards a comprehensive catalog of zebrafish behavior 1.0 and beyond. Zebrafish 10(1): 70-86.
- Avdesh, A, Chen, M, Martin-Iverson, MT, Mondal, A, Ong, D, Rainey-Smith, S, Taddei, K, Lardelli, M, Groth, DM, Verdile, G, and Martins, RN. (2012). Regular care and maintenance of a zebrafish (Danio rerio) laboratory: an introduction. J Vis Exp (69): 4196.
- Miller, N, and Gerlai, R. (2012). From schooling to shoaling: patterns of collective motion in zebrafish (Danio rerio). PLoS One (7)
Additional Resources:
Levin, ED, and Cerutti, DT. (2009). Chapter 15: Behavioral Neuroscience of Zebrafish. Methods of Behavior Analysis in Neuroscience, 2nd ed. CRC Press/Taylor & Francis.
