Introduction
Cerebrovascular disease, where blood flow to all or part of the brain is reduced to dangerous levels, is a serious and highly prevalent disorder. It ranks with heart disease as one of the leading causes of death in the modern age, with around ten percent of all deaths globally attributed to it. Most of these individuals are elderly (over the age of 70) but the disease can also strike the young and middle-aged.[1]
Cerebrovascular disease and consequent reduced vascular flow can arise from a number of factors including atherosclerosis from diet or smoking, high blood pressure, diabetes or a genetic predisposition. It also confers a range of pathological behavioral symptoms and leads to a neurodegenerative disorder known as vascular cognitive impairment (VCI) or vascular dementia in more extreme cases.
Understanding the behavioral consequences of this disease is crucial for assisting and treating its sufferers, and mouse models of reduced vascular flow have proven key in that respect. In this article, we will begin by discussing what vascular flow is, and how it relates to the brain and human behavior. We will then go on to examine in detail the different aspects of behavioral change that mouse models of reduced vascular flow have helped elucidate.
The Basics of Vascular Flow and the Brain
The well-regulated flow of blood through the vessels of the cardiovascular system is crucial to the survival and health of every large animal, including humans. Animals larger than an insect require a constant, active circulation of vital gases and minerals around their body because they are too large for passive diffusion between their cells to suffice. This circulation is achieved by a large mass of muscle tissue (viz. the heart) pumping blood through vessels in and around every organ.
One of the most crucial substances carried by blood is the gas oxygen, which is transported by haem-containing proteins in red blood cells. Oxygen accepts an electron at the end of respiration, the process by which cells produce the vital coenzyme ATP. Without oxygen, cells are unable to respire, all their active processes cease, and they die within minutes.
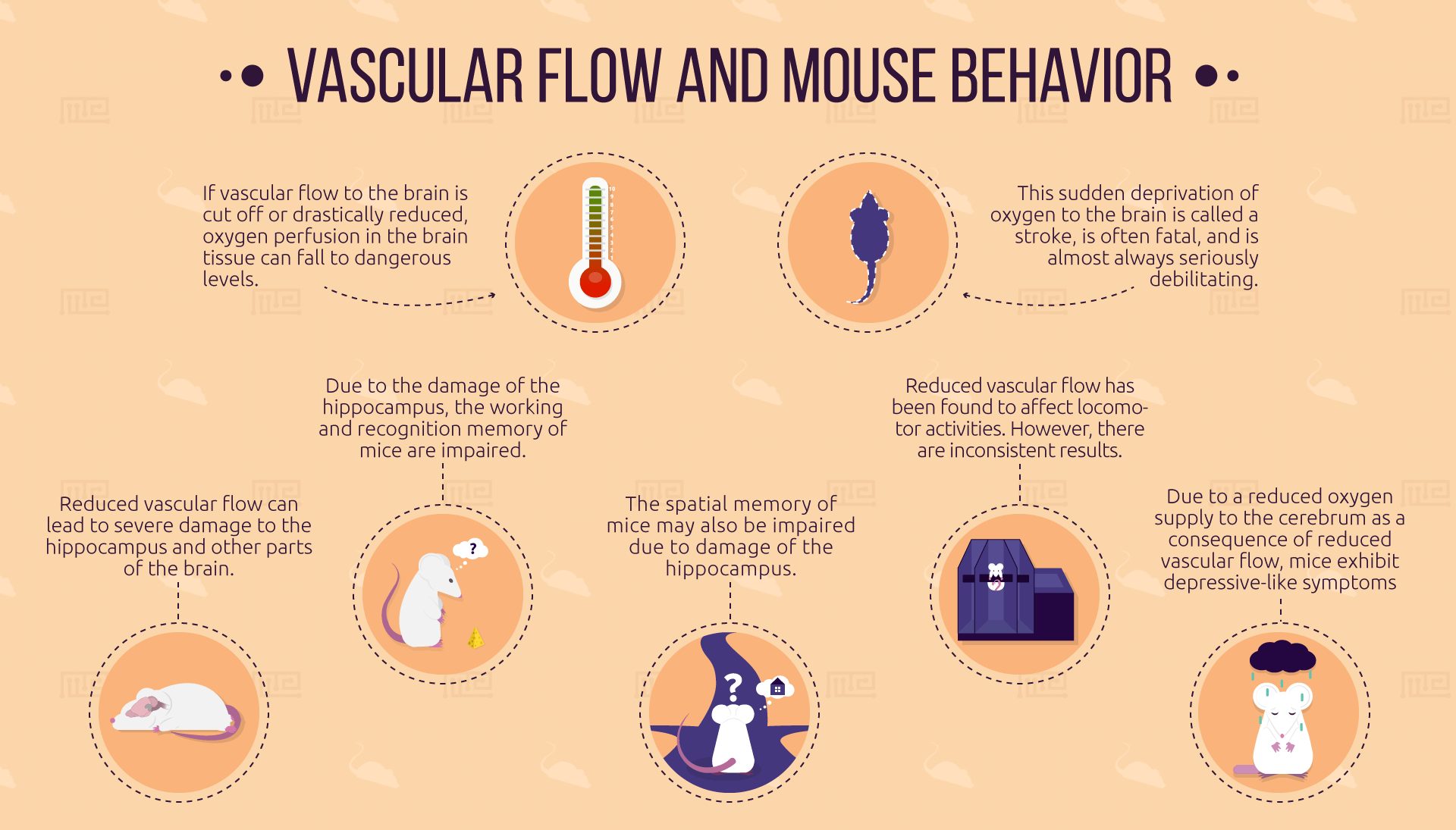
This issue is especially acute with the brain, which requires more energy to function than any other part of the body. If vascular flow to the brain is cut off or drastically reduced, oxygen perfusion in the brain tissue can fall to dangerous levels. This sudden deprivation of oxygen to the brain, called stroke, is often fatal and is almost always seriously debilitating. Furthermore, the gradual death of brain cells due to reduced blood flow can induce vascular cognitive impairment (VCI) and dementia (VCD).
As the organ that engages in cognition, and controls voluntary motor action, the brain is the central factor in animal behavior. A drastic reduction in the presence of oxygen and other key substances (such as sodium, potassium, and water) in the brain, as a result of insufficient vascular flow, can have a number of noticeable behavioral consequences, including deficits in working memory and mood, and possible deficits in spatial memory and motor coordination.
Vascular Flow and Recognition Memory
An American study[2] published in 2015 looked into the behavioral effects of severe damage to the hippocampus of the mouse brain, caused by reduced vascular flow. In this case, the flow reduction was achieved by blocking one of the carotid arteries, the main vessels in the neck that supply blood to the brain. The mice then showed a marked reduction in the size of their hippocampi, the parts of the brain mainly responsible for various forms of learning and memory.
Compared to controls who had undergone “sham surgery”, these vascularly impaired mice displayed impaired recognition memory in the novel object recognition test. Normal mice spend more time investigating objects that they have not seen before than familiar objects. The control mice showed this normal behavior, but the mice with reduced vascular flow spent equal time investigating both novel and familiar objects, suggesting they have impaired recognition memory.
It is interesting that this damage to the hippocampus, which is most commonly associated with spatial memory and long-term episodic memory formation, manifested as deficits to non-spatial recognition memory. And as detailed in the following section, this recognition memory impairment was not the only effect on non-spatial memory seen with a reduced vascular flow in mice.
Vascular Flow and Working Memory
Deficits have also been observed with such mice in working memory. A Japanese study[3] from the following year, using mice whose vascular flow impairment had been achieved in the same way, reports that these mice performed significantly worse than controls in the Y maze test. The authors note that the mice displayed a phenotype that was markedly similar to that of humans with vascular dementia.
The Y maze test is similar to the more commonly used T maze test, but with its arms positioned at a slightly different angle. In the first trial, the mouse finds and retrieves food from one of the arms. In further trials, mice are expected to alternate between the arms in search of food, remembering where they searched before. Mice that do not display this alternated searching are considered to have reduced working memory capacity.
These results are corroborated by a British study from 2011,[4] where mice with brain hypoperfusion induced through carotid microcoils exhibited impaired working memory in the radial arm maze. This maze is similar to the Y and T mazes, but has many spokes radiating from the center; mice that repeatedly re-enter the same spokes significantly more frequently than controls are said to have worse working memory.
It seems clear that mice with impaired vascular flow show deficits in working and recognition memory. However, the Y maze, T maze, and radial arm maze tests are more precise tests of spatial working memory. Thus, results from tests that focus more specifically on spatial memory allow us to tease apart the consequences of this condition for spatial memory and working memory.
Vascular Flow and Spatial Memory
The consequences of vascular flow impairment for spatial memory in mice are more controversial, and no strong conclusions can be drawn from the literature available.
The working memory deficits caused by vascular flow abnormalities seen in the 2015 American study[2] did not appear to extend to pure spatial memory. The control and diseased mice in that study were also tested in the Morris water maze, where mice must use spatial learning and memory to find a raised platform in a pool of water (to which mice show a naturally aversive behavior). No significant difference was seen between the two groups of mice in this test.
The 2011 British study mentioned above,[4] which found spatial working memory deficits in the radial arm maze, concurs with the results of this 2015 American study; the researchers report no significant difference between vascular flow impaired mice and normal mice in the Morris water maze. The 2016 Japanese study mentioned above[3] also reports the same result, and suggests, based on MRI data, that hippocampal neuronal death in the mice was not extensive enough to impair spatial memory.
However, the reliability of these observations has been called into question by other studies. A German study, also from 2016,[5] describes inconsistent Morris water maze performance by vascular flow impaired mice. In this case, the vascular impedance was induced by constricting the carotid arteries with microcoils. In some experiments, diseased mice performed worse than controls, and in others, they did not.
Experiments detailed in an Indian paper published in 2009[6] included a Morris water maze test, in which mice with cerebral blood flow reduction caused by the antibiotic quercetin performed worse than controls. It is questionable however how comparable this situation is to more common forms of vascular impairment, and so how generalisable these results would be. Other unknown effects of the drug should also be taken into account as a possible confounding variable.
Vascular Flow and Locomotor Capability
The investigation of the locomotor effects of vascular flow impairment has also returned inconsistent results. Observations obtained from the rotarod and wire hang tests in several studies do not concur.
In the rotarod, mice are placed on a rotating cylinder and must keep running forwards in order to avoid falling off. Mice that fall off significantly more quickly or at slower speeds than controls are interpreted as possessing motor coordination deficits. The wire hang test requires the mouse to cling onto a frame of narrow metal wires until it falls to the ground. More rapid falling is taken as a sign of motor impairment. The wire hang test is more focused on strength and endurance than coordination.
The Japanese study published in 2016[3] observes that VCI mice performed worse than controls in both the wire hang and rotarod tests. However, another Japanese study[7] from the same year gives the exact opposite observations, reporting no significant difference between bilateral carotid artery stenosis mice and controls in the rotarod or wire hang test. Nevertheless, the third study from 2014[8] corroborates the findings of the first study, reporting deficits with vascular flow impaired mice in both of these paradigms.
The reason for the unreliability of the locomotor observations is not clear. A systematic review[9] of animal vascular dementia models suggests that the differences between results may reflect both a temporal and severity dependency of the stenosis. That is, mice that underwent stenosis for longer, or that had a more severe occlusion of the carotid arteries suffered greater brain damage and then manifested motor deficits not seen in the mice with more intact brains.
The use of different procedures in the creation of rodent vascular flow impairment models also seems to have had an important effect. The systematic review observes that motor deficits have been reported with rats after severe bilateral carotid occlusion, as well as with mice who have undergone the same procedure, but unilateral carotid occlusion did not produce the same results. Motor abnormalities are not seen in gerbils with bilateral carotid stenosis, nor in mice with hyperhomocysteinaemic vasculopathy (where vascular flow reduction is caused by excess cysteine in the blood).
Hence, given the various significant procedural dependencies found in the literature, it is unlikely that the results seen in locomotor tests with mouse models of vascular flow impairment are suitably robust; it would be hard to justify using them as a model for the motor impairments frequently observed with human vascular cognitive dementia patients.
Vascular Flow and Depression
Human patients with cerebral ischemia (greatly reduced oxygen to the cerebrum, the main part of the brain) display a chronic depressive mood. A similar phenotype can also be inferred from the depressive-like behavior of VCI mouse models. For example, in a Chinese study published in 2018,[10] cerebral ischemia was induced in mice via bilateral occlusion of the carotid arteries. These mice then exhibited depressive-like symptoms in the sucrose preference, tail suspension and forced swim tests.
The sucrose preference test measures the preference of mice for sweet tasting food. A reduced preference for sweet stimuli compared to controls is seen as a sign of anhedonia i.e. reduced ability to experience positive feelings from such stimuli. Anhedonia is frequently reported by human sufferers of chronic depression.
The tail suspension test and forced swimming test look at the immobility of mice in an aversive situation. In the former, mice are dangled upside down by the tail and are expected to wriggle in order to escape. In the latter test, mice are dropped into a tank of water and are expected to swim frantically to get to the edge. Higher immobility in these tests indicates possible depressive symptoms.
In the forced swim and tail suspension tests, it is difficult to separate the effects of mood abnormalities from the effects of locomotor deficits, given that both these tests require mice to perform a dynamic behavior. This would be especially concerning given the inconsistent motor performance observed with these models. However, the inclusion of a non-motor paradigm, as the Chinese researchers have done with the sucrose preference test, allows for the confounding motor variable to be ruled out.
Researchers should bear in mind however that literature linking vascular flow with depression in mice is scarce, and that strong conclusion cannot be based entirely on one study. While other research can be found linking ischemia with depression[11] (in this case, ischemia-induced via lesion), the 2018 Chinese study is the only paper reporting such findings that specifically focuses on vascular flow.
Vascular Flow and Anxiety
While depression and anxiety are often comorbid mood disorders in humans, researchers do not find anxiety in vascularly impaired mouse models.
The Japanese study[7] from 2007 did not find a significant difference in behavior between carotid artery constricted mice and control mice in either the light-dark box test or the open field. In the light-dark box test, mice spending more time in the dark portion of the enclosure compared to the light part is interpreted as a sign of elevated anxiety. Less movement during the open field test, where mice are left free to roam within a square enclosure, is interpreted in the same way.
These open field results are corroborated by the American study published in 2015,[2] where researchers also saw no increased anxiety in carotid occluded mice compared to sham mice in the elevated zero maze. In this paradigm, mice are placed on an elevated circular platform with exposed and closed off sections, and reduced tendency to explore the open areas compared to controls is taken as an index of increased anxiety.
However, one paper published by a Spanish research group in 2017[12] does report elevated anxiety in certain models of mouse cerebral ischemia. In this case, ischemia was induced through direct ligation of cerebral arteries. Occluding the middle cerebral artery in this way increased anxiety-like behavior in the elevated plus maze, but occluding the anterior cerebral artery did not. The elevated plus maze is very similar to the elevated zero maze, but forms a cross shape instead of a circle.
While the results of this paper appear to contradict the general conclusion that vascular flow deficits do not impact anxiety, a more careful analysis of the data is required. The models used in this study differ from the standard carotid occlusion model, which is designed to mirror carotid occlusion in humans. Although some forms of vascular dementia are indeed associated with cerebral artery occlusion.
Nonetheless, it is unsurprising that a more specific brain ischemia can induce anxiety; one would expect that, since mood arises primarily from the brain, it would be possible to induce an anxious mood through some kind of brain lesion. This does not contradict the separate conclusion that reduced vascular flow is not typically associated with such symptoms.
Conclusion
Experimentation with mouse models of impaired vascular flow has uncovered a range of deleterious behavioral effects stemming from this condition, including depressive-like symptoms, impaired working memory, and possible spatial memory and locomotor issues. This research aids in the understanding of and development of treatments for cerebrovascular disease in human patients.
References
- World Health Organisation. The global burden of cerebrovascular disease. Thomas Truelsen, Stephen Begg, Colin Mathers. [ONLINE]. Available at: http://www.who.int/healthinfo/statistics/bod_cerebrovasculardiseasestroke.pdf. Accessed: 7 September 2018.
- Zuloaga, K. L., Zhang, W., Yeiser, L. A., Stewart, B., Kukino, A., Nie, X., … Alkayed, N. J. (2015). Neurobehavioral and Imaging Correlates of Hippocampal Atrophy in a Mouse Model of Vascular Cognitive Impairment. Translational Stroke Research, 6(5), 390–398.
- Hattori, Y., Enmi, J., Iguchi, S., Saito, S., Yamamoto, Y., Tsuji, M., … Ihara, M. (2016). Gradual Carotid Artery Stenosis in Mice Closely Replicates Hypoperfusive Vascular Dementia in Humans. Journal of the American Heart Association: Cardiovascular and Cerebrovascular Disease, 5(2), e002757.
- Coltman R1, Spain A, Tsenkina Y, Fowler JH, Smith J, Scullion G, Allerhand M, Scott F, Kalaria RN, Ihara M, Daumas S, Deary IJ, Wood E, McCulloch J, Horsburgh K. Selective white matter pathology induces a specific impairment in spatial working memory. Neurobiol Aging. 2011 Dec;32(12):2324.e7-12.
- Füchtemeier, M., Brinckmann, M. P., Foddis, M., Kunz, A., Po, C., Curato, C., … Farr, T. D. (2015). Vascular change and opposing effects of the angiotensin type 2 receptor in a mouse model of vascular cognitive impairment. Journal of Cerebral Blood Flow & Metabolism, 35(3), 476–484.
- Santoshkumar Tota, Himani Awasthi, Pradeep Kumar Kamat, Chandishwar Nath, Kashif Hanif. Protective effect of quercetin against intracerebral streptozotocin induced reduction in cerebral blood flow and impairment of memory in mice. Behavioural Brain Research. 209. 2010. 73–79.
- Shibata M, Yamasaki N, Miyakawa T, Kalaria RN, Fujita Y, Ohtani R, Ihara M, Takahashi R, Tomimoto H. Selective impairment of working memory in a mouse model of chronic cerebral hypoperfusion. Stroke. 2007 Oct;38(10):2826-32.
- Hattori Y, Kitamura A, Tsuji M, Nagatsuka K, Ihara M. Motor and cognitive impairment in a mouse model of ischemic carotid artery disease. Neurosci Lett. 2014 Oct 3;581:1-6.
- Jiwa NS, Garrard P, Hainsworth AH. Experimental models of vascular dementia and vascular cognitive impairment: a systematic review. J Neurochem. 2010 Nov;115(4):814-28.
- Liu, S., Han, S., Dai, Q., Li, S., & Li, J. (2018). BICAO-induced ischaemia caused depressive-like behaviours and caspase-8/-9-dependent brain regional neural cell apoptosis in mice. Stroke and Vascular Neurology, 3(1), 1–8.
- Vahid-Ansari, F., Lagace, D. C., & Albert, P. R. (2016). Persistent post-stroke depression in mice following unilateral medial prefrontal cortical stroke. Translational Psychiatry, 6(8), e863–.
- Hernández-Jime ́nez M, Peña-Mart ́ınez C, Godino MdC, D ́ıaz-Guzma ́n J, Moro MA ́, Lizasoain I. 2017. Test repositioning for functional assessment of neurological outcome after experimental stroke in mice. PLoS ONE 12(5): e0176770.